Management of Radiation, Electrical, and Chemical Burns of the Hand
Key Takeaway
Radiation, electrical, and chemical burns of the hand present complex reconstructive challenges. Radiation exposure can induce chronic dermatitis and squamous cell carcinoma, necessitating wide excision and grafting. High-voltage electrical injuries cause deep dielectric tissue breakdown and myoglobinuria, requiring aggressive fluid resuscitation and urgent fasciotomy. Chemical burns demand specific neutralizing agents, such as calcium gluconate for hydrofluoric acid, followed by meticulous surgical debridement and early rehabilitation to preserve hand function.
INTRODUCTION TO COMPLEX HAND BURNS
Thermal, radiation, electrical, and chemical burns of the upper extremity represent a highly specialized subset of orthopedic and reconstructive surgery. Unlike standard thermal injuries, radiation, electrical, and chemical burns possess unique pathophysiological mechanisms that dictate highly specific diagnostic and therapeutic algorithms. The hand, with its dense concentration of critical neurovascular, tendinous, and articular structures situated immediately deep to a thin cutaneous envelope, is particularly vulnerable to profound functional impairment following these injuries.
This comprehensive guide delineates the evidence-based management of radiation dermatitis, high- and low-voltage electrical injuries, and specific chemical exposures (hydrofluoric acid, phenol, and white phosphorus) affecting the upper extremity.
RADIATION BURNS OF THE HAND
Pathophysiology and Occupational Hazards
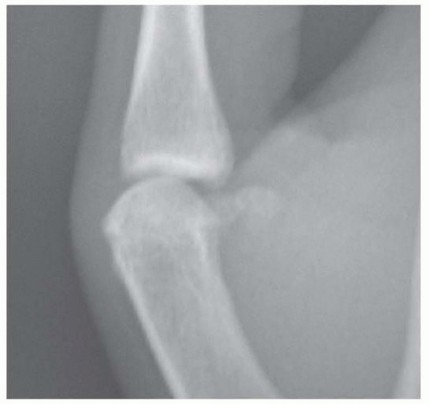
Radiation burns (radiation dermatitis) of the hand are historically and contemporarily linked to the occupational exposure of medical professionals, particularly orthopedic surgeons, interventional radiologists, and cardiologists. Chronic overexposure to roentgen rays (X-rays) during fluoroscopy—often from holding cassettes or manipulating fractures directly within the primary beam without adequate lead shielding—results in cumulative cellular damage.
The pathophysiology is driven by ionizing radiation inducing DNA double-strand breaks, leading to cellular apoptosis and a profound depletion of the basal keratinocytes and dermal fibroblasts. Crucially, radiation induces an endarteritis obliterans—a progressive, irreversible obliteration of the microvasculature. This results in a profoundly ischemic, hypoxic, and fibrotic tissue bed that is incapable of normal wound healing.
Clinical Presentation
The clinical progression of radiation-induced hand injury is insidious:
* Acute Phase (Weeks post-exposure): Erythema, intense pruritus, blistering, and desquamation.
* Chronic Phase (Months to Years): The skin becomes pale, dry, atrophic, and severely wrinkled. Scattered hyperkeratotic plaques (radiation keratoses) develop. The fingernails frequently exhibit longitudinal splitting, ridging, and dystrophy.
* Malignant Degeneration: The ischemic, damaged tissue is highly susceptible to malignant transformation. Multiple squamous cell carcinomas (SCC) frequently develop within the irradiated field, presenting as painful, non-healing, ulcerated lesions.
Clinical Pearl: Pain in a chronic radiation burn is a highly concerning symptom. As the skin becomes increasingly ischemic and ulcerated, the pain can become intractable, often requiring narcotic analgesia. Severe, escalating pain or the breakdown of previously intact atrophic skin should raise immediate suspicion for malignant transformation to squamous cell carcinoma.
Surgical Management and Resurfacing
When conservative measures fail, or when tissue breakdown, intractable pain, or malignant changes occur, surgical resurfacing of the hand is mandatory.
Preoperative Planning:
The goal is complete excision of the irradiated, ischemic tissue. Because the underlying microvasculature is compromised, local random-pattern flaps are generally contraindicated due to an unacceptably high risk of necrosis.
Surgical Technique:
1. Excision: The area of excision must be generous. It is a surgical axiom in radiation burns of the hand that all questionably involved skin must be removed. Typically, this requires the excision of all dorsal skin from the wrist crease distally to the proximal interphalangeal (PIP) joints or beyond.
2. Deep Margin: Excision is carried down to healthy, well-vascularized tissue. Care must be taken to preserve the paratenon over the extensor tendons to provide a vascularized bed for subsequent grafting. If the paratenon is destroyed or involved in malignancy, a vascularized flap (e.g., pedicled groin flap, radial forearm flap, or free tissue transfer) will be required instead of a skin graft.
3. Grafting: Once a healthy bed is established, split-thickness skin grafts (STSG) or full-thickness skin grafts (FTSG) are applied simultaneously. FTSGs provide better durability and less secondary contracture for the dorsum of the hand, though STSGs are more reliable in beds with marginal vascularity.
4. Amputation: In cases of advanced, invasive squamous cell carcinoma with deep structural involvement (bone, joint, or flexor apparatus), ray amputation or more proximal amputation may be the only definitive, life-saving option.
ELECTRICAL INJURIES OF THE UPPER EXTREMITY
Electrical injuries are devastating traumas that often present with a deceptive clinical picture. The visible cutaneous burn frequently represents only a fraction of the total tissue destruction—a phenomenon often termed the "iceberg effect."
Biomechanics and Pathophysiology
The severity of an electrical injury is determined by the voltage, current (amperage), type of current (alternating vs. direct), pathway through the body, and duration of contact. High-voltage injuries are generally defined as those involving greater than 1,000 volts.
Tissue resistance dictates the pattern of deep injury. The resistance of human tissues to electrical current, from least to greatest, is: Nerve < Blood < Muscle < Skin < Tendon < Fat < Bone.
Because bone has the highest resistance, it generates the most heat as current passes through it. Consequently, the deepest muscles immediately adjacent to the bone (e.g., the deep flexor compartment of the forearm) sustain the most severe thermal damage, while superficial muscles may initially appear viable.
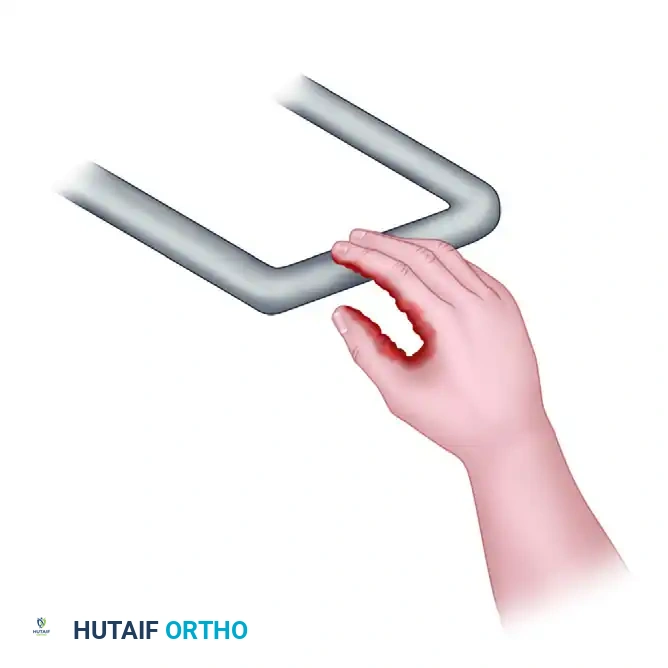
Figure 70-8: Electrical contact. Various damaging forces involved in high-energy electrical shock. With voltages greater than 1000 volts, electrical contact (arc mediated) precedes mechanical contact. High surface temperatures at contact points produce deep burns. Current passage through the extremity leads to the electrical breakdown of muscle and nerve membranes (dielectric breakdown). Prolonged contacts of several seconds result in substantial deep tissue burning. High-energy arcs produce shock waves that can cause blunt trauma.
Mechanisms of Injury:
1. Direct Contact: Current passes directly through the body, causing deep thermal and dielectric breakdown of cell membranes (electroporation).
2. Arc Burns: Current arcs through the air from the source to the patient. Arcs can generate temperatures up to 50,000°C, causing catastrophic, deep, localized thermal burns and explosive shockwaves that result in concomitant blunt trauma (fractures, dislocations).
3. Flash Burns: Superficial thermal burns caused by the heat of a nearby electrical arc, without current passing through the body.
4. Flame Burns: Ignition of the patient's clothing.
Systemic Resuscitation and Evaluation
Electrical injuries are multisystem traumas. The current pathway can involve the central and peripheral nervous systems, cardiopulmonary system, kidneys, and musculoskeletal system.
- Fluid Resuscitation: Standard thermal burn formulas (e.g., Parkland formula) severely underestimate the fluid requirements for electrical burns because they rely on Total Body Surface Area (TBSA), which does not account for massive deep muscle necrosis. Patients require aggressive intravenous fluid resuscitation. The target urinary output is 50 to 100 mL/hr in adults.
- Myoglobinuria: Massive muscle necrosis releases myoglobin, which is highly nephrotoxic and can precipitate acute renal failure. If myoglobinuria is present (dark, "port-wine" urine), fluid rates must be increased, and alkalinization of the urine with sodium bicarbonate may be indicated to increase myoglobin solubility.
- Cardiac Monitoring: Arrhythmias are common. Continuous ECG monitoring is mandatory, alongside serial cardiac enzymes.
- Diagnostics: Obtain radiographs of the affected extremities to rule out fractures/dislocations caused by tetanic muscle contractions or explosive arc shockwaves. Baseline serum chemistries, arterial blood gases (ABG), and liver/renal panels are essential.
Local Hand and Upper Extremity Management
Initial evaluation requires a meticulous assessment of the entire body for entrance and exit wounds (though these terms are technically inaccurate for alternating current, they denote contact points). Contact points typically feature a central charred, depressed area surrounded by a zone of coagulation.
Vascular Assessment and Compartment Syndrome:
The upper extremity must be evaluated for perfusion. Assess skin color, warmth, capillary refill, and peripheral pulses. A Doppler probe is essential for flow assessment.
Electrical injuries frequently cause massive subfascial edema. Combined with deep muscle necrosis, this rapidly leads to acute compartment syndrome.
Surgical Warning: Because of the profound risk of sudden, catastrophic hemorrhage from electrically damaged, necrotic major vessels (e.g., radial or ulnar arteries), a sterile pneumatic tourniquet must be kept immediately available at the patient’s bedside at all times.
Surgical Intervention:
1. Fasciotomy and Escharotomy: If circulatory compromise is detected, or if compartment pressures are elevated, urgent decompression is indicated. Unlike thermal burns which may only require escharotomy (release of the burned skin), high-voltage electrical burns almost universally require true fasciotomies (release of the muscle fascia) of the hand, forearm, and sometimes the arm, to decompress the swelling muscle bellies.
2. Debridement: Serial surgical debridements are the hallmark of electrical burn management. Necrotic muscle must be aggressively excised to prevent sepsis and gas gangrene. Because tissue necrosis can progress over several days due to progressive microvascular thrombosis, patients are typically returned to the operating room every 48 to 72 hours for re-evaluation and further debridement until a stable, viable wound bed is achieved.
3. Reconstruction: Once the wound bed is clean and viable, coverage is achieved via skin grafts, pedicled flaps, or free tissue transfer, depending on the exposure of deep structures (bone, tendon, nerve).
CHEMICAL BURNS OF THE HAND
Chemical burns of the upper extremity cause progressive tissue destruction that continues as long as the offending agent remains active in the tissue. The severity of the burn is dictated by the agent's concentration, volume, duration of contact, and mechanism of action.
Hydrofluoric Acid (HF) Burns
Hydrofluoric acid is a highly corrosive agent used in glass etching, rust removal, and industrial cleaning. It is unique among acids because it causes deep, liquefactive-like necrosis rather than the superficial coagulative necrosis typical of other acids.
Pathophysiology:
The damage is twofold. First, the hydrogen ion causes superficial corrosive burns. Second, and more dangerously, the highly lipophilic fluoride ion penetrates deeply into the tissues where it avidly binds to intracellular and extracellular calcium and magnesium. This causes profound local hypocalcemia, cell death, and the release of potassium, leading to excruciating pain that is classically described as "out of proportion to the visible injury." Systemic absorption can lead to fatal cardiac arrhythmias due to profound systemic hypocalcemia and hyperkalemia.
Treatment Protocol:
1. Copious Irrigation: Immediate and prolonged water irrigation (at least 15-30 minutes).
2. Topical Neutralization: Application of a 2.5% calcium gluconate gel. The gel should be massaged into the burn continuously. The calcium in the gel binds the free fluoride ions, neutralizing them.
3. Infiltration: If pain is not promptly relieved by topical gel, local infiltration of 10% calcium gluconate or magnesium sulfate deep to and surrounding the lesions is indicated. (Note: Calcium chloride should never be injected into tissues as it is highly caustic and will cause further necrosis).
4. Intra-arterial Infusion: For severe, persistent pain or extensive digital burns where local infiltration might cause compartment syndrome, intra-arterial infusion of calcium gluconate via the radial or brachial artery is highly effective.
Phenol (Carbolic Acid) Burns
Phenol is an aromatic hydrocarbon used in industrial disinfectants and chemical peels. It causes severe coagulative necrosis and acts as a local anesthetic, which can dangerously mask the severity of the burn. Systemic absorption can lead to central nervous system depression, cardiovascular collapse, and renal failure.
Treatment Protocol:
Crucially, phenol is not water-soluble. Standard water irrigation is ineffective and may merely spread the chemical over a larger surface area.
Removal must be achieved using a lipophilic solvent. Polyethylene glycol (PEG 300 or 400) or glycerol are the recommended agents to dissolve and remove phenol from the skin. Once the phenol is neutralized with PEG, the area can be washed with water.
White Phosphorus Burns
White phosphorus is an incendiary agent used in military munitions and fireworks. It is highly reactive and spontaneously ignites upon exposure to oxygen in the air, causing severe thermal and chemical burns.
Treatment Protocol:
1. Smothering: The affected area must be immediately submerged in water or covered with saline-soaked dressings to deprive the phosphorus of oxygen and halt combustion. White phosphorus particles will continue to smoke and burn as long as they are exposed to air.
2. Neutralization and Identification: The wound should be irrigated with a 1% to 3% copper sulfate solution. The copper sulfate reacts with the phosphorus to form a black cupric phosphide coating. This blackens the particles, making them easily identifiable, and temporarily impedes further combustion.
3. Underwater Debridement: The blackened particles must be meticulously removed surgically. This debridement should ideally be performed underwater in a water bath to prevent re-ignition of the particles if the cupric phosphide coating is breached.
Surgical Pitfall: Failure to irrigate with copper sulfate prior to water exposure in a non-submerged environment can cause the phosphorus to violently ignite on contact with small amounts of water.
Late Management and Reconstruction of Chemical Burns
Significant chemical burns presenting late require hospitalization. The hand and digital circulation must be continuously monitored using Doppler probes and digital oximetry.
If severe edema results in circulatory compromise from a circumferential burn eschar, urgent surgical decompression (escharotomy/fasciotomy) is indicated. Deeper chemical burns that result in full-thickness skin loss will require formal surgical debridement. Depending on the depth of the defect and the exposure of underlying tendons, nerves, or bone, closure is achieved with split-thickness skin grafts, local pedicle flaps, or free tissue transfer.
POSTOPERATIVE PROTOCOLS AND REHABILITATION
Regardless of the burn etiology (radiation, electrical, or chemical), the postoperative rehabilitation of the burned hand is as critical as the surgical intervention itself.
- Splinting: In the immediate postoperative period, the hand should be splinted in the "safe position" (intrinsic-plus position): wrist extended 20-30 degrees, metacarpophalangeal (MCP) joints flexed 70-90 degrees, and interphalangeal (IP) joints in full extension. This prevents collateral ligament contracture and preserves functional length.
- Elevation: Strict elevation is mandatory to minimize edema, which is a primary driver of joint stiffness and fibrosis.
- Early Motion: Once grafts or flaps have achieved primary adherence (typically 5 to 7 days postoperatively), a rigorous, therapist-directed active and active-assisted range of motion (ROM) program must be initiated.
- Scar Management: Long-term management includes the use of custom compressive garments, silicone gel sheeting, and aggressive scar massage to prevent hypertrophic scarring and secondary joint contractures. Recovery is usually prompt and functional outcomes are optimized only when meticulous surgical treatment is seamlessly integrated with a dedicated hand therapy rehabilitation program.
You Might Also Like