Operative Management of Hand Soft Tissue Tumors: Hemangioma and Glomus Tumor
Key Takeaway
Cavernous hemangiomas and glomus tumors represent distinct soft tissue pathologies in the hand requiring precise surgical management. While hemangiomas present as soft, collapsible masses with distended venous sinuses, glomus tumors are characterized by the classic triad of pain, cold sensitivity, and point tenderness. This guide details the pathophysiology, diagnostic criteria, and step-by-step surgical techniques for marginal and complete excision of these complex vascular and neuromyoarterial lesions.
Introduction to Vascular and Neuromyoarterial Tumors of the Hand
Soft tissue tumors of the hand and upper extremity encompass a diverse array of neoplastic and non-neoplastic lesions. Because of the dense, complex anatomy of the hand, even benign lesions can cause significant functional impairment, sensory deficits, and debilitating pain. Among the most clinically significant of these are vascular malformations—specifically cavernous hemangiomas—and neuromyoarterial lesions, such as the glomus tumor.
This comprehensive guide details the pathophysiology, clinical presentation, and operative management of these specific lesions, alongside the surgical principles for managing other common soft tissue masses like giant cell tumors of the tendon sheath (GCTTS) and fibromas.
Cavernous Hemangioma
The following principles are strictly limited to the cavernous hemangioma and do not apply to the capillary superficial infantile hemangioma (strawberry hemangioma), which typically undergoes spontaneous involution by age 7 and rarely requires surgical intervention.
Cavernous hemangiomas of the upper extremity are complex vascular malformations composed of dilated, endothelium-lined venous sinuses. They can infiltrate local tissues, including subcutaneous fat, muscle, and occasionally bone, making complete surgical eradication challenging.
Clinical Presentation and Pathophysiology
A cavernous hemangioma typically presents as a soft, collapsible mass that may enlarge a digit or specific compartment of the hand.
* Visual Inspection: When the lesion occurs close to the dermal surface, it produces a distinct bluish discoloration.
* Palpation: The mass is usually slightly to moderately tender. It is highly compressible and rapidly refills upon release of pressure due to the distended venous sinuses.
* Positional Changes: The tumor often engorges and becomes more symptomatic when the extremity is in a dependent position.
Clinical Pearl: The presence of calcifications (phleboliths) on standard plain radiographs is a hallmark diagnostic sign of a cavernous hemangioma. These represent calcified thrombi within the stagnant venous sinuses of the lesion.
Kasabach-Merritt Syndrome
A critical, albeit rare, complication associated with large cavernous hemangiomas (typically those larger than 5 cm in diameter) is Kasabach-Merritt Syndrome.
This is a life-threatening consumptive coagulopathy caused by secondary platelet sequestration within the extensive vascular labyrinth of the tumor. The trapping of platelets leads to profound thrombocytopenia, localized intravascular coagulation, and a paradoxical systemic bleeding diathesis. Early, aggressive treatment—often requiring a multidisciplinary approach with hematology—is strictly indicated before any surgical intervention is attempted.
Management Strategies
Conservative Treatment
Conservative management is often the first line of defense for diffuse, asymptomatic, or mildly symptomatic lesions. Custom-fitted compression garments (e.g., Isotoner gloves or custom Coban wrapping) can be highly effective in collapsing the venous sinuses, reducing engorgement, and alleviating aching pain.
Surgical Warning: Radiation therapy is strongly discouraged for benign cavernous hemangiomas of the hand due to the high risk of radiation-induced contractures, skin necrosis, and secondary malignant transformation.
Operative Indications and Technique
Surgery is the treatment of choice when symptoms (pain, functional limitation, or severe cosmetic deformity) justify the risks of intervention.
Surgical Steps for Excision:
1. Anesthesia and Positioning: The patient is placed supine with the arm on a hand table. Regional anesthesia (axillary or supraclavicular block) is preferred to provide postoperative vasodilation and pain control.
2. Tourniquet Control (The "Partial Exsanguination" Technique):
* Crucial Step: Do not tightly exsanguinate the limb with an Esmarch bandage. Instead, simply elevate the arm for 2 to 3 minutes before inflating the pneumatic tourniquet.
* Rationale: This allows blood to partially fill the cavernous sinuses, outlining the true extent of the tumor with a bluish hue at the time of surgical dissection, making it significantly easier to differentiate from surrounding healthy tissue.
3. Incision and Dissection: Utilize a Brunner zigzag or mid-axial incision depending on the lesion's location. Meticulous dissection is required to separate the vascular malformation from the neurovascular bundles.
4. Staged Procedures: The tumor can be so extensive that a single-stage removal would compromise digit viability. In such cases, a staged procedure is required. Vessel ligation of the primary feeding branches during the first stage can assist in shrinking the lesion for a safer second-stage excision.
5. Closure and Prognosis: Complete excision is usually curative if the tumor is fairly well localized. However, in diffuse, infiltrative lesions, persistence (rather than true recurrence) is common. If complete excision is impossible without sacrificing critical structures, tumor debulking should be the primary emphasis of the procedure.
Glomus Tumor
Originally described by Wood in 1812 and histologically clarified by Masson in 1924, the glomus tumor is a rare, benign hamartoma arising from the glomus body.
Anatomy and Pathophysiology
A normal glomus body is a specialized neuromyoarterial receptor located in the reticular layer of the dermis. It functions as an arteriovenous dermal shunt that regulates skin temperature and blood pressure. The normal glomus body is composed of:
* An afferent arteriole
* An anastomotic Sucquet-Hoyer canal
* An afferent venule
* Actin-containing glomus cells surrounding the canals
* An intraglomerular retinaculum and capsule
Hyperplasia of one or more parts of this glomus body causes the tumor. Histologically, glomus cells are specialized perivascular muscle cells that are round or oval with a dense, granular cytoplasm. The tumor is heavily innervated by nonmyelinated nerve fibers intermixed with thick-walled capillaries, which are directly responsible for the severe, lancinating pain characteristic of this lesion.
Clinical Evaluation
Glomus tumors are typically tiny—usually less than 1 cm in diameter, and often only a few millimeters. Despite their small size, they produce profound morbidity. They occur more often in the hand (75% of all cases) than anywhere else in the body. Between 25% and 65% of patients present with subungual lesions, which may be visible through the overlying nail plate as a deep red or purple discoloration.
However, 25% of glomus tumors are not subungual (occurring in the digital pulp or palm), posing a significant diagnostic challenge.
The Classic Diagnostic Triad:
1. Paroxysmal Pain: Severe, lancinating pain that seems disproportionate to the visual findings.
2. Cold Sensitivity: Extreme discomfort when exposed to cold.
3. Point Tenderness: Exquisite, localized tenderness.
Provocative Clinical Tests:
* Love Test: Direct pressure applied to the tumor by a small, firm object (such as the head of a pin or a paperclip) causes excruciating pain. Pressure applied even slightly to one side of the lesion elicits no pain.
* Ice Water Test: Immersing the involved hand or digit in ice water causes severe, reproducible discomfort.
* Hildreth Test: Inflation of a proximal tourniquet to exsanguinate the limb relieves the pain of the Love test. Release of the tourniquet causes the pain to return.
Diagnostic Pearl: When the clinical triad is present but the lesion is not visible, High-Resolution MRI (with gadolinium contrast) is the gold standard imaging modality. The tumor typically appears as a well-demarcated mass with low signal intensity on T1 and high signal intensity on T2-weighted images. Bone scans may also show focal uptake due to adjacent bony erosion.
Surgical Technique: Subungual Glomus Tumor Excision
- Preparation: Digital block anesthesia and a sterile finger tourniquet are utilized.
- Nail Plate Removal: The nail plate is carefully elevated and removed to expose the sterile matrix.
- Matrix Incision: A longitudinal incision is made directly over the bluish discoloration in the nail bed.
- Tumor Enucleation: The tumor is usually well-encapsulated. It is carefully enucleated using a small curette or tenotomy scissors.
- Matrix Repair: The sterile matrix must be meticulously repaired using 6-0 absorbable sutures (e.g., chromic gut) to prevent postoperative nail deformity (split nail). The native nail plate (or a silicone substitute) is placed back into the eponychial fold to stent the matrix open during healing.
Excision of Other Common Hand Tumors: Giant Cell Tumors and Fibromas
To fully understand the surgical approach to hand masses, one must also be familiar with the marginal excision techniques used for solid, encapsulated tumors such as Giant Cell Tumors of the Tendon Sheath (GCTTS) and Fibromas.
Giant Cell Tumor of the Tendon Sheath (GCTTS)
GCTTS is the second most common benign tumor of the hand (after ganglion cysts). It presents as a slow-growing, firm, non-tender mass, typically on the volar aspect of the digits.
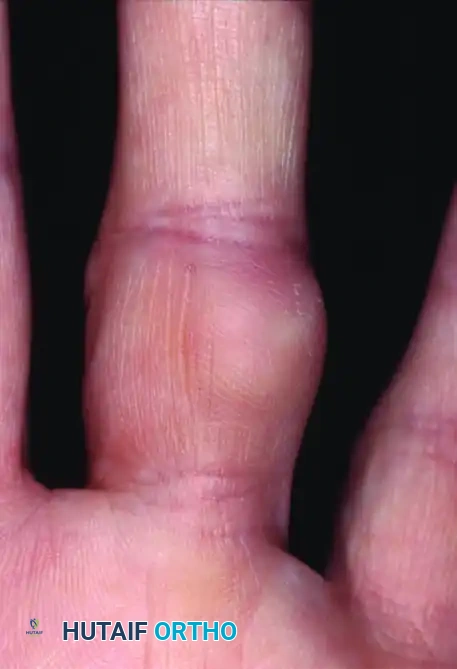
Figure 1: Clinical presentation of a Giant Cell Tumor on the flexor surface of the ring finger, demonstrating a firm, lobulated mass.
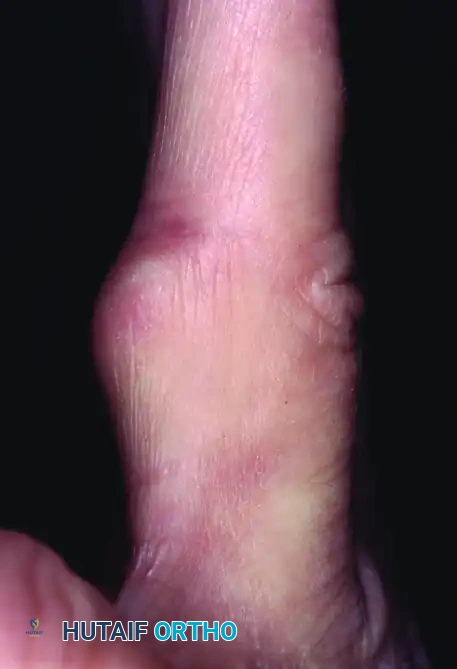
Figure 2: Preoperative marking of the Brunner zigzag incision designed to expose the flexor surface mass while preventing flexion contractures.
During surgical dissection, GCTTS is easily identifiable by its characteristic yellowish-brown color, which is due to the presence of hemosiderin-laden macrophages and foam cells.
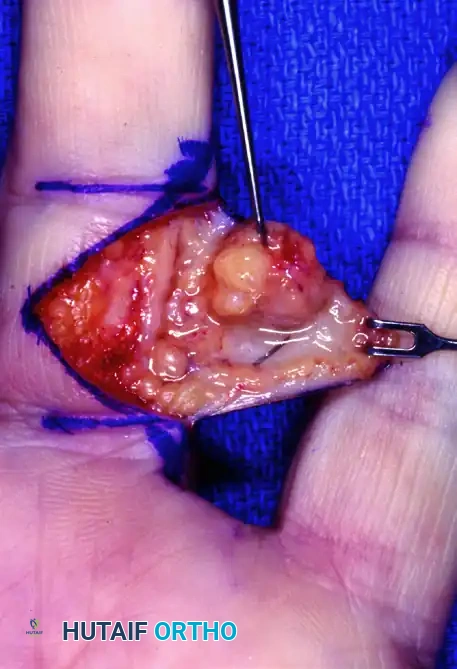
Figure 3: Surgical dissection showing a well-encapsulated giant cell tumor with its typical yellowish-brown color, carefully separated from the digital neurovascular bundle.
Digital Fibroma Excision
Fibromas of the digit can result in gradual pulp enlargement and secondary sensory nerve compromise due to mass effect.
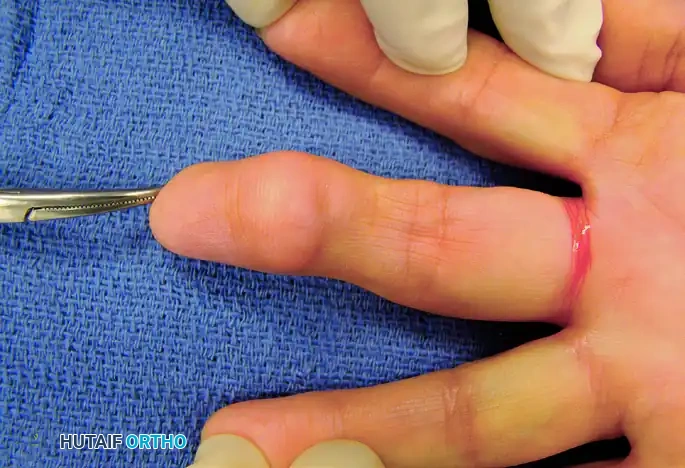
Figure 4: Clinical presentation of a fibroma resulting in gradual pulp enlargement of the digit.
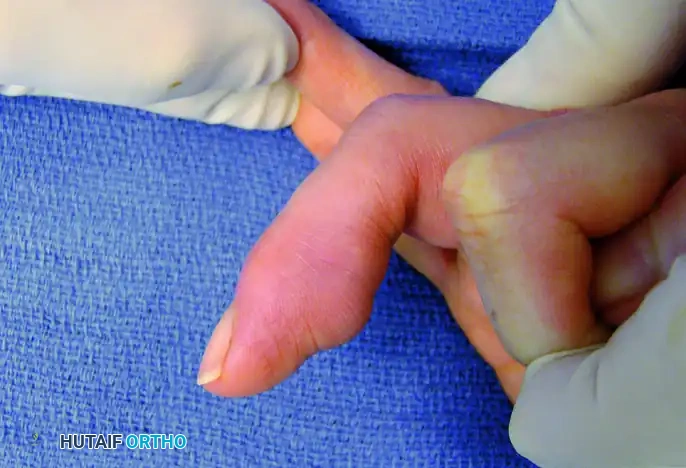
Figure 5: Lateral view demonstrating the asymmetric volar swelling caused by the underlying fibroma.
Surgical excision requires a careful approach to avoid damaging the terminal branches of the digital nerve. A midline palmar oblique approach is often utilized for optimal exposure.
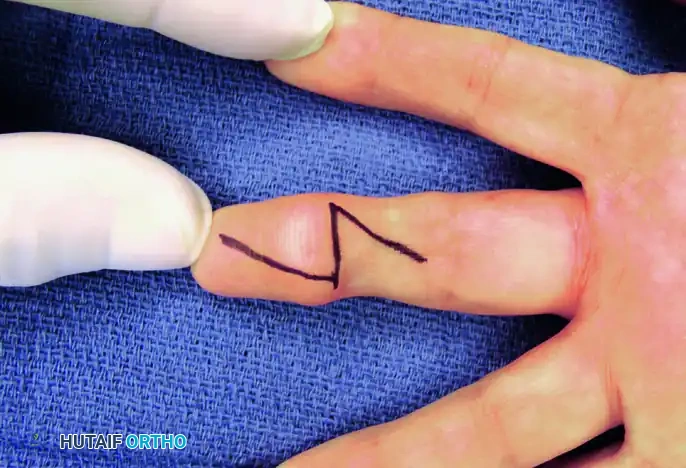
Figure 6: Midline palmar oblique approach marked over the volar aspect of the affected digit.
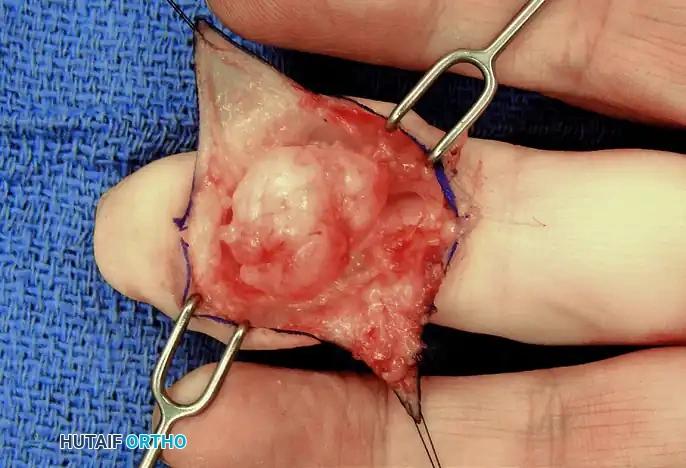
Figure 7: Exposure of the well-encapsulated fibroma tumor in situ, demonstrating displacement of the local subcutaneous tissues.
Once exposed, marginal excision is performed. It is critical to inspect the adjacent joints and tendon sheaths to ensure no satellite lesions are left behind.
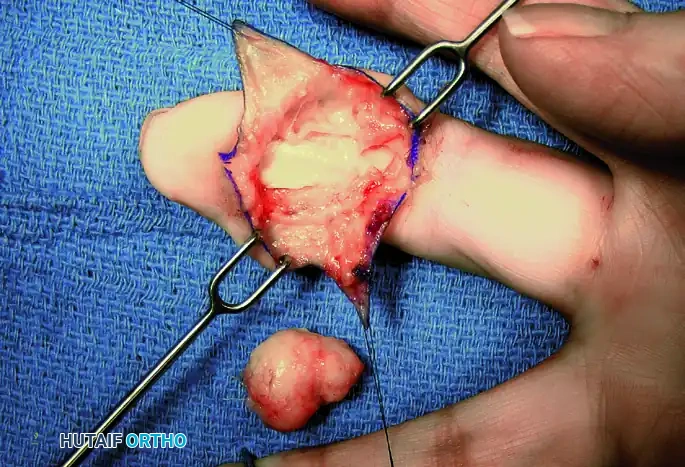
Figure 8: Marginal excision of the tumor, carefully peeling the capsule away from the underlying flexor tendon sheath.
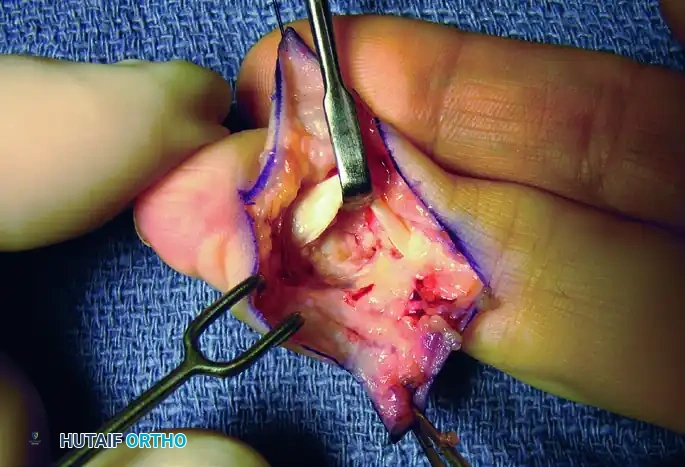
Figure 9: Final inspection of the distal interphalangeal (DIP) joint and flexor sheath for any additional tumor deposits prior to closure.
Malignant Transformation in Peripheral Nerve Sheath Tumors
While discussing soft tissue tumors, it is imperative to note the risk of malignancy in certain patient populations. In patients with Neurofibromatosis Type 1 (von Recklinghausen disease), plexiform neurofibromas carry a significant risk of malignant transformation into Malignant Peripheral Nerve Sheath Tumors (MPNST).
Surgical Warning: Malignant transformation in neurofibromatosis has been reported to occur in up to 15% of patients. Complete, wide excision is absolutely necessary for any neurofibromatous lesions that suddenly enlarge, become painful at rest, or cause new-onset neurologic deficits.
Postoperative Protocol and Rehabilitation
Regardless of the tumor type (hemangioma, glomus, GCTTS, or fibroma), postoperative management follows similar principles aimed at minimizing edema and restoring function:
- Immobilization: A bulky soft dressing with a volar orthosis is applied in the operating room. The hand is immobilized in an intrinsic-plus position (wrist extended 20 degrees, MCP joints flexed 70 degrees, IP joints fully extended) for 3 to 5 days to allow for initial wound healing and hematoma prevention.
- Elevation: Strict elevation above the level of the heart is maintained for the first 48-72 hours to minimize throbbing pain and swelling.
- Early Mobilization: Once the bulky dressing is removed, active and active-assisted range of motion (ROM) exercises are initiated under the guidance of a certified hand therapist.
- Scar Management: Suture removal occurs at 10 to 14 days. Following complete epithelialization, aggressive scar massage and silicone gel sheeting are employed to prevent tendon adhesions and perineural fibrosis.
- Desensitization: Particularly for glomus tumors and fibromas involving the digital pulp, a formal desensitization program (using varying textures and fluidotherapy) is critical to overcome postoperative hypersensitivity.
You Might Also Like
