Targeted Ablation of Musculoskeletal Tumors: A Safer Approach
Key Takeaway
Here are the crucial details you must know about Targeted Ablation of Musculoskeletal Tumors: A Safer Approach. Photodynamic ablation (PDA) of musculoskeletal tumors is a treatment using a photosensitizer drug, administered systemically or locally, that accumulates in tumor cells. When illuminated with visible light, the drug excites to generate cytotoxic species, causing specific tumor cell death and destruction. This method provides an option for targeting microscopic disease remaining after surgical removal, minimizing harm to healthy tissues.
Introduction and Epidemiology
Malignant tumors of the musculoskeletal system, particularly primary bone and soft tissue sarcomas, present a formidable challenge in surgical oncology. Historically, the management of these solid tumors has necessitated a true and wide surgical resection to achieve complete extirpation and maximize disease-free survival. The oncologic concept of "wide margins," as formally established by the Enneking surgical staging system, dictates the en bloc resection of the tumor alongside a continuous, uninterrupted cuff of normal, healthy tissue surrounding the visible tumor mass and its reactive pseudocapsule. However, even radical resections do not absolutely guarantee a cure. Microscopic disease frequently extends through the reactive zone into the surrounding surgical field via satellite nodules or micrometastases, leading to local recurrence if not adequately addressed.

To mitigate the substantial risk of local recurrence, surgical management is almost universally coupled with neo-adjuvant or adjuvant treatments, predominantly external beam radiation therapy and systemic cytotoxic chemotherapy. While these modalities significantly lower the likelihood of local and systemic relapse, they carry a considerable and often dose-limiting morbidity profile. Traditional adjuvant therapies are not inherently tissue-specific; they inflict profound collateral damage on healthy surrounding tissues. This non-selective cytotoxicity leads to severe complications, including impaired wound healing, radiation-induced fibrosis, osteonecrosis, peripheral neuropathy, secondary radiation-induced malignancies, and profound systemic toxicity.
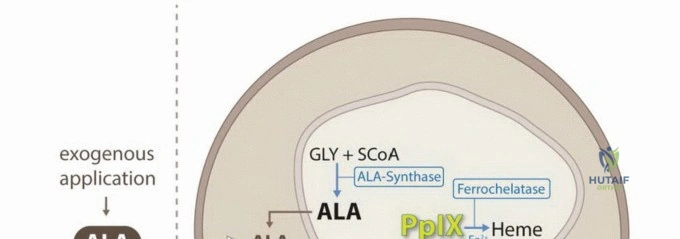
Photodynamic ablation (PDA) represents a paradigm shift in the management of microscopic residual disease in orthopedic oncology. By facilitating specific tumor cell cytotoxicity, PDA provides an elegant, targeted treatment option for eradicating the microscopic disease remaining in the surgical bed following intralesional or marginal tumor cytoreduction. Photodynamic ablation is defined by the administration of a non-toxic photosensitizer either systemically, locally, or topically. The photosensitizer exhibits a preferential affinity for neoplastic tissue due to the altered metabolic pathways, high vascular permeability, and acidic microenvironment characteristic of tumors. Following its accumulation within the target cells, the surgical site is illuminated with specific wavelengths of visible light. This illumination excites the photosensitizer, initiating a Type II photochemical reaction that generates reactive oxygen species (ROS) and highly cytotoxic singlet oxygen. These reactive molecules rapidly induce tumor cell apoptosis, necrosis, and localized microvascular thrombosis.
The fundamental realization that the combination of non-toxic dyes and visible light could eradicate cellular structures was first documented by Oscar Raab, a medical student working under Professor Herman Von Tappeiner in Munich in 1900. While investigating the effects of acridine dyes on malaria-causing Paramecia, Raab discovered that the combination of acridine red and light resulted in protozoal death. He correctly postulated that this effect was mediated by the transfer of energy from light to the chemical agent, a process analogous to botanical photosynthesis. This seminal discovery led to the first therapeutic medical applications using topical eosin combined with white light illumination to treat cutaneous malignancies. Subsequent biophysical research identified that molecular oxygen is an absolute requirement for this chain of reactions, leading to the formal coinage of the term "photodynamic action."
Surgical Anatomy and Biomechanics
The application of photodynamic ablation in orthopedic oncology requires a profound understanding of the tumor microenvironment, compartmental anatomy, and the unique optical properties of musculoskeletal tissues. Tumors of the musculoskeletal system expand radially by compressing surrounding normal tissue, creating a reactive zone or "pseudocapsule." This pseudocapsule is not a true anatomic barrier; rather, it is composed of compressed fibrous tissue, inflammatory cells, and tortuous neovasculature. In malignant lesions, this pseudocapsule is frequently breached by satellite tumor nodules, and skip metastases may exist further proximally or distally within the same anatomic compartment.
Traditional wide resection mandates the removal of this entire compartment or a significant cuff of normal tissue. In the extremities, this often necessitates the sacrifice of critical neurovascular structures, major stabilizing muscle groups, and large segments of diaphyseal or metaphyseal bone. This results in profound biomechanical deficits that fundamentally alter the load-bearing capacity and kinematics of the limb. Endoprosthetic reconstruction or massive structural allografting is subsequently required to span the intercalary or articular defect. These complex reconstructions carry inherent and significant long-term risks, including aseptic loosening, mechanical structural failure, periprosthetic fracture, and catastrophic deep periprosthetic joint infection.
Conversely, the biomechanical rationale for utilizing intralesional or marginal resection combined with photodynamic ablation is the maximal preservation of host bone and soft tissue architecture. By selectively ablating the microscopic disease within the reactive zone photochemically, the surgeon can preserve the structural integrity of the native diaphysis or metaphysis. This joint-sparing and bone-sparing approach maintains the native entheses, preserving the origin and insertion of critical stabilizing musculature, thereby vastly improving postoperative proprioception and dynamic joint stability.
Optical Properties of Musculoskeletal Tissues
The clinical efficacy of photodynamic ablation is heavily dependent on the penetration depth of light through heterogeneous tissues such as cortical bone, cancellous bone, cartilage, and striated muscle. The optical penetration depth is defined mathematically by the absorption and scattering coefficients of the target tissue, governed by the Beer-Lambert law. Cortical bone, due to its dense hydroxyapatite matrix, exhibits high scattering properties, while tissues rich in chromophores such as hemoglobin, myoglobin, and melanin exhibit high absorption.
Light in the red and near-infrared spectrum (600 nm to 800 nm) demonstrates the deepest tissue penetration due to minimal absorption by endogenous chromophores, making it the preferred wavelength for deeper volumetric ablation. However, certain photosensitizers, such as acridine orange, are optimally excited by blue light (approximately 466 nm). While blue light has a much shallower tissue penetration depth (often limited to 1-2 mm), it provides excellent superficial fluorescence for visually guiding the surgical resection of microscopic disease prior to the administration of broader-spectrum, deep-penetrating ablative light.

Indications and Contraindications
The integration of photodynamic ablation into the surgical armamentarium is primarily indicated for patients where wide resection would result in unacceptable functional morbidity, mandate an amputation, or where the complex anatomic location precludes the achievement of negative margins without catastrophic neurologic or vascular injury.
Primary Indications
Photodynamic ablation is highly advantageous in the management of locally aggressive benign bone tumors, such as Giant Cell Tumors (GCT) of bone and Aneurysmal Bone Cysts (ABC), where recurrence rates following standard mechanical curettage remain unacceptably high. In malignant pathology, it is indicated as an adjunct to marginal resection for low-grade chondrosarcomas (atypical cartilaginous tumors), parosteal osteosarcomas, and selected soft tissue sarcomas situated immediately adjacent to critical neurovascular bundles (e.g., the sciatic nerve or popliteal vessels).
Furthermore, PDA serves as a powerful palliative and local control tool in the management of metastatic bone disease, particularly in the spine and pelvis. In cases of epidural spinal cord compression secondary to metastatic disease, achieving wide negative margins is anatomically impossible. PDA can be utilized following separation surgery to ablate the residual epidural tumor, maximizing local tumor control, preserving neurologic function, and providing durable pain relief.
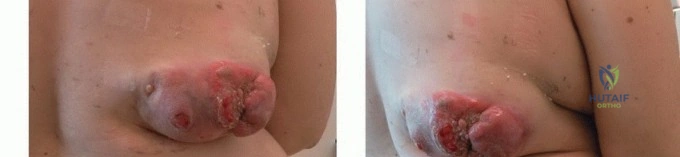
Contraindications
Absolute contraindications to systemic photodynamic therapy include patients with known porphyrias or documented hypersensitivity to porphyrin-based photosensitizers. Relative contraindications involve large, bulky, un-resectable tumor masses. Because photodynamic ablation relies on the physical penetration of photons, its effective depth is typically limited to a few millimeters to a centimeter, depending heavily on the wavelength and tissue optical density. Therefore, PDA cannot replace meticulous gross surgical cytoreduction. Additionally, because the Type II photodynamic reaction is strictly oxygen-dependent, highly necrotic or severely hypoxic tumor regions may exhibit resistance to photodynamic ablation, rendering the treatment sub-optimal in ischemic beds.
Comparison of Treatment Modalities
| Clinical Parameter | Traditional Wide Resection | Intralesional Resection with Photodynamic Ablation |
|---|---|---|
| Surgical Margins | En bloc removal with a continuous normal tissue cuff | Marginal or intralesional cytoreduction |
| Microscopic Disease Control | Achieved via anatomic barrier excision | Achieved via targeted photochemical cytotoxicity |
| Biomechanical Impact | High structural loss requiring complex reconstruction | Preservation of native host bone and joint surfaces |
| Adjuvant Therapy Need | High reliance on systemic chemo/radiation | Reduced reliance on local adjuvant radiation |
| Functional Outcome | Often compromised due to muscle/nerve sacrifice | Maximized preservation of native anatomy and kinematics |
| Complication Profile | High risk of implant failure, aseptic loosening, and deep infection | Risk of cutaneous phototoxicity and localized wound healing issues |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is mandatory to ensure both complete gross tumor resection and optimal photodynamic ablation of the resulting surgical bed. Advanced cross-sectional imaging is critical. Gadolinium-enhanced magnetic resonance imaging (MRI), utilizing T1 post-contrast, STIR, and T2-weighted sequences, is utilized to precisely map the tumor volume, delineate the extent of the reactive pseudocapsule, and evaluate the proximity of the tumor to major neurovascular structures and fascial boundaries.
Photosensitizer Administration and Pharmacokinetics
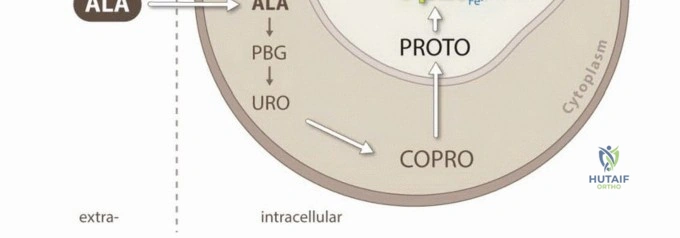
The selection and administration route of the specific photosensitizer dictate the preoperative timeline and logistical workflow. Systemic agents, such as porfimer sodium (Photofrin) or 5-aminolevulinic acid (5-ALA), must be administered intravenously or orally at a highly specific "drug-light interval" prior to surgery. This interval (often 24 to 72 hours) allows for the maximum differential accumulation of the agent within the neoplastic tissue while simultaneously clearing from normal, healthy tissues. Conversely, for topically applied agents like acridine orange, the application occurs intraoperatively, directly into the surgical cavity for a specified dwell time following gross tumor removal.
Patient Positioning and Operating Room Setup
Patient positioning must facilitate not only the traditional surgical approach but also the unimpeded, geometric delivery of light to the entire complex topography of the surgical cavity. The operating room must be equipped with specialized biophotonic light delivery systems, including tunable laser modules or xenon arc lamps, coupled with fiber optic cylindrical diffusers or microlens fibers to ensure uniform light distribution. Depending on the photosensitizer utilized, the ambient lighting in the operating room may need to be strictly controlled (e.g., utilizing specialized filters or darkened rooms) to prevent premature photobleaching of the agent or unintended phototoxicity to the patient's exposed skin and retinas during the surgical approach.

Detailed Surgical Approach and Technique
The surgical execution of targeted photodynamic ablation is a meticulously staged procedure that seamlessly combines the established principles of traditional surgical oncology with advanced intraoperative biophotonics. The procedure is typically divided into four distinct phases: gross cytoreduction, fluorescence-guided micro-resection, photodynamic ablation, and structural reconstruction.
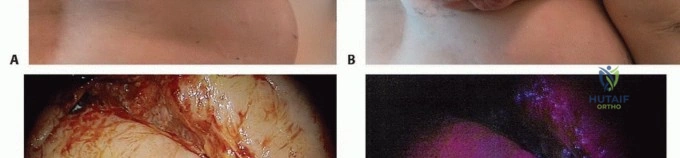
Phase One Gross Cytoreduction
The surgical approach is planned to allow direct, unhindered access to the tumor while minimizing the contamination of adjacent, uninvolved anatomic compartments. Following meticulous hemostasis and compartmental isolation, the tumor is exposed. In the context of an intralesional approach (e.g., for a Giant Cell Tumor), the tumor capsule or cortical window is incised, and gross tumor debulking is aggressively performed using a combination of hand curettes, rongeurs, and high-speed motorized burrs to extend the curettage into normal cancellous bone. For soft tissue sarcomas or surface bone lesions, a marginal resection is performed, dissecting meticulously just outside the reactive pseudocapsule. The objective of this initial phase is to remove all macroscopic disease, leaving only the microscopic cellular burden embedded within the walls of the surgical cavity.
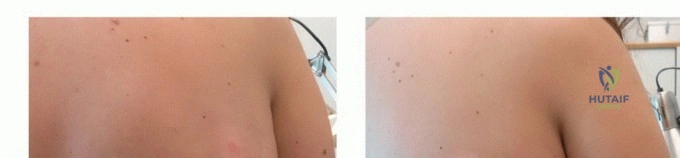
Phase Two Fluorescence Guided Surgery
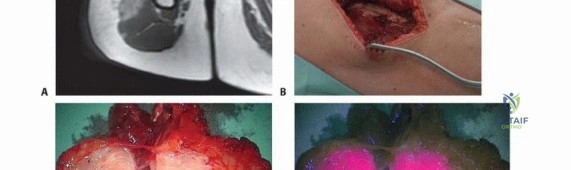
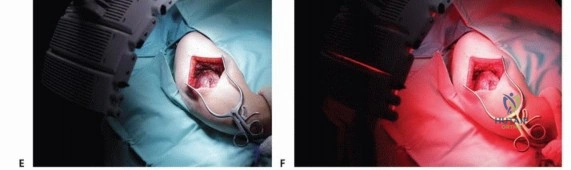
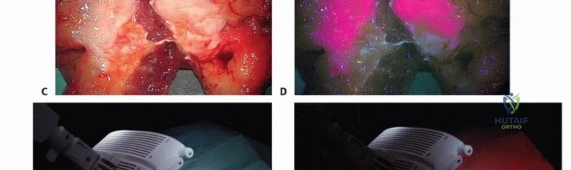
Once gross cytoreduction is deemed visually complete, the surgical field is rigorously evaluated for microscopic residual disease. If a fluorophore such as acridine orange or 5-ALA has been utilized, the surgical field is illuminated with a specific excitation light source (typically blue light around 400-460 nm). Acridine orange, for example, binds selectively to the acidic microenvironment and high nucleic acid content of tumor cell lysosomes and DNA. Under blue light illumination, the residual tumoral tissue emits a distinct, high-contrast green or red fluorescence depending on the specific intracellular binding site.

This emitted fluorescence is detected using a designated surgical microscope or exoscope equipped with appropriate optical emission filters. The surgeon systematically evaluates the entire complex geometry of the cavity walls. Any tissue demonstrating positive fluorescence is meticulously excised. This micro-resection is often performed utilizing an ultrasonic surgical aspirator (CUSA) or a fine ophthalmic scalpel, allowing for precise, millimeter-by-millimeter resection of the fluorescent tissue while safely sparing adjacent critical structures such as peripheral nerves, major vessels, or articular cartilage.
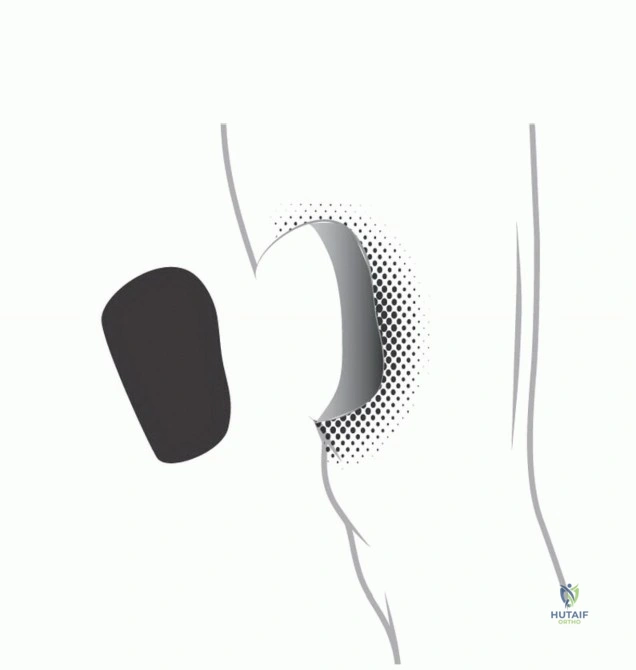
Phase Three Photodynamic Ablation
Following the completion of fluorescence-guided micro-resection, the final ablative phase is initiated. The entire surgical field is subjected to high-intensity, specific-wavelength illumination to activate the photosensitizer within any remaining, non-visible microscopic disease. In protocols utilizing acridine orange, the surgical field is typically illuminated with unfiltered light from a xenon lamp or a specifically tuned laser system.
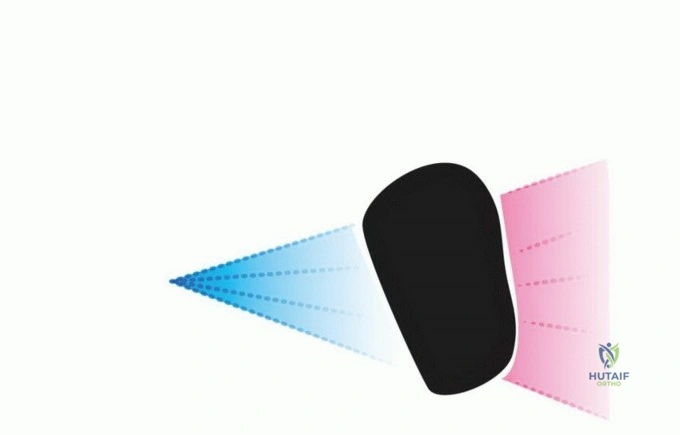
The dosimetry of the light application is highly critical and must be strictly calculated based on the surface area and depth of the cavity. The light energy is measured in Joules per square centimeter ($J/cm^2$), while the irradiance is measured in milliwatts per square centimeter ($mW/cm^2$). Adequate oxygenation of the tissue must be maintained during this phase (avoiding prolonged tourniquet ischemia if possible during illumination), as the generation of singlet oxygen is the primary mechanism of cellular destruction. The photodynamic reaction induces rapid microvascular thrombosis within the tumor bed, followed by direct tumor cell apoptosis and necrosis.
Phase Four Structural Reconstruction
Upon completion of the targeted ablation, the surgical cavity is thoroughly irrigated with pulsatile lavage to remove necrotic debris. Because the host bone architecture and joint surfaces have been largely preserved, reconstruction is often significantly less complex than following wide en bloc resection. Cavitary defects in bone may be reconstructed using polymethylmethacrylate (PMMA) bone cement. PMMA provides immediate structural stability and imparts an additional thermal ablative effect during its exothermic polymerization, synergizing with the PDA. Alternatively, structural allografts, autografts, or synthetic bone substitutes combined with rigid internal fixation using plates and screws may be employed to restore durable biomechanical integrity to the limb.

Complications and Management
While photodynamic ablation significantly reduces the systemic toxicity associated with traditional cytotoxic chemotherapy and the severe local tissue fibrosis associated with external beam radiation, it is not without specific, modality-related complications. Orthopedic oncologists must be adept at anticipating, recognizing, and managing these unique adverse events.
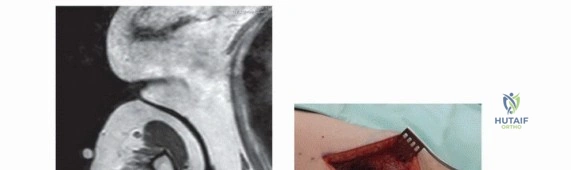
Cutaneous Phototoxicity
The most common systemic complication associated with intravenously administered photosensitizers (e.g., Photofrin) is prolonged cutaneous phototoxicity. Because the agent circulates systemically and accumulates in the dermal layers, exposure to ambient sunlight or bright indoor lighting can induce severe, burn-like erythema, blistering, and edema. Standard sunscreens are ineffective as they block UV light, whereas photosensitizers are activated by visible light. This risk is mitigated by strict adherence to postoperative light precautions, gradually reintroducing the patient to normal lighting conditions over weeks as the photosensitizer is cleared. Topically applied agents like acridine orange carry a negligible risk of systemic phototoxicity.
Local Wound Complications
The intense local generation of reactive oxygen species and subsequent microvascular thrombosis can compromise the viability of the immediate surgical margins, leading to delayed wound healing, marginal skin flap necrosis, or persistent seroma formation. Careful, tension-free layered closure over closed suction drains is imperative. If a major neurovascular bundle was exposed within the ablation field, there is a theoretical risk of thermal or photochemical injury to the vasa nervorum or adventitia, potentially leading to neuropraxia or vascular blowout, though the clinical incidence remains low when appropriate, calculated light dosimetry is utilized.
Summary of Complications and Salvage Strategies
| Complication | Estimated Incidence | Etiology and Risk Factors | Management and Salvage Strategy |
|---|---|---|---|
| Cutaneous Phototoxicity | 10 - 30% (Systemic PS) | Premature exposure to visible light post-infusion | Strict light avoidance protocols, topical corticosteroids, standard burn management |
| Delayed Wound Healing | 5 - 15% | Photochemical damage to skin flaps, local ischemia | Aggressive debridement, negative pressure wound therapy, local rotational muscle flaps |
| Local Tumor Recurrence | 10 - 20% | Incomplete gross resection, inadequate light dosimetry, hypoxic tumor bed | Repeat intralesional resection with PDA, escalation to wide resection, or adjuvant radiotherapy |
| Pathological Fracture | 5 - 10% | Inadequate structural reconstruction of the ablated cavity | Prophylactic internal fixation, revision osteosynthesis with structural bone grafting |
| Neurovascular Injury | < 5% | Direct photochemical toxicity to adjacent nerves/vessels | Intraoperative physical shielding of nerves, careful light dosimetry, neuropathic pain management |
Post Operative Rehabilitation Protocols
The postoperative rehabilitation protocol following photodynamic ablation is dictated by two primary factors: the mechanical stability of the reconstructed limb (bony vs. soft tissue defect) and the specific pharmacokinetic profile of the photosensitizer utilized during the procedure.
Light Avoidance and Systemic Clearance
For patients receiving systemic photosensitizers, a strict, documented light-restriction protocol is initiated immediately postoperatively. Patients are maintained in a darkened environment, with ambient light levels strictly controlled and measured via lux meters. Over a period of several weeks, the patient is gradually exposed to increasing levels of ambient light to facilitate the controlled photobleaching of the residual drug in the skin without inducing a massive phototoxic reaction. Direct sunlight must be absolutely avoided for four to six weeks, depending on the specific half-life of the agent.
Functional Rehabilitation
Because photodynamic ablation allows for the preservation of major muscle groups, tendons, and articular joint surfaces, functional rehabilitation can often progress significantly more rapidly than following wide en bloc resections and endoprosthetic replacements. If the bony defect was reconstructed with polymethylmethacrylate cement and rigid internal fixation, patients may often begin early, protected weight-bearing. Range of motion exercises for adjacent joints are initiated immediately in the postoperative period to prevent capsular contracture, tendon adhesions, and complex regional pain syndrome.
Routine oncologic surveillance is mandatory. Serial radiographs are obtained to assess the integrity of the reconstruction, monitor graft incorporation, and evaluate for signs of local recurrence (e.g., progressive osteolysis). Magnetic resonance imaging, with and without gadolinium contrast, is utilized to evaluate the soft tissues of the surgical bed, though interpretation can be complex due to postoperative inflammatory changes, fibrosis, and susceptibility artifacts from reconstruction materials.
Summary of Key Literature and Guidelines
The integration of photodynamic ablation into orthopedic oncology has been driven by a select group of pioneering researchers and academic surgeons. The seminal work by Matsubara et al. provided early, compelling evidence for the efficacy of this approach. In their prospective evaluation, they reported on patients with locally aggressive bone and soft tissue sarcomas of the extremities who underwent intralesional resection followed by topical acridine orange application.
The protocol utilized a rigorous three-step approach: fluorescence-guided micro-resection using blue light excitation, broad-spectrum illumination of the surgical field with a xenon lamp for photodynamic ablation, and a single, low-dose session of intraoperative or immediate postoperative radiation therapy to address deeper microscopic extensions. When this cohort was compared to a matched group of patients undergoing traditional wide tumor resection, the photodynamic ablation group demonstrated equivalent rates of local tumor control but vastly superior functional outcomes, as measured by standardized limb salvage scoring systems (e.g., MSTS scores).
Subsequent studies have successfully expanded the application of photodynamic ablation to spinal metastasis, demonstrating significant reductions in epidural tumor recurrence following separation surgery and decompression. Current academic guidelines suggest that while photodynamic ablation remains an adjunct rather than a complete replacement for sound oncologic surgical principles, it offers a highly valuable tool for maximizing local control while minimizing the profound morbidity associated with radical resections. Future research in musculoskeletal oncology is heavily focused on the development of targeted nanoparticle delivery systems, which aim to enhance the specificity of photosensitizer uptake via antibody conjugation, and the utilization of near-infrared light sources to exponentially increase the depth of tissue ablation, further solidifying photodynamic ablation as a safer, highly effective, and joint-sparing approach.
