Adjuvant Treatment in Orthopaedic Oncology: A Comprehensive Surgical Guide
Key Takeaway
Adjuvant treatment in orthopaedic oncology integrates radiation therapy, chemotherapy, and precise surgical resection to eradicate primary musculoskeletal malignancies and palliate metastatic disease. This comprehensive guide details the biomechanical principles, indications, and surgical techniques required for optimal limb salvage. By mastering neoadjuvant protocols, brachytherapy application, and biopsy principles, orthopaedic surgeons can significantly improve patient survival rates while minimizing severe complications like pathological fractures and radiation-induced sarcomas.
Introduction to Adjuvant Treatment in Musculoskeletal Malignancies
The overarching goal of treatment in a patient presenting with a primary malignancy of the musculoskeletal system is to achieve a disease-free state while maximizing functional limb salvage. Conversely, the primary objective in treating a patient with metastatic carcinoma to the bone is palliative: to minimize pain, prevent impending pathological fractures, and preserve mechanical function.
Achieving these goals is rarely accomplished through surgical resection alone. The optimal management of musculoskeletal tumors mandates a highly coordinated, multidisciplinary approach integrating radiation therapy, chemotherapy, and surgery. The precise sequencing of these modalities—whether neoadjuvant (preoperative) or adjuvant (postoperative)—dictates not only the survival of the patient but the viability of the reconstructed limb.
Clinical Pearl: The success of adjuvant therapy is inextricably linked to the quality of the initial surgical biopsy. A poorly planned biopsy can contaminate previously uninvolved anatomical compartments, forcing the expansion of radiation fields, increasing the complexity of the definitive resection, or even necessitating amputation.
The Critical Role of Biopsy Planning
Before initiating any neoadjuvant chemotherapy or radiation protocol, a definitive histological diagnosis must be established. The biopsy is the first, and arguably the most critical, surgical step in orthopaedic oncology.
Meticulous adherence to oncologic biopsy principles is mandatory. The biopsy tract must be considered contaminated with tumor cells and must be excised en bloc with the primary tumor during the definitive resection. Failure to adhere to these principles leads to catastrophic complications.
Examples of Poorly Performed Biopsies
The following clinical examples illustrate severe deviations from standard oncologic biopsy protocols, leading to compromised adjuvant treatment and surgical outcomes:

A and B: Biopsy resulting in an irregular cortical defect. Creating a large, non-circular window in the bone creates a massive stress riser. This significantly weakens the biomechanical integrity of the bone, leading directly to a pathological fracture before neoadjuvant therapy can even be completed.
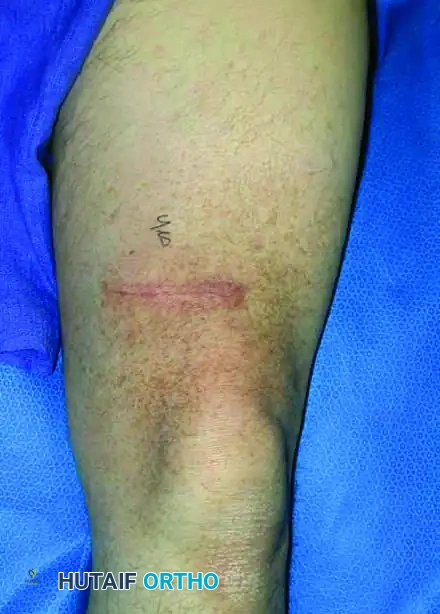
C: Transverse incisions. A transverse incision violates multiple longitudinal anatomical compartments and neurovascular planes. Biopsy incisions must always be longitudinal and placed perfectly in line with the planned definitive surgical approach.
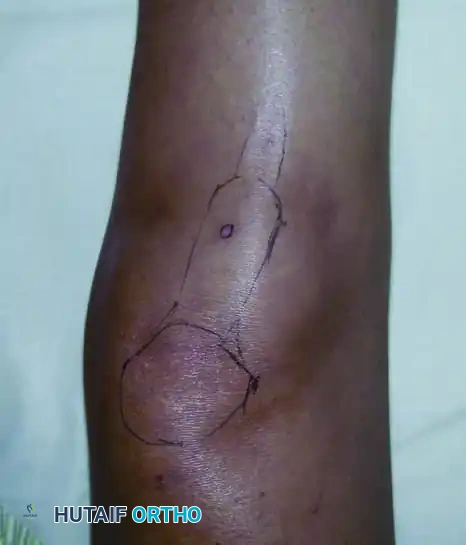
D: Needle biopsy track contaminating the patellar tendon. Contamination of the extensor mechanism requires resection of the tendon during definitive surgery, severely compromising postoperative knee mechanics.
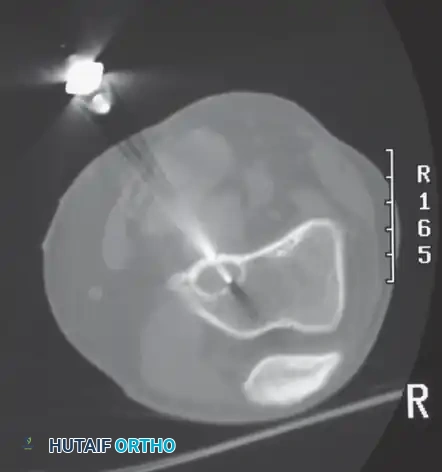
E: Posteriorly placed needle track. A posterior approach to a lesion that requires an anterior or lateral definitive resection makes en bloc excision of the biopsy tract virtually impossible without sacrificing critical posterior neurovascular structures.
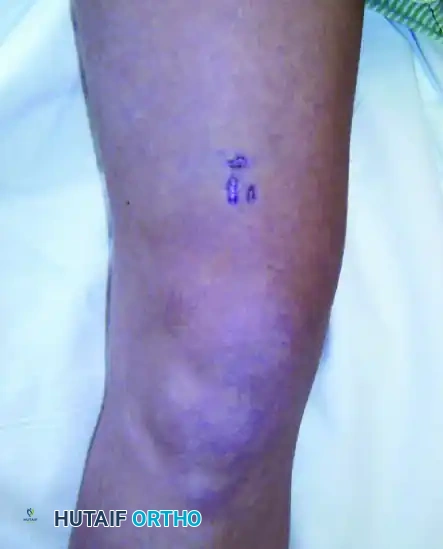
F: Multiple needle tracks. Multiple passes that exit the skin at different sites massively expand the zone of contamination, requiring a much larger soft-tissue resection and a wider radiation field.
G: Improper drain placement. The drain site was not placed in line with the primary incision. Because the drain tract is considered contaminated, it must be excised. An offset drain requires a wider, more morbid skin excision.
Radiation Therapy: Radiobiology and Clinical Application
Radiation therapy is a cornerstone of adjuvant treatment, particularly for soft-tissue sarcomas and specific radiosensitive bone tumors.
Cellular Mechanisms of Radiation
Radiation induces cell death primarily by generating intracellular free radicals (reactive oxygen species) that subsequently cause lethal double-strand DNA breaks. The sensitivity of a neoplastic cell to radiation is governed by several critical factors:
- Cell Cycle Position: Cells are most radiosensitive during the actively mitotic (M) phase and late G2 phase. They are most radioresistant during the late S (DNA synthesis) phase.
- Tissue Oxygenation: Local tissue hypoxia provides a protective effect to the tumor. Because oxygen is required to form the damaging free radicals (the Oxygen Enhancement Ratio), hypoxic necrotic cores of tumors are highly resistant to radiation.
- Apoptotic Capability: The cell’s inherent ability to repair DNA damage or its inability to undergo apoptosis (programmed cell death) dictates its survival post-exposure.
Dosimetry and Fractionation
The absorbed dose of radiation is measured in gray (Gy), where 1 Gy equals 1 joule of absorbed energy per kilogram of tissue. (Note: 1 rad = 1 centigray [cGy]).
The therapeutic goal is to deliver a tumoricidal dose while sparing adjacent normal tissues. This is achieved using modern linear accelerators that deliver highly conformal doses with sharp lateral field edges. Furthermore, therapeutic advantage is maximized through fractionation—dividing the total dose into smaller daily treatments.
Fractionation exploits the "Four Rs" of radiobiology:
* Reassortment: Allows surviving cells to progress into a more sensitive phase of the cell cycle (e.g., M phase) before the next dose.
* Reoxygenation: As outer tumor cells die, previously hypoxic inner cores gain access to blood supply, becoming oxygenated and thus more radiosensitive.
* Repair: Allows normal, healthy cells time to repair sublethal DNA damage between fractions.
* Repopulation: Normal tissues repopulate faster than tumor tissues during the treatment breaks.
Standard protocols deliver 150 to 200 cGy per day. Total target doses range from 30 to 40 Gy for highly sensitive lesions (e.g., multiple myeloma) up to 60 to 65 Gy for high-grade soft-tissue sarcomas.
Indications and Tumor Sensitivity
Most primary bone malignancies (e.g., classic osteosarcoma, chondrosarcoma) are relatively radioresistant. For these tumors, radiation plays a limited role, and local control is best achieved via wide surgical resection.
However, exceptions exist. Marrow-derived tumors—including multiple myeloma, lymphoma, and Ewing sarcoma—are exquisitely radiosensitive. Carcinomas metastatic to bone (breast, prostate, lung, thyroid) also respond well to palliative radiation, with the notable exception of renal cell carcinoma, which is notoriously radioresistant.
For soft-tissue sarcomas, radiotherapy is frequently utilized to reduce the incidence of local recurrence following a marginal resection, particularly when achieving wide margins would require a functionally devastating amputation. Preoperative (neoadjuvant) radiation is often preferred as it can encapsulate the tumor, reduce its volume, and thicken the pseudocapsule, making surgical resection safer and more effective.
Complications of Radiation Therapy
Radiation therapy carries significant acute and long-term morbidities that the orthopaedic surgeon must anticipate:
- Acute Complications: Skin erythema progressing to moist desquamation (especially when combined with cytotoxic chemotherapy), gastrointestinal upset, fatigue, and extremity edema.
- Late Complications: Chronic lymphedema, severe soft-tissue fibrosis, joint contractures, and osteonecrosis.
- Pathological Fractures: Radiation-induced osteonecrosis severely compromises bone biomechanics. A landmark study by Lin et al. demonstrated a 29% incidence of pathological fracture at 5 years following treatment of thigh soft-tissue sarcomas involving radiation and wide resection with periosteal stripping. This risk skyrocketed to 66% in female patients who also received chemotherapy. These fractures are notoriously difficult to treat, often resulting in nonunion due to the avascular nature of the irradiated bone.
- Secondary Radiation Sarcomas: Malignant transformation of irradiated tissues is a devastating late complication, particularly in survivors of childhood cancers. These secondary sarcomas (often high-grade osteosarcomas or undifferentiated pleomorphic sarcomas) occur with a mean latency period of approximately 10 years and carry a dismal prognosis.
- Pediatric Sequelae: In children, radiation across open physes causes growth arrest, leading to severe limb-length discrepancies, scoliosis, kyphosis, and hypoplasia of the irradiated bones (e.g., ilium).
Surgical Warning: Due to the severe long-term sequelae, radiotherapy is rarely indicated for benign conditions. Rare exceptions include aggressive, recurrent pigmented villonodular synovitis (PVNS) uncontrollable by synovectomy, or massive, surgically inaccessible spinal giant cell tumors.
Advanced Delivery: Brachytherapy
In addition to conventional external beam radiation therapy (EBRT), radiation can be delivered via brachytherapy (from the Greek brachys, meaning "close"). This technique involves implanting radioactive sources directly into the tumor bed.
Surgical Technique for Brachytherapy:
At the time of surgical resection, hollow catheters are meticulously implanted into the tumor bed. To maintain precise geometric spacing (usually 1 cm apart), the catheters are often woven through a polyglactin 910 (Vicryl) mesh. This mesh is then sutured directly over the high-risk margins, such as adjacent bone or major neurovascular bundles. The catheters are brought out through the skin via separate stab incisions, away from the primary surgical wound.
Postoperatively, computer dosimetry calculates the optimal loading of radioisotopes (e.g., Iridium-192) into the catheters. This allows a massive, highly localized dose of radiation to be delivered directly to the target tissue, with radiation levels falling off exponentially at the field edges, thereby sparing the overlying skin and surrounding normal tissues.
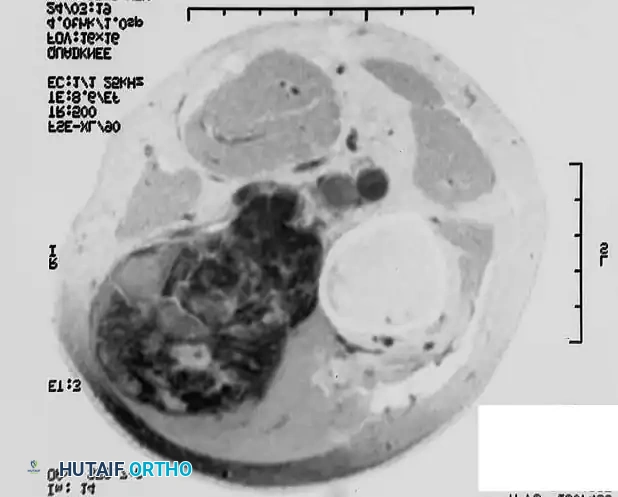
A: Preoperative MRI of a high-grade soft-tissue sarcoma in an 85-year-old man, demonstrating the tumor's intimate proximity to the distal femur and the superficial femoral vessels.
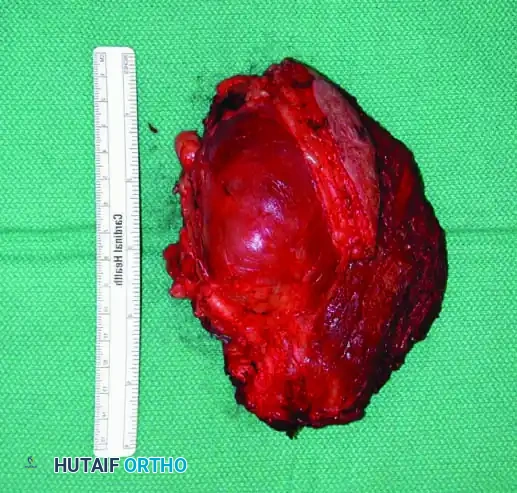
B: The resected gross specimen, excised with marginal margins due to the proximity of critical structures.
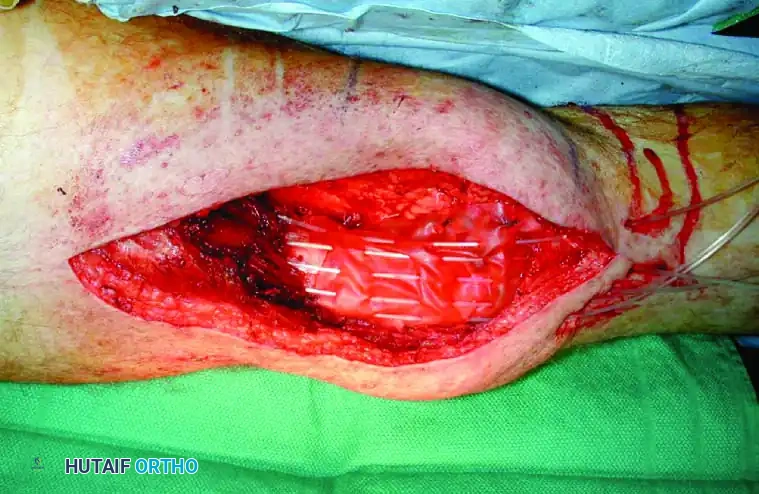
C: The surgical tumor cavity. Note the exposed distal femur and the skeletonized femoral vessels (arrow), representing areas of high risk for microscopic residual disease.
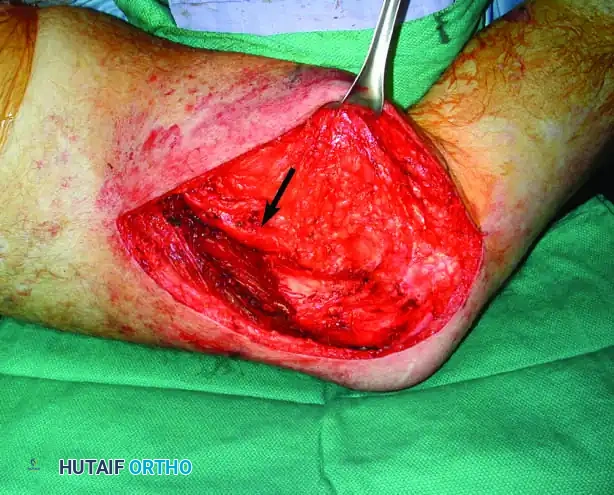
D: Brachytherapy application. Catheters are woven through a polyglactin 910 (Vicryl) mesh to maintain strict parallel spacing. The mesh is secured along the vessels and bone where margins were closest. The catheters exit through separate stab wounds, and the primary wound is closed over the array.
Chemotherapy in Musculoskeletal Malignancies
The advent of systemic chemotherapy revolutionized the field of orthopaedic oncology, shifting the standard of care from radical amputation to functional limb salvage.
The Paradigm Shift: Treating Micrometastatic Disease
Historically, before the routine use of chemotherapy, patients diagnosed with osteosarcoma were treated with immediate radical amputation. While this achieved excellent local control, approximately 80% of these patients eventually died from pulmonary metastatic disease within two years. This grim statistic proved that at the time of initial diagnosis, 80% of patients already harbored undetectable micrometastases.
The introduction of multi-agent systemic chemotherapy directly targets these micrometastases. Today, with modern chemotherapy protocols, the 5-year survival rate for localized osteosarcoma has improved dramatically to approximately 70%. Similar survival benefits are seen in Ewing sarcoma, malignant fibrous histiocytoma of bone (pleomorphic undifferentiated sarcoma), and high-grade pediatric soft-tissue sarcomas like rhabdomyosarcoma.
Neoadjuvant vs. Adjuvant Chemotherapy
- Adjuvant Chemotherapy: Administered after surgical resection to eradicate presumed micrometastases.
- Neoadjuvant Chemotherapy: Administered before surgical resection.
While large-scale studies have not definitively proven a long-term overall survival advantage of neoadjuvant over adjuvant timing, neoadjuvant chemotherapy offers profound clinical and surgical advantages:
- Tumor Regression: Preoperative chemotherapy frequently induces tumor necrosis and shrinkage, converting an unresectable lesion into one amenable to limb salvage. (Malawar et al. demonstrated that 75% of initially unresectable lesions became resectable following neoadjuvant therapy).
- In Vivo Chemosensitivity Testing: Resecting the tumor after chemotherapy allows the pathologist to calculate the percentage of tumor necrosis (e.g., Huvos grading). A high necrosis rate (>90%) is the single most valuable prognostic indicator for long-term survival. Poor responders can have their postoperative chemotherapy regimens altered.
- Early Treatment of Micrometastases: Systemic therapy begins immediately, halting the progression of microscopic pulmonary disease while surgical planning takes place.
- Surgical Planning Time: The 10 to 12 weeks of neoadjuvant treatment provides crucial time to manufacture custom endoprosthetic implants and allows the patient and family to psychologically prepare for complex limb salvage or amputation.
Surgical Timing and Complications:
Neoadjuvant chemotherapy is not without risks. Cytotoxic agents impair fibroblast proliferation and angiogenesis, significantly increasing the risk of delayed wound healing, wound dehiscence, and deep periprosthetic joint infections. To mitigate these risks, orthopaedic oncologists typically schedule the definitive resection 3 to 4 weeks after the final dose of preoperative chemotherapy, allowing bone marrow and nutritional indices to recover. Postoperative chemotherapy is resumed approximately 2 to 3 weeks after surgery, provided the surgical wound has completely healed.
Tumor Heterogeneity and Combination Regimens
Malignancies are not uniform; they are composed of a heterogeneous population of cells resulting from rapid turnover and genetic instability. Consequently, different cell clones within the same tumor develop varying mechanisms of chemoresistance. To combat this, modern protocols utilize combinations of synergistic cytotoxic drugs (e.g., Methotrexate, Doxorubicin, Cisplatin, and Ifosfamide for osteosarcoma). These agents are most effective when the tumor burden is small, highlighting the importance of aggressive surgical debulking.
Exceptions to Adjuvant Therapy: Low-Grade and Cartilaginous Lesions
It is critical to recognize that chemotherapy and radiation are generally ineffective for cartilaginous lesions (chondrosarcomas) and most other low-grade malignancies. Because these tumors have a low mitotic index and poor vascularity, systemic cytotoxic agents cannot effectively penetrate or disrupt their cell cycles.
For these lesions, wide surgical resection is the only curative option. In highly selected cases of low-grade lesions with classic radiographic features, primary resection without a prior biopsy may be indicated to avoid contaminating adjacent critical structures.
Case Study: Primary Resection of a Low-Grade Chondrosarcoma
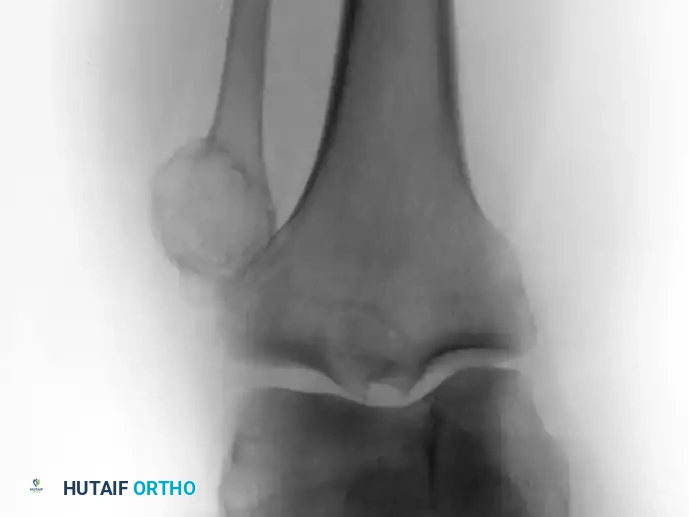
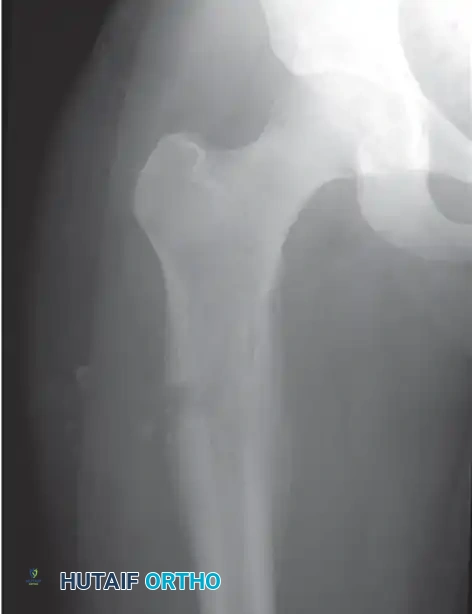
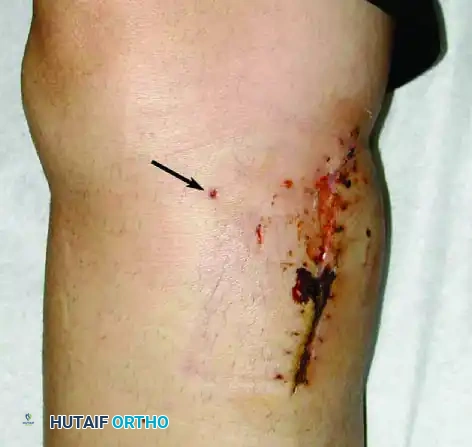
A: Anteroposterior radiograph of the right knee in a 50-year-old woman presenting with progressive pain. An expansile lesion is visible in the proximal fibula, containing classic "popcorn" calcifications highly suggestive of a cartilaginous tumor.
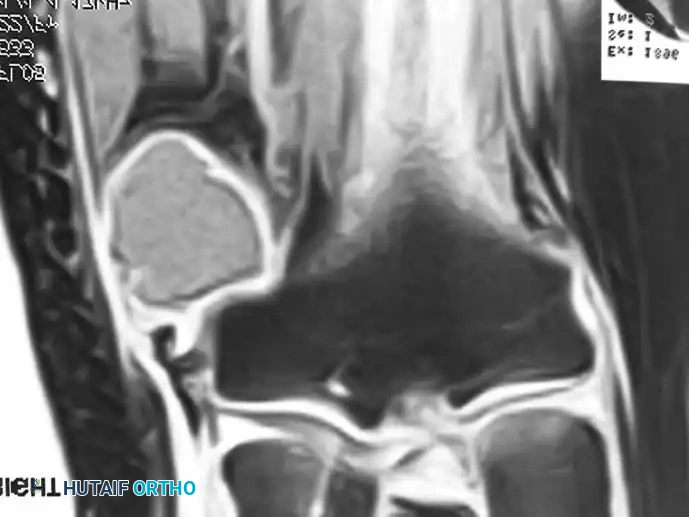
B: Coronal T1-weighted MRI confirms the lesion remains entirely contained within the fibular cortex, with no soft-tissue extension.
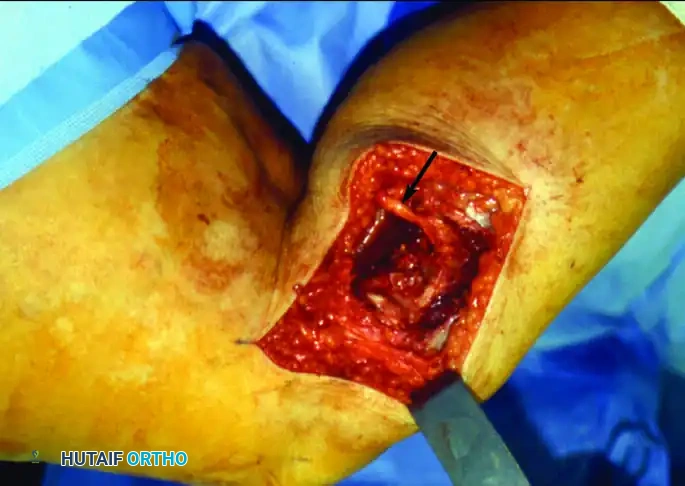
C: Intraoperative view. Because the clinical and radiographic picture overwhelmingly pointed to a low-grade chondrosarcoma (which is chemo/radio-resistant), a primary en bloc resection of the proximal fibula was performed without a prior biopsy. This strategic decision prevented biopsy tract contamination, allowing the surgeon to achieve wide margins while safely dissecting and preserving the common peroneal nerve (arrow).
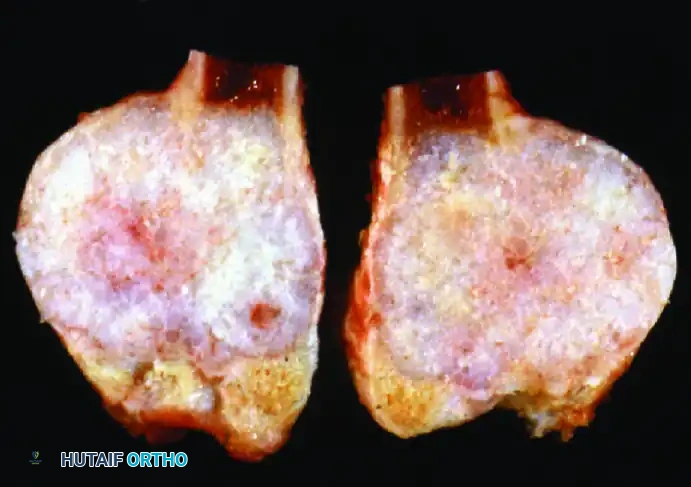
D: Gross pathological specimen sectioned longitudinally. Histological analysis confirmed a low-grade chondrosarcoma with negative surgical margins.
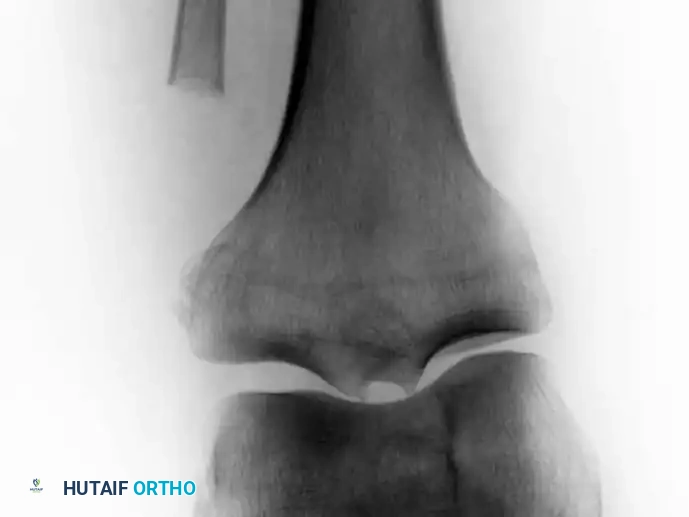
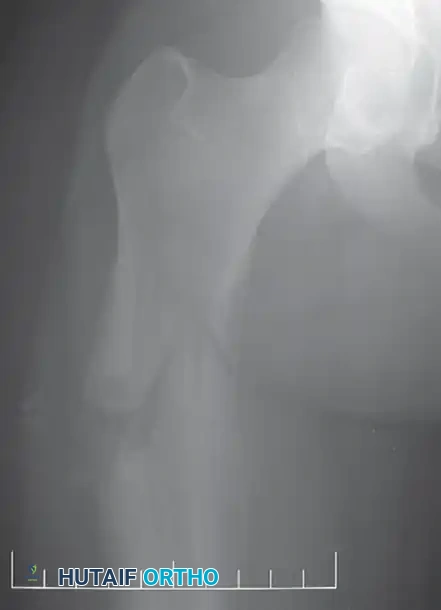
E: Postoperative radiograph demonstrating the complete primary resection of the proximal fibula. The patient required no adjuvant chemotherapy or radiation, and retained full function of the peroneal nerve.
Conclusion
The integration of adjuvant treatments in orthopaedic oncology requires a profound understanding of tumor biology, biomechanics, and surgical anatomy. By meticulously planning biopsies, leveraging the radiobiological advantages of fractionation and brachytherapy, and timing surgical resections perfectly within neoadjuvant chemotherapy windows, the orthopaedic surgeon
You Might Also Like