Proximal Tibia Resection With Endoprosthetic Reconstruction
Comprehensive Introduction and Patho-Epidemiology
The surgical management of primary and secondary osseous malignancies of the proximal tibia represents one of the most formidable challenges in orthopedic oncology. Historically, the proximal tibia was considered an anatomic site where limb-sparing surgery was nearly impossible, and above-knee amputations were the standard of care for malignant tumors such as osteosarcoma and Ewing sarcoma. The proximal tibia is widely acknowledged as the most complicated anatomic location for major bone resections and endoprosthetic reconstructions. This complexity arises from three primary factors: the profound lack of muscular coverage along the anteromedial aspect of the tibia, the relatively small caliber and intimate proximity of the major neurovascular structures of the leg, and the absolute necessity of resecting and subsequently reconstructing the insertion site of the extensor mechanism to maintain a functional extremity.
Advances in neoadjuvant chemotherapy, high-resolution cross-sectional imaging, and modular endoprosthetic design have catalyzed a paradigm shift, making limb salvage the standard of care for the vast majority of proximal tibial sarcomas. However, despite these advancements, the proximal tibia remains the site associated with the highest rates of post-operative complications and the poorest relative functional outcomes compared to other major long bone resections, such as the distal femur or proximal humerus. The thin, vulnerable soft tissue envelope predisposes patients to wound dehiscence and deep periprosthetic joint infections, which frequently necessitated secondary amputations in the early eras of limb salvage.
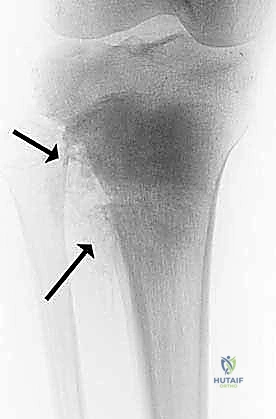
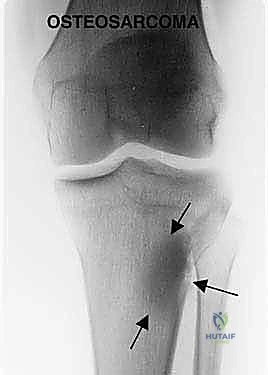
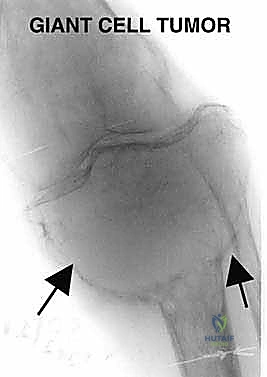
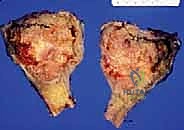
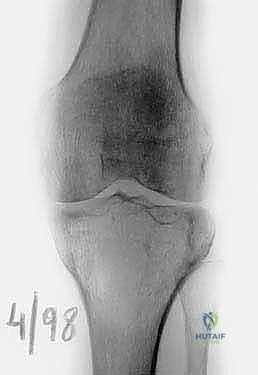
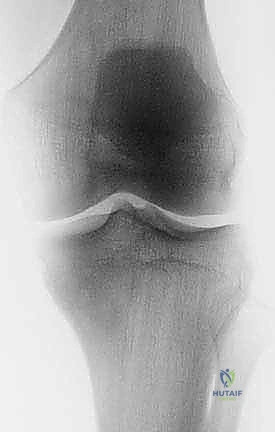
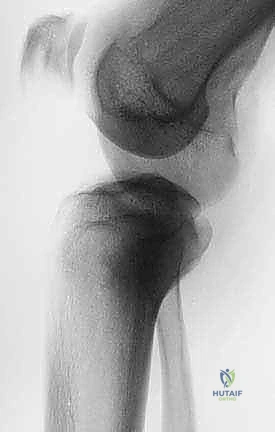
The patho-epidemiology of proximal tibial tumors is dominated by primary bone sarcomas, predominantly osteosarcoma, which exhibits a bimodal age distribution peaking in adolescence and again in late adulthood. The metaphysis of the proximal tibia is the second most common site for osteosarcoma, following the distal femur. Other pathologies frequently encountered include Ewing sarcoma, chondrosarcoma, and aggressive benign lesions such as Campanacci Grade III giant cell tumors (GCTs) that present with extensive cortical destruction and metaphyseal-epiphyseal ballooning. Metastatic carcinomas, particularly from renal, thyroid, and lung primaries, may also present with massive destructive lesions in this region, necessitating en bloc resection and reconstruction for local tumor control and functional restoration.
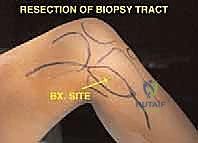
A critical tenet of the patho-epidemiology in this region is the mechanism of local tumor extension. Tumors of the proximal tibia rarely invade the knee joint directly due to the robust barrier provided by the articular cartilage and the physeal plate in skeletally immature patients. When intra-articular invasion does occur, it is almost invariably iatrogenic or traumatic in nature—most commonly resulting from a pathologic fracture, capsular contamination due to an improperly placed biopsy tract, or direct tumor extension along the cruciate ligaments. Consequently, adherence to strict oncologic principles during the initial biopsy, ensuring the tract is placed entirely within the planned resection field, is paramount to avoiding catastrophic joint contamination that would mandate an extra-articular resection or amputation.
Detailed Surgical Anatomy and Biomechanics
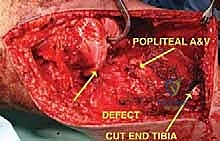
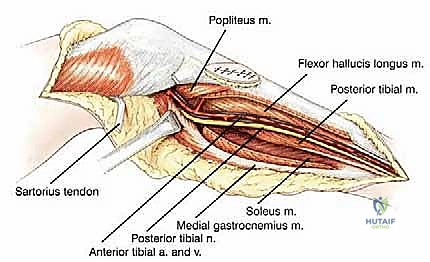
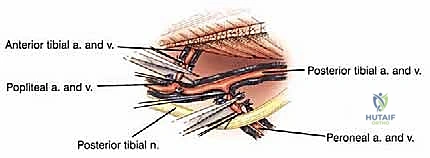
A profound, three-dimensional understanding of the surgical anatomy of the popliteal fossa and the proximal leg is non-negotiable for the orthopedic oncologist. The vascular anatomy is defined by the popliteal trifurcation, which is, in reality, composed of two distinct bifurcations. The popliteal artery descends through the popliteal fossa and gives rise to its first major branch, the anterior tibial artery, precisely at the inferior border of the popliteus muscle. Following this first bifurcation, the main vessel continues as the tibioperoneal trunk. The second bifurcation occurs slightly distal to the anterior tibial takeoff, where the tibioperoneal trunk divides into the posterior tibial artery and the peroneal artery. During a proximal tibia resection, it is almost always an anatomic necessity to ligate the anterior tibial artery to mobilize the neurovascular bundle away from the posterior cortex of the tibia and the tumor mass.
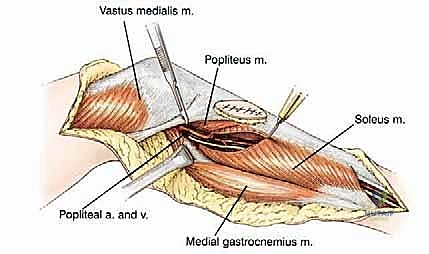
The soft tissue anatomy of the posterior proximal tibia provides a unique and fortuitous oncologic boundary. The popliteus muscle drapes intimately over the posterior surface of the proximal tibia, serving as a robust, natural anatomical barrier between the posterior intraosseous or extraosseous extension of the tumor and the critical neurovascular bundle of the lower extremity. This anatomic reality is in stark contrast to the distal femur, where the posterior aspect of the bone is covered solely by thin popliteal fat, offering minimal resistance to posterior tumor extension. The presence of an intact popliteus muscle often allows the surgeon to achieve a negative microscopic margin while safely dissecting the popliteal vessels off the posterior tumor mass.
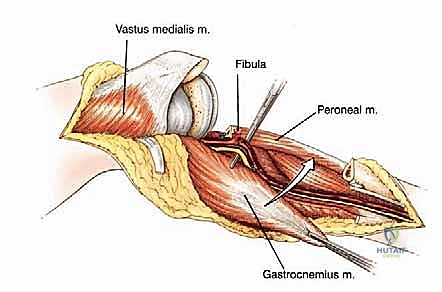
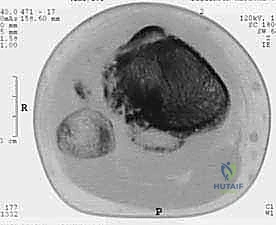
The proximal tibiofibular joint presents another critical anatomic consideration. Situated closely to the posterolateral aspect of the proximal tibia, this synovial joint is frequently implicated in tumor extension. Histologic and advanced imaging studies have consistently demonstrated that sarcomas involving the proximal tibia have a remarkably high incidence of microscopic and macroscopic extension into the periscapular tissues of the tibiofibular joint. Consequently, to obtain a satisfactory, wide surgical margin, it is a routine and mandatory procedure for all high-grade sarcomas of the proximal tibia to resect the proximal tibiofibular joint en bloc with the primary specimen, constituting an extra-articular resection of this specific joint. The peroneal nerve, which wraps around the fibular neck, must be meticulously identified, neurolysed, and preserved during this maneuver.
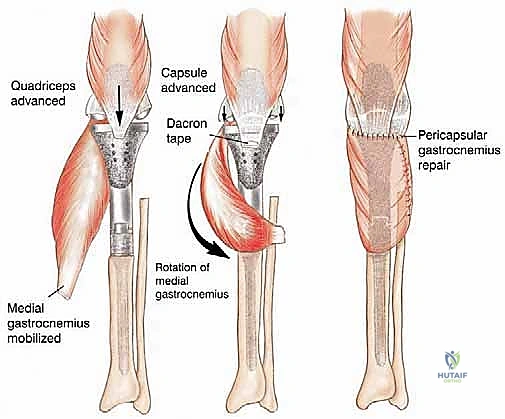
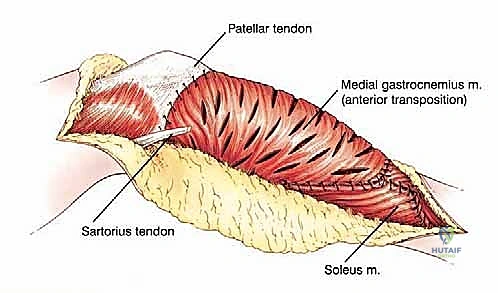
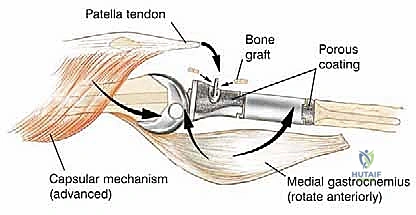
Biomechanically, the proximal tibia serves as the insertion site for the entire extensor mechanism of the lower extremity via the patellar tendon's attachment to the tibial tuberosity. Resection of the proximal one-half to two-thirds of the tibia mandates the en bloc removal of this insertion site. The extensor mechanism is subjected to massive tensile forces during normal ambulation, stair climbing, and rising from a seated position. Therefore, the biomechanical reconstruction of this mechanism is arguably the most critical determinant of the patient's ultimate functional outcome. Failure to achieve rigid, biologic fixation of the extensor mechanism to the endoprosthesis results in a profound extensor lag, rendering the limb functionally inferior to an above-knee amputation. This biomechanical imperative necessitates the routine use of a medial gastrocnemius rotational flap, which not only provides robust soft tissue coverage over the metallic prosthesis but also serves as a vascularized, dynamic tissue bed to which the patellar tendon can be securely sutured and healed.
Exhaustive Indications and Contraindications
The indications for proximal tibia resection with endoprosthetic reconstruction are primarily oncologic, dictated by the biologic behavior and anatomic extent of the lesion. The most frequent indication is the presence of a primary high-grade bone sarcoma, such as osteosarcoma or Ewing sarcoma, localized to the proximal tibia without encasement of the major neurovascular structures. Limb salvage is indicated when a wide surgical margin can be achieved while preserving a functional extremity. Additionally, aggressive benign tumors, notably Campanacci Grade III giant cell tumors that have caused massive, uncontained destruction of the meta-epiphyseal bone, are prime candidates for this procedure. In these benign-aggressive cases, joint salvage via curettage and cementation is often mechanically unfeasible or carries an unacceptably high risk of local recurrence and secondary joint collapse.
Metastatic bone disease also constitutes a significant indication, particularly for patients with solitary metastases from renal cell carcinoma, thyroid carcinoma, or breast carcinoma who present with impending or completed pathologic fractures. In the metastatic setting, the goal shifts from absolute oncologic cure to durable local control, immediate pain relief, and rapid restoration of weight-bearing capacity. Endoprosthetic reconstruction allows for immediate mechanical stability, bypassing the delayed healing and high failure rates associated with osteosynthesis in heavily irradiated or biologically compromised bone. The decision to proceed with a massive endoprosthesis in the metastatic patient must be carefully weighed against the patient's overall prognosis, life expectancy, and systemic disease burden.
Contraindications to limb-sparing surgery in the proximal tibia are stringent and must be respected to avoid catastrophic oncologic and functional failures. The absolute major contraindication is direct encasement or gross invasion of the major neurovascular bundle (the popliteal artery, tibioperoneal trunk, or tibial nerve) by the tumor mass. While resection and vascular grafting are technically feasible, the combination of a massive bone resection, vascular bypass, and compromised soft tissue coverage yields unacceptably high amputation rates and poor functional outcomes. Furthermore, extensive soft tissue tumor involvement that precludes the ability to achieve adequate soft tissue coverage of the prosthesis—even with the utilization of regional or free flaps—is a strong relative, if not absolute, contraindication.
Other critical contraindications include the presence of an intractable, deep-seated infection within the tumor bed, which absolutely precludes the implantation of a massive metallic endoprosthesis. In cases of massive intra-articular contamination—either from a poorly planned biopsy, a large intra-articular pathologic fracture, or extensive tumor tracking along the cruciate ligaments—a standard intra-articular resection is contraindicated. In such scenarios, if limb salvage is still attempted, a highly complex extra-articular resection of the entire knee joint (en bloc removal of the proximal tibia, joint capsule, and distal femur) must be performed, though this significantly increases the morbidity and complexity of the reconstruction.
| Category | Indications | Contraindications |
|---|---|---|
| Primary Malignancies | Enneking Stage IIA/IIB Osteosarcoma, Ewing Sarcoma, Chondrosarcoma | Major neurovascular bundle encasement |
| Benign-Aggressive | Campanacci Grade III Giant Cell Tumor, massive Aneurysmal Bone Cyst | Intractable deep periprosthetic or tumor bed infection |
| Metastatic Disease | Solitary metastasis with massive bone destruction/pathologic fracture | Extensive soft tissue involvement precluding flap coverage |
| Anatomic/Local | Contained extra-osseous extension, clear margins attainable | Massive intra-articular contamination (relative contraindication for standard resection; requires extra-articular approach) |

Pre-Operative Planning, Templating, and Patient Positioning
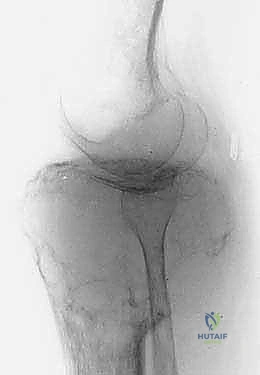
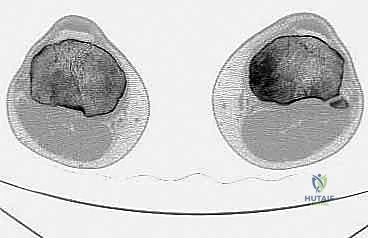
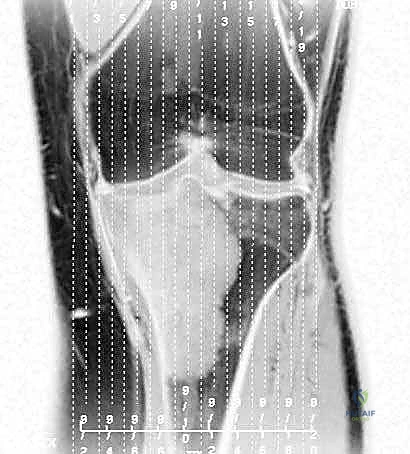
Thorough pre-operative evaluation and meticulous surgical planning are the cornerstones of a successful proximal tibia resection and reconstruction. The imaging protocol must be exhaustive. Plain radiographs (anteroposterior and lateral) provide a baseline assessment of bone destruction and matrix mineralization. However, magnetic resonance imaging (MRI) of the entire tibia is the gold standard and absolutely essential for determining the precise intraosseous and extraosseous extent of the tumor. T1-weighted coronal and sagittal sequences are utilized to accurately map the proximal-to-distal intramedullary marrow involvement, which dictates the level of the tibial osteotomy. Oncologic principles dictate that the osteotomy should be planned 3 to 5 centimeters distal to the furthest extent of the intramedullary tumor signal. Furthermore, whole-bone MRI is critical for identifying skip lesions, the presence of which would drastically alter the level of resection or necessitate a total tibial replacement.
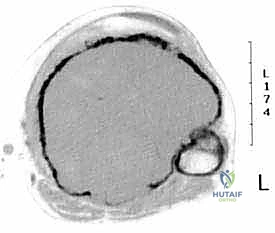
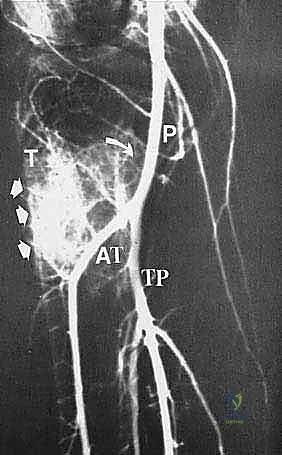
Computed tomography (CT) of the limb is highly synergistic with MRI, providing superior resolution of cortical bone integrity, subtle pathologic fractures, and the three-dimensional architecture of the remaining host bone. CT is particularly useful for assessing the integrity of the proximal tibiofibular joint and the degree of periscapular tissue involvement. In conjunction with cross-sectional imaging, biplane angiography remains a highly valuable tool, particularly when MRI or CT reveals significant posterior soft tissue extension. The lateral angiographic view is essential for evaluating the interval between the posterior cortex of the tibia and the main neurovascular bundle. A clear interval, often maintained by an intact popliteus muscle, suggests that a safe resection margin can be achieved. Angiography also confirms the patency of the posterior tibial and peroneal arteries, which is critical knowledge given that the anterior tibial artery will almost certainly be ligated during the resection.
Digital and physical templating based on calibrated radiographs and CT scout films is a mandatory pre-operative step. The surgeon must determine the precise length of the required bone resection and select the appropriate modular endoprosthetic components to restore the patient's native leg length and joint line. The diameter of the distal tibial diaphysis must be measured to select the correct intramedullary stem size. Given the narrow medullary canal of the tibial diaphysis, custom or ultra-short stems may occasionally be required. The surgeon must also ensure that the chosen implant system possesses the necessary modularity to accommodate intra-operative adjustments and provides a reliable mechanism for reattaching the extensor mechanism, such as a porous metal segment or specialized suturing holes.
Patient positioning and operating room setup must be optimized for a complex, multi-hour procedure. The patient is positioned supine on a radiolucent operating table to facilitate unimpeded intra-operative fluoroscopy. A sterile pneumatic tourniquet is applied to the proximal thigh, though it is often kept uninflated during the initial vascular dissection to allow for accurate identification of pulsatile vessels; it may be inflated later during the bone resection and cementing phases to minimize blood loss. The entire lower extremity, from the toes to the groin, is prepped and draped free to allow for full manipulation of the knee and hip joints, assessment of limb alignment, and harvesting of distal autograft if necessary. A bump is placed under the ipsilateral hip to internally rotate the leg to a neutral position, facilitating the anteromedial surgical approach.

Step-by-Step Surgical Approach and Fixation Technique
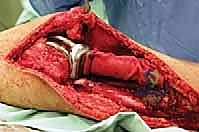
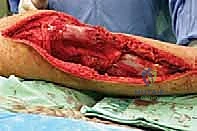
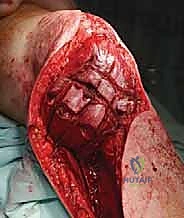
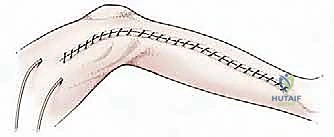
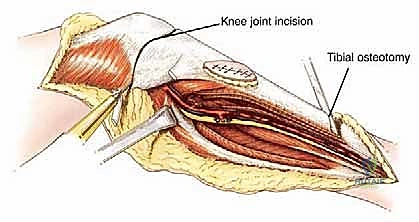
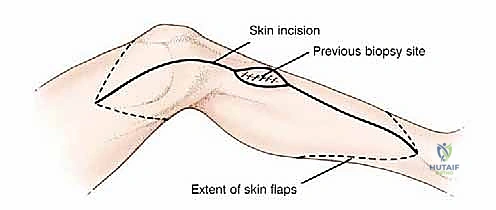
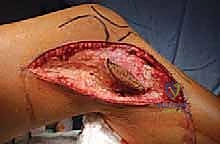
The surgical execution of a proximal tibia resection with endoprosthetic reconstruction is logically divided into three major phases: the oncologic resection, the prosthetic reconstruction of the skeletal defect, and the soft tissue/extensor mechanism reconstruction. The procedure commences with a single, extensile anteromedial incision. This incision begins proximally at the distal third of the femur, traverses the medial aspect of the patella, and extends distally to the middle or distal third of the tibia, depending on the planned osteotomy level. It is imperative that the previous biopsy tract is elliptically excised en bloc with the surgical specimen, maintaining a margin of at least 2 centimeters of healthy skin around the tract. Medial and lateral fasciocutaneous flaps are raised; these flaps must be kept deliberately thick, incorporating the deep fascia, to preserve the subdermal vascular plexus and minimize the profound risk of post-operative flap necrosis.
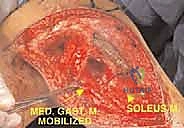
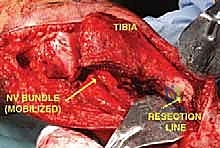
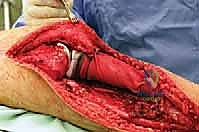
Following flap elevation, the critical phase of vascular exploration and detachment of the neurovascular bundle begins. The popliteal fossa must be explored early in the procedure to definitively determine resectability, particularly for tumors with large posterior extraosseous components. The popliteal space is accessed medially by detaching the medial head of the gastrocnemius muscle from its femoral origin and splitting the soleus muscle along its tibial attachment. By applying gentle posterior traction proximal to the popliteal artery, the surgeon can visualize the critical takeoff of the anterior tibial artery and its accompanying venae comitantes as they pass anteriorly through the interosseous membrane. The anterior tibial vessels are meticulously isolated, ligated, and divided. This crucial maneuver releases the tethering of the neurovascular bundle, allowing the popliteal artery and tibioperoneal trunk to fall away posteriorly, safely distancing them from the posterior aspect of the tibia and the tumor mass.
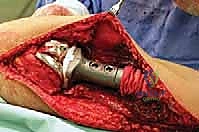
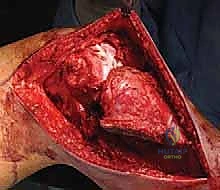
Once the neurovascular bundle is protected, the en bloc resection proceeds. The patellar tendon is sharply detached from the tibial tuberosity, often leaving a small wafer of bone attached if oncologically safe, to facilitate later repair. The proximal tibiofibular joint is addressed via an extra-articular resection; the fibula is osteotomized distal to the joint, and the peroneal nerve is carefully neurolysed and retracted laterally. The tibial diaphysis is then exposed at the pre-operatively templated level, and a transverse osteotomy is performed using an oscillating saw. The knee joint capsule is incised circumferentially, and the cruciate ligaments are transected at their femoral origins. If pre-operative MRI indicated cruciate involvement, the resection must be modified to an extra-articular knee resection, taking the femoral condyles en bloc. The entire proximal tibia, along with the popliteus muscle, proximal fibula, and surrounding tumor pseudocapsule, is removed as a single specimen and sent for margin analysis.
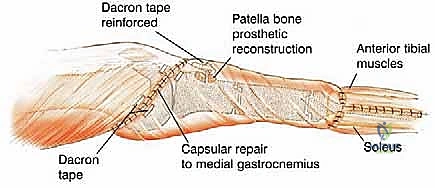
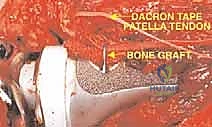
The reconstruction phase begins with the preparation of the tibial diaphysis and distal femur. The medullary canal of the tibia is sequentially reamed to accommodate the selected prosthetic stem. The distal femur is prepared with standard cutting jigs to accept the femoral component of the rotating hinge knee mechanism. The tibial stem is typically cemented using highly viscous, antibiotic-loaded polymethylmethacrylate (PMMA) bone cement, utilizing modern cementing techniques including pulsatile lavage, a distal cement restrictor, and retrograde filling. Once the cement has cured, the modular intercalary segments are assembled to restore the exact length of the resected bone, and the rotating hinge mechanism is linked.
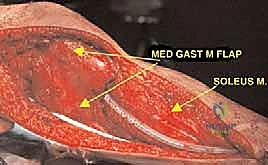
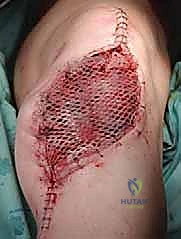
The final, and arguably most critical, step is the soft tissue reconstruction. The entire medial aspect of the metallic tibia lies in a subcutaneous location, posing an extreme risk of infection and wound breakdown. Furthermore, the extensor mechanism must be rigidly reconstructed. Today, the routine transfer of the medial gastrocnemius muscle anteriorly is the gold standard. The muscle belly is mobilized, rotated anteriorly, and draped over the exposed prosthesis. The patellar tendon is then securely sutured into the robust tendinous insertion of the gastrocnemius flap and, depending on the implant design, directly into a porous metal segment or specialized synthetic mesh attached to the prosthesis. This vital flap provides immediate, vascularized soft tissue coverage, obliterates dead space, and provides a dynamic, biologic bed for extensor mechanism healing, drastically reducing the incidence of infection and secondary amputation.
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, proximal tibia endoprosthetic reconstruction is burdened by a high complication profile, necessitating vigilant post-operative surveillance and a readiness to intervene. The most devastating early-to-intermediate complication is deep periprosthetic joint infection (PJI). Due to the massive hardware volume, extensive soft tissue dissection, and the immuno-compromised state of patients undergoing neoadjuvant chemotherapy, infection rates historically approached 20-30%, though the routine use of gastrocnemius flaps has reduced this to approximately 10-15%. Acute infections require aggressive debridement, antibiotics, and implant retention (DAIR) protocols, often supplemented by additional soft tissue coverage. Chronic or recalcitrant infections mandate a two-stage revision protocol, utilizing an articulating or static antibiotic-loaded cement spacer, followed by re-implantation once the infection is eradicated. In cases of intractable infection with massive soft tissue loss, above-knee amputation remains the ultimate salvage procedure.
Failure of the extensor mechanism is the most common functional complication, occurring in up to 20% of patients. This presents as an inability to actively extend the knee against gravity, resulting in a profound functional deficit, an obligate antalgic gait, and an increased risk of falls. Extensor lag can result from avulsion of the patellar tendon from its reconstructed insertion, attenuation of the repair over time, or ischemic necrosis of the distal aspect of the gastrocnemius flap. Management of extensor mechanism failure is notoriously difficult. Acute avulsions may be primarily repaired and augmented with synthetic mesh (e.g., Marlex or Trevira tube) or allograft tissue. Chronic, massive failures may require complex reconstructions utilizing Achilles tendon allografts with a calcaneal bone block fixed to the tibial prosthesis, though outcomes remain guarded and many patients ultimately require permanent bracing with a drop-lock knee-ankle-foot orthosis (KAFO).
Aseptic loosening and structural implant failure represent the primary long-term complications, typically manifesting 5 to 10 years post-operatively. The rotating hinge mechanism subjects the cemented tibial stem to significant torsional and bending moments. Over time, this can lead to progressive radiolucent lines at the
Clinical & Radiographic Imaging Archive