Revision Arthroplasty with Tibial Bone Loss: Bone Grafting Solutions
Key Takeaway
Looking for accurate information on Revision Arthroplasty with Tibial Bone Loss: Bone Grafting Solutions? Tibial bone loss is a complex issue in revision knee arthroplasty, presenting as significant bone defects that challenge implant stability. Successful management of **arthroplasty with tibial bone** loss utilizes methods like cement fill, metal augments, or bone grafting to restore anatomical support. Smaller defects are addressed with morselized graft, while larger ones may necessitate metallic wedges or structural allografts.
Introduction and Epidemiology
Substantial bone loss and complex osseous defects represent some of the most formidable challenges encountered by orthopedic surgeons performing revision total knee arthroplasty (TKA). As the volume of primary TKA procedures continues to rise globally, the corresponding burden of revision arthroplasty has increased proportionally. Tibial bone loss in the setting of a failed TKA is a particularly complex problem, demanding meticulous preoperative planning, advanced reconstructive techniques, and a thorough understanding of implant biomechanics.

Awareness and proper management of metaphyseal and diaphyseal bone loss are crucial for achieving immediate mechanical stability and ensuring the long-term longevity of the newly implanted revision components. While modern highly porous metallic augments, cones, and sleeves have gained popularity, bone grafting—both morselized and structural—remains a fundamental biologic solution, particularly for restoring bone stock in younger patients or those with catastrophic osseous deficiency.
Pathogenesis of Tibial Bone Loss
The etiology of bone stock deficiency following TKA is multifactorial, often representing a combination of biologic and mechanical failure modes.

- Stress Shielding: The presence of rigid metallic implants alters the normal load-sharing biomechanics of the proximal tibia. Stress shielding of the proximal tibia and distal femur can cause clinically significant osteopenia surrounding the knee, rendering the remaining bone structurally inadequate upon implant extraction.
- Osteolysis: Particulate wear debris (predominantly polyethylene, but also polymethylmethacrylate or metal) triggers a macrophage-mediated biologic response. The subsequent release of inflammatory cytokines (e.g., TNF-alpha, IL-1, IL-6) upregulates osteoclastic activity, resulting in progressive, often asymptomatic, periprosthetic bony destruction.
- Aseptic Implant Loosening: Failure of initial fixation or late debonding results in pathologic micromotion at the implant–bone interface. This micromotion acts as a fluid pump, driving wear debris deeper into the effective joint space and promoting the formation of a biologically active, osteolytic membrane.
- Iatrogenic Bone Loss: Removal of well-fixed implants, even when executed using proper microsurgical techniques, inevitably results in some degree of bone loss, particularly from the critical subchondral and metaphyseal regions.
- Infection: Periprosthetic joint infection (PJI) induces aggressive osteomyelitis and enzymatic degradation of the bone-cement interface, often leading to rapid and massive cavitary defects.
Natural History of Aseptic Loosening
Regardless of the initial mechanism of bone loss, once significant bony destruction becomes radiographically visible, the natural history is one of inexorable progression. In this downward spiral toward catastrophic implant failure, patients may remain asymptomatic during the early stages of osteolysis. However, as mechanical stability is compromised, pain, swelling, and instability ensue. Hyperextension or progressive varus/valgus thrust due to loss of tibial height and collateral ligament incompetence are expected sequelae of a failing TKA with significant tibial bone loss.
Surgical Anatomy and Biomechanics
Tibial Plateau Topography and Bone Quality
Tibial bone loss during revision TKA follows predictable anatomical patterns dictated by the initial primary resection, the mechanism of failure, and the inherent bone density of the proximal tibia. The most common areas of structural deficiency involve the posterolateral and medial tibial plateaus.

The proximal tibial metaphysis consists of a thin cortical shell surrounding a cancellous core. In the setting of revision arthroplasty, this cancellous core is frequently absent, replaced by fibrous tissue, cement mantles, or osteolytic cysts.

Understanding the zonal anatomy of the tibia is critical for reconstructive planning:
* Zone 1 (Epiphysis/Joint Line): Often heavily compromised or entirely absent in revision scenarios.
* Zone 2 (Metaphysis): The primary site of cavitary and uncontained defects. Restoration of this zone is the primary goal of structural bone grafting or metaphyseal cones.
* Zone 3 (Diaphysis): Typically preserved and serves as the ultimate anchor point for diaphyseal engaging stems to bypass proximal structural deficits.
Biomechanics of Tibial Component Stability
The fundamental biomechanical goal in revision TKA with severe bone loss is achieving rigid initial fixation to allow for biologic incorporation of bone grafts and to prevent early mechanical failure.

When structural allografts are utilized to reconstruct uncontained defects, the construct must be protected from excessive shear and compressive forces. This necessitates the use of diaphyseal engaging stems. The biomechanical principle dictates that the stem must bypass the most distal aspect of the bone defect or structural graft by a minimum of two cortical diameters to adequately transfer stress away from the healing host-graft junction and prevent cantilever bending forces from inducing graft fracture or nonunion.
Indications and Contraindications
Clinical Evaluation and Symptomatology
Patient history and physical findings in the setting of aseptic loosening and bone loss can range from completely asymptomatic (identified only on surveillance radiography) to debilitating pain and gross instability during normal ambulation.

A comprehensive history and physical examination are mandatory. The assessment must delineate the type, quality, location, and duration of pain. Any new, severe, or progressive pain in a previously well-functioning implant, particularly during weight-bearing, is a red flag for mechanical loosening or infection. Eliciting a history of "start-up pain" (pain upon initiating movement that improves with continued ambulation) is classic for aseptic loosening.
Instability must be carefully evaluated. A new onset of slowly progressive "giving way" or knee weakness often indicates asymmetric bone collapse leading to collateral ligament laxity. On physical examination, local tenderness along the interface between the tibial implant and the tibia is highly suggestive of tibial component loosening, though this must be clinically distinguished from localized pes anserine bursitis.
Crucially, as with all patients undergoing revision knee replacement, periprosthetic joint infection must be definitively ruled out prior to committing to a single-stage reconstruction with massive bone grafting.
Operative Versus Non Operative Management
| Indication / Contraindication | Operative Management (Revision TKA with Bone Grafting) | Non-Operative Management |
|---|---|---|
| Primary Indications | Aseptic loosening with progressive bone loss; Symptomatic osteolysis; Gross instability due to asymmetric bone collapse; PJI (as part of a two-stage exchange). | Asymptomatic, non-progressive radiolucencies; High-risk surgical candidates (severe cardiopulmonary comorbidities). |
| Defect Characteristics | AORI Type 2 or 3 defects requiring structural support; Contained cavitary defects amenable to impaction grafting. | Small, stable, contained defects in asymptomatic patients (surveillance only). |
| Patient Factors | Younger patients where restoration of bone stock is desirable for potential future revisions; Adequate soft tissue envelope. | Active systemic infection (absolute contraindication for single-stage allografting); Severe peripheral vascular disease; Charcot arthropathy (relative). |
| Contraindications | Active, uncontrolled PJI (requires staged approach, structural allograft contraindicated in stage 1); Inadequate extensor mechanism (relative). | N/A |
Pre Operative Planning and Patient Positioning
Imaging and Diagnostic Modalities
A thorough clinical and radiographic evaluation is an absolute prerequisite for successful revision TKA. The surgeon must determine the extent and location of bone loss, the quality of the remaining host bone, the degree of cortical continuity, and the anatomical joint line.

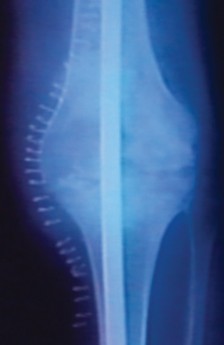
Standing anteroposterior (AP), lateral, and patellar (Merchant) radiographs are the initial standard for assessing tibial bone loss. These provide a baseline understanding of component migration, osteolysis, and gross alignment.

However, plain radiography frequently underestimates the true volumetric extent of osteolysis. Computed Tomography (CT) scans, particularly with metal artifact reduction sequence (MARS) protocols, are significantly more accurate in estimating the degree of bone loss.

Three-dimensional (3D) CT reconstructions are invaluable when dealing with massive, uncontained AORI Type 3 defects or highly abnormal anatomy, allowing the surgeon to conceptualize the exact dimensions of the structural allograft required.

Preoperative templating is mandatory. It dictates the anticipated size of the revision components, the length and diameter of the diaphyseal stems required to bypass defects, and the volume or size of the bone graft necessary to restore the joint line and metaphyseal support.
Infection Workup and Laboratory Analysis
All patients scheduled for revision TKA must undergo a rigorous infection workup. Standard laboratory studies include a complete blood count (CBC), serum C-reactive protein (CRP), and erythrocyte sedimentation rate (ESR).
Furthermore, an attempt at knee aspiration is mandatory. Synovial fluid must be sent for Gram stain, total nucleated cell count, differential (neutrophil percentage), and aerobic/anaerobic cultures. If the initial aspiration is dry or inconclusive, serial aspirations or the measurement of synovial biomarkers (e.g., alpha-defensin) should be utilized. The presence of infection fundamentally alters the surgical algorithm, typically necessitating a two-stage approach and precluding the immediate use of massive structural allografts.
Patient Positioning and Operating Room Setup
The patient is positioned supine on a radiolucent operating table to facilitate intraoperative fluoroscopy. A proximal thigh tourniquet is applied but typically inflated only during cementation to minimize ischemic time, unless excessive bleeding obscures the surgical field during exposure. A robust leg holder or a sterile bump is utilized to allow the knee to be flexed past 90 degrees and hyperflexed during component extraction and tibial preparation.
The operating room must be equipped with a comprehensive array of revision instruments, including extraction devices, flexible osteotomes, high-speed burrs, diaphyseal reamers, and an extensive selection of bone grafts (morselized cancellous chips, femoral head allografts, and distal femoral structural allografts).
Detailed Surgical Approach and Technique
Surgical Exposure and Component Removal
The surgical approach typically utilizes the previous midline skin incision. A standard medial parapatellar arthrotomy is performed. In the setting of a stiff knee or extensive scarring, the surgeon must be prepared to utilize extensile exposures to prevent catastrophic avulsion of the extensor mechanism. Options include a quadriceps snip, V-Y quadricepsplasty, or an extended tibial tubercle osteotomy (TTO). A TTO is particularly advantageous in revision scenarios as it drastically improves exposure of the proximal tibia and facilitates the safe extraction of well-fixed long stems.

Component removal must be executed with extreme patience and precision to preserve the remaining host bone stock.

The implant-bone or implant-cement interface is systematically disrupted using thin, flexible osteotomes, oscillating microsaws, and Gigli saws. Extraction slaps or customized removal tools are utilized only after the interfaces have been fully mobilized to prevent iatrogenic fracture or further uncontained bone loss.
Defect Classification and Preparation
Following component extraction, meticulous debridement of all particulate debris, fibrous tissue, and residual polymethylmethacrylate (PMMA) cement is performed.

The osseous defects are then objectively assessed and classified. The Anderson Orthopaedic Research Institute (AORI) classification is the most widely utilized system for categorizing bone defects in revision TKA:
* AORI Type 1: Intact metaphyseal bone, minor contained defects. Joint line is preserved.
* AORI Type 2: Damaged metaphyseal bone requiring reconstruction.
* Type 2A: Involves one condyle (medial or lateral).
* Type 2B: Involves both condyles.
* AORI Type 3: Deficient metaphyseal segment compromising the proximal diaphysis, often associated with collateral ligament detachment.

Smaller contained defects (AORI Type 1) can often be addressed with morselized bone graft or cement alone. Larger, uncontained defects (AORI Type 2 and 3) require the use of metallic wedges, highly porous cones/sleeves, or structural allografts.
Management of Contained Defects with Morselized Graft
For contained cavitary defects where the peripheral cortical rim remains intact, impaction bone grafting is a highly effective biologic solution.

The sclerotic neo-membrane lining the cyst or cavity must be aggressively curetted or burred to expose punctate bleeding host bone, which is essential for graft incorporation.

Fresh-frozen or freeze-dried cancellous allograft is milled into small morsels (typically 3-5mm in diameter). The graft is sequentially introduced into the defect and vigorously compacted using specialized tamps and a mallet. This impaction process creates a dense, mechanically stable bed capable of supporting a revision tibial tray or augment. If the defect has a small uncontained cortical blowout, a wire mesh can be secured to the periphery to convert the defect into a contained cavity prior to impaction grafting.
Management of Uncontained Defects with Structural Allograft
Uncontained defects (AORI Type 2 and 3) lack a supportive peripheral rim and require structural support to transfer loads from the tibial tray to the host diaphysis. While metallic cones are increasingly popular, structural allografting remains an excellent option, particularly for massive defects or when restoration of bone stock is prioritized.

Femoral head allografts are typically utilized for Type 2 defects, while massive distal femoral or proximal tibial allografts may be required for Type 3 defects.
The host bone bed is prepared by resecting sclerotic bone to create a flat, bleeding surface. Step-cuts or oblique resections are often utilized to maximize the contact area between the host and the graft.

The structural allograft is meticulously shaped using an oscillating saw and high-speed burr to perfectly match the host defect. The cartilage is completely removed from the allograft to expose subchondral bone.

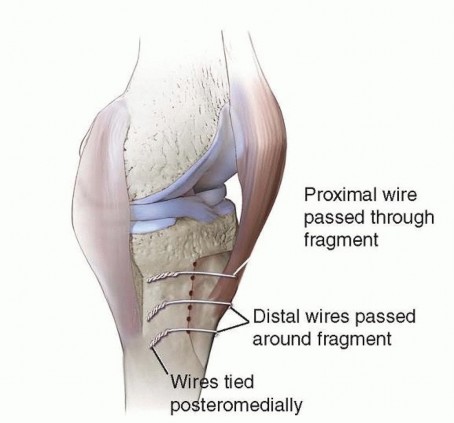
Once a precise fit is achieved, the graft is provisionally stabilized with Kirschner wires and definitively fixed to the host tibia using fully threaded cancellous lag screws or cortical screws directed from the graft into the host bone. The screws must be countersunk to avoid interfering with the seating of the tibial baseplate.
Implant Selection and Stem Fixation
Following graft fixation, the medullary canal is reamed to accommodate a diaphyseal engaging stem.

Trialing is performed to assess alignment, joint line restoration, and ligamentous stability. The tibial tray must be supported by host bone for at least 50% of its surface area whenever possible, though massive allografts may require the tray to rest entirely on the graft construct.

The definitive implant is then impacted into place. When a structural allograft is utilized, a diaphyseal stem is absolutely mandatory. The stem serves to bypass the graft-host junction, transferring axial and shear loads directly to the intact diaphyseal bone (Zone 3 fixation). Stems can be cemented or uncemented (press-fit). Uncemented fluted stems are frequently preferred as they provide rigid initial stability without the risk of cement interposition at the graft-host interface, which could inhibit biologic incorporation.
Complications and Management
Revision TKA with massive bone grafting is a complex procedure associated with a significantly higher complication profile than primary arthroplasty.
Intraoperative and Postoperative Complications
| Complication | Incidence / Risk Factors | Prevention and Management Strategies |
|---|---|---|
| Graft Resorption | Higher in massive structural allografts over time; Immunologic response or stress shielding. | Ensure rigid diaphyseal stem fixation to prevent micromotion; optimize host-graft contact area. Late severe resorption may require re-revision with metallic cones. |
| Graft Nonunion | 5-10% in structural grafts; Inadequate fixation, smoking, poor host bone vascularity. | Meticulous preparation of bleeding host bed; rigid screw fixation of the graft; strict adherence to postoperative weight-bearing restrictions. |
| Periprosthetic Infection (PJI) | 3-8% in revision settings; Prolonged operative time, massive allograft use (avascular tissue). | Rigorous preoperative screening; meticulous sterile technique; copious irrigation; extended postoperative antibiotic prophylaxis; consider antibiotic-loaded cement for the articular surface. |
| Intraoperative Fracture | High risk during component extraction or diaphyseal reaming of osteopenic bone. | Prophylactic cerclage wiring of the tibia prior to reaming or impaction; use of flexible osteotomes; extended exposures (TTO) to reduce tension. |
| Aseptic Loosening | Secondary to graft failure, inadequate stem bypass, or progressive osteolysis. | Ensure stem bypasses the defect by at least 2 cortical diameters; achieve rigid Zone 3 diaphyseal engagement. |

Graft resorption is a known long-term complication of massive structural allografts. While the graft provides immediate mechanical support, the biologic creeping substitution is often incomplete, leading to late structural collapse if the diaphyseal stem fails to adequately offload the metaphyseal segment.

Nonunion at the host-graft interface typically presents with recurrent pain and progressive radiolucent lines. Management often requires re-revision, removal of the un-incorporated graft, and substitution with highly porous metaphyseal cones or sleeves.

Periprosthetic joint infection remains the most
Clinical & Radiographic Imaging



























You Might Also Like