Vascularized Bone Grafting of Avascular Scaphoid Nonunions
Introduction and Epidemiology
Scaphoid fractures represent the most frequently encountered carpal bone injury in clinical practice, accounting for approximately 60% to 70% of all carpal fractures and predominantly affecting young, active male populations. Despite appropriate initial conservative or operative management, nonunions occur in a clinically significant proportion of cases, with contemporary literature reporting nonunion rates ranging from 5% to 15%. The etiology of scaphoid nonunion is multifactorial, with predisposing variables including delayed diagnosis, inadequate or abbreviated periods of immobilization, initial fracture displacement greater than 1 mm, proximal pole involvement, and the presence of avascular necrosis (AVN). The insidious onset of symptoms and often subtle, easily missed radiographic findings on initial presentation frequently contribute to delayed treatment, exponentially exacerbating the risk of nonunion.
Avascular necrosis of the scaphoid proximal pole represents a particularly formidable reconstructive challenge, directly stemming from the bone's unique, precarious, and retrograde intraosseous blood supply. Once AVN is established, the biological milieu of the proximal fragment is severely compromised. Consequently, the likelihood of achieving successful bony union utilizing conventional non-vascularized bone grafting (NVBG) techniques (e.g., Matti-Russe or Fisk-Fernandez procedures) diminishes considerably, with failure rates often exceeding 50% in the setting of true proximal pole ischemia.
In these complex clinical scenarios, vascularized bone grafting (VBG) has emerged as a critical, limb-salvaging surgical intervention. VBG provides immediate structural support (osteoconduction) while simultaneously introducing a viable, autonomous blood supply containing live osteocytes and osteoblasts (osteogenesis and osteoinduction). This dual mechanism promotes accelerated healing, facilitates revascularization of the ischemic fracture fragments via creeping substitution, and addresses the profound biological deficiencies inherent in avascular nonunions. The ultimate surgical objective is to maximize union rates, restore scaphoid morphology, and prevent the predictable, progressive cascade of carpal collapse and secondary degenerative arthritis, universally recognized as scaphoid nonunion advanced collapse (SNAC).
Surgical Anatomy and Biomechanics
Scaphoid Vascularity
A profound understanding of scaphoid vascularity is the cornerstone of managing scaphoid pathology. The scaphoid’s blood supply is predominantly intraosseous and derives primarily from branches of the radial artery. Classical anatomical studies by Gelberman and Menon demonstrated that the dorsal carpal branch of the radial artery provides the dominant blood supply. This vessel enters the scaphoid dorsally through a dense plexus of vessels within the dorsal capsule and the dorsal intercarpal ligamentous attachments, primarily at the level of the scaphoid waist.
Crucially, these intraosseous vessels traverse the scaphoid in a highly specific distal-to-proximal direction. This retrograde arborization supplies approximately 70% to 80% of the bone, encompassing the entire proximal pole and the proximal aspect of the waist. Minor supplementary contributions arise from the palmar carpal branch and the superficial palmar branch of the radial artery, which enter the distal tubercle and supply the distal 20% to 30% of the bone.
This obligate retrograde blood flow pattern renders the proximal pole exquisitely vulnerable to ischemia following a fracture through the scaphoid waist or proximal pole. A fracture line traversing this region physically disrupts the primary intraosseous vascular channels, immediately isolating the proximal fragment from its blood supply. Microangiographic studies have revealed that in approximately 30% of human scaphoids, there is either a single, highly tenuous vascular channel or no significant collateral channel reaching the proximal pole, underscoring the anatomical predisposition to AVN following trauma.
Scaphoid Biomechanics and Kinematics
Biomechanically, the scaphoid functions as a critical mechanical linkage—an intercalated segment—that bridges the proximal and distal carpal rows. Its complex, twisted "S" shape and robust ligamentous attachments (particularly the scapholunate interosseous ligament and the radioscaphocapitate ligament) enable the coordinated, synchronous movement of the wrist while transmitting substantial axial loads across the radiocarpal joint. During radial deviation of the wrist, the normal scaphoid flexes palmarward; during ulnar deviation, it extends dorsally. This intricate kinematic behavior is an absolute prerequisite for maintaining global carpal stability and preserving a physiological range of motion.
A scaphoid nonunion catastrophically disrupts this delicate biomechanical equilibrium. The loss of cortical integrity leads to a predictable pattern of carpal derangement. Subjected to compressive loads across the wrist, the distal scaphoid fragment tends to flex excessively, while the proximal fragment extends with the lunate. This results in the classic "humpback" deformity of the scaphoid. Concurrently, the uncoupled lunate extends, resulting in a dorsal intercalated segmental instability (DISI) pattern. This abnormal, uncoupled carpal alignment drastically alters joint contact mechanics, exponentially increasing contact pressures at the radioscaphoid and capitolunate articulations. This altered tribology is the direct precursor to the progressive, staged degenerative arthritis characteristic of the SNAC wrist. Therefore, the reconstructive goal with VBG is bipartite: achieving solid bony union and meticulously restoring normal scaphoid length, alignment, and carpal kinematics to halt the progression of arthropathy.
Vascularized Bone Graft Donor Site Anatomy
Several highly reliable sources for vascularized bone grafts have been described and validated in the literature. These are broadly categorized into pedicled grafts (primarily from the distal radius) and free vascularized grafts (predominantly from the medial femoral condyle). The selection of the donor site is dictated by the specific morphological characteristics of the nonunion, the volumetric requirement for bone graft, the presence of deformity, and the microvascular proficiency of the operating surgeon.
1,2-Intercompartmental Supraretinacular Artery (1,2-ICASRA) Pedicle Graft
First popularized by Zaidemberg in 1991, the 1,2-ICASRA graft remains the workhorse pedicled vascularized bone graft for scaphoid nonunions. The 1,2-ICASRA is a consistent branch arising from the radial artery at the level of the radial styloid, typically 5 cm proximal to the radiocarpal joint. It courses dorsally and distally within the retinacular tissue between the first extensor compartment (abductor pollicis longus and extensor pollicis brevis) and the second extensor compartment (extensor carpi radialis longus and brevis). It provides a rich arborization to the dorsal cortex of the distal radius. A robust cortical or corticocancellous bone graft, typically measuring 1.5 cm to 2.5 cm in length, can be safely harvested from the dorsal radius based on this predictable pedicle. Its immediate anatomical proximity to the scaphoid allows for a tension-free transposition, making it an exceptional choice for dorsal approaches to proximal pole AVN.
Distal Radius Palmar Pedicle Grafts
For nonunions approached volarly (often to correct significant humpback deformities), palmar-based pedicled grafts from the distal radius are highly advantageous.
* 2,3- or 2,4-ICASRA Grafts: These grafts rely on minute but consistent vascular branches arising from the palmar aspect of the radial artery, distal to the flexor carpi radialis (FCR) tendon. They course distally between the respective extensor/flexor intervals. While highly effective for volar defects, they are technically more demanding to dissect due to the smaller caliber of the vessels and tighter anatomical confines compared to the dorsal 1,2-ICASRA.
* Anterior Interosseous Artery (AIA) Graft: A vascularized graft can be harvested from the palmar distal radius or ulna, pedicled on the terminal branches of the anterior interosseous artery. While possessing a reliable vascular pedicle, it requires a more extensive dissection and is generally reserved as a secondary option.
Medial Femoral Condyle (MFC) Free Vascularized Graft
For highly complex scaphoid nonunions—specifically those characterized by massive bone loss, extensive cystic degeneration, revision scenarios with scarred local tissue beds, or total avascularity of a large proximal pole—the free vascularized graft from the medial femoral condyle (MFC) is the gold standard. The MFC provides a massive, structurally robust corticocancellous bone block with a highly reliable and prolific blood supply derived from the descending genicular artery (DGA) and its specific medial genicular branch. The MFC graft offers superior osteogenic potential and structural rigidity compared to distal radius grafts. The vascular pedicle (artery and paired venae comitantes) is harvested en bloc with the bone and requires formal microvascular anastomosis to recipient vessels in the anatomical snuffbox or volar forearm (typically the radial artery or its venae comitantes).
Indications and Contraindications
The clinical decision-making process regarding the implementation of vascularized bone grafting is highly nuanced. It requires a comprehensive synthesis of patient demographics, functional demands, precise nonunion morphology, and the biological status of the scaphoid fragments.
Indications for Vascularized Bone Grafting
- Established Scaphoid Nonunion with Proximal Pole Avascular Necrosis (AVN): This represents the absolute, primary indication for VBG. The severely diminished osteogenic potential of a necrotic proximal fragment dictates the necessity of introducing a living, vascularized graft to achieve union.
- Failed Conventional Non-Vascularized Bone Grafting: In the setting of a recalcitrant nonunion following a prior Matti-Russe or similar NVBG procedure, the local biological bed is often depleted and scarred. VBG introduces a fresh, independent blood supply, significantly improving the salvage rate.
- Scaphoid Nonunion with Volumetric Bone Loss: Nonunions presenting with large cystic cavities or significant fragment resorption require substantial structural augmentation. Vascularized grafts (particularly the MFC) provide the necessary volume without succumbing to the central necrosis often seen in massive, non-vascularized autografts.
- Revision Scaphoid Nonunion Surgery: Any revision scenario, particularly those involving prior hardware failure or infection (once eradicated), benefits from the enhanced biological healing environment provided by a VBG.
- Advanced Nonunion Morphology with Deformity: Nonunions exhibiting severe structural collapse (humpback deformity) and dense sclerotic margins indicate a hostile local environment. VBG aids in structural correction while bypassing the poor intrinsic healing capacity of the sclerotic bone.
- Prophylactic VBG in Acute, High-Risk Fractures: In highly selected cases—such as severely comminuted, highly displaced proximal pole fractures in elite athletes or manual laborers—primary VBG may be indicated to preemptively mitigate the exceptionally high risk of AVN and nonunion.
Contraindications for Vascularized Bone Grafting
- Active Local or Systemic Infection: The presence of active osteomyelitis or soft tissue infection at the wrist or the proposed donor site is an absolute contraindication. Eradication of infection is mandatory prior to any reconstructive attempt.
- Severe Degenerative Arthritis (SNAC Wrist Stage III or IV): If advanced, irreversible articular cartilage destruction has occurred, particularly involving the capitolunate joint or the entire radiocarpal articulation, attempting to restore scaphoid anatomy is futile and will not relieve pain. Salvage procedures such as Proximal Row Carpectomy (PRC), Four-Corner Fusion (4CF), or Total Wrist Arthrodesis (TWA) are the indicated interventions.
- Severe Patient Comorbidities: Systemic conditions that severely compromise microvascular flow (e.g., severe peripheral vascular disease, uncontrolled diabetes mellitus, heavy active tobacco use) or preclude prolonged general anesthesia are relative or absolute contraindications, particularly for free tissue transfer.
- Inadequate Recipient Bone Stock: If the proximal pole is entirely fragmented or resorbed to a mere cartilaginous shell, there may be insufficient viable osseous architecture to accept and stabilize a graft.
- Compromised Soft Tissue Envelope: Extensive scarring or prior radiation therapy at the recipient site may preclude safe surgical dissection or compromise the viability of the transferred pedicle.
Operative vs Non-Operative Indications
The management algorithm for scaphoid pathology requires careful stratification. VBG is reserved for the most biologically compromised operative scenarios.
| Feature | Non-Operative Treatment Indications | Operative Treatment Indications (General) | Operative Treatment Indications (Vascularized Bone Graft Specific) |
|---|---|---|---|
| Nonunion Status | Stable, asymptomatic fibrous nonunion in a low-demand patient. No progressive deformity. | Symptomatic nonunion (pain, weakness). Unstable nonunion. Progressive deformity. | Scaphoid nonunion with MRI-proven proximal pole AVN. Failed prior NVBG. |
| Fracture Type/Location | Acute, undisplaced distal pole or waist fractures. | Displaced fractures (>1mm), proximal pole fractures, unstable patterns. | Proximal pole nonunions (high AVN risk), large structural defects, revision surgery. |
| Bone Viability | Proximal pole fully vascularized (confirmed on MRI). | Viable proximal pole, nonunion driven purely by mechanical instability. | Established proximal pole avascularity. Dense sclerotic or cystic fragment margins. |
| Deformity/Collapse | Normal carpal alignment (normal scapholunate angle, no DISI). | Humpback deformity, DISI instability, early SNAC wrist (Stage I-II). | Severe scaphoid collapse requiring structural wedge correction and vascularized bone fill. |
| Patient Factors | Low functional demands, elderly, high surgical risk, absolute refusal of surgery. | Young, active patient, manual laborer, high functional demands. | Young, active patient requiring the highest statistical probability of union in a high-risk biological scenario. |
| Associated Pathology | No concurrent ligamentous injury or degenerative changes. | Associated perilunate instability, early localized degenerative changes. | "Kissing lesion" arthritis (early radioscaphoid degeneration) where joint-sparing salvage is still biologically feasible. |
Pre Operative Planning and Patient Positioning
Exacting preoperative planning is the sine qua non of successful vascularized bone grafting. The procedure demands a rigorous assessment of the bony architecture, vascular status, and soft tissue envelope.
Diagnostic Imaging
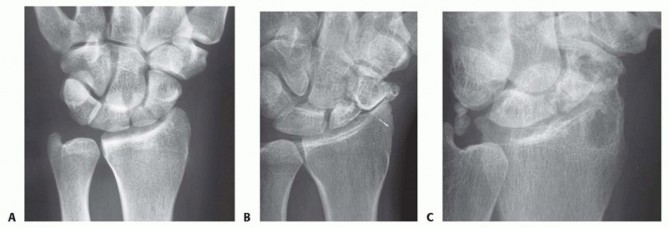
- Plain Radiographs: Standardized orthogonal views (posteroanterior, lateral, oblique) and dedicated scaphoid views (with the wrist in ulnar deviation and slight extension) are the baseline diagnostic modalities. These identify the nonunion, assess for sclerosis and cystic resorption, and evaluate carpal alignment (measuring the scapholunate and radiolunate angles to quantify DISI and humpback deformity).
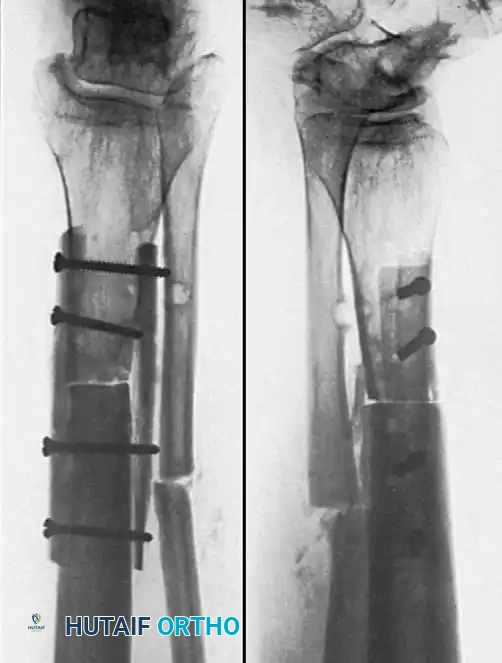
- Computed Tomography (CT) Scan: A high-resolution, thin-cut (1 mm or less) CT scan with multiplanar reconstructions (MPR) oriented along the true longitudinal and transverse axes of the scaphoid is the gold standard for morphological assessment. It precisely quantifies the volumetric bone loss, maps the exact location of the nonunion, and is critical for preoperative templating to determine the required size and shape of the bone graft. CT is also the definitive modality for confirming solid bony union post-operatively.
- Magnetic Resonance Imaging (MRI): MRI without and with intravenous gadolinium contrast is the definitive study for evaluating the vascularity of the proximal pole. A necrotic proximal pole will demonstrate a lack of gadolinium enhancement and a loss of the normal hyperintense fatty marrow signal on T1-weighted images (often appearing hypointense on both T1 and T2 sequences). MRI also provides excellent visualization of the intrinsic carpal ligaments and the integrity of the articular cartilage.
- Arteriography/CT Angiography (CTA): While generally unnecessary for standard pedicled grafts (1,2-ICASRA), preoperative CTA or formal angiography is highly recommended prior to a free MFC graft. This maps the recipient vessels in the forearm (ensuring patency of the radial artery and its venae comitantes) and assesses the donor site vascularity, particularly in patients with a history of prior wrist trauma, multiple surgeries, or peripheral vascular disease.
Clinical Evaluation

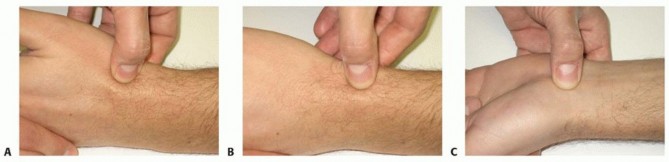
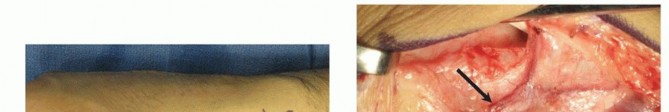
* FIG 1 • A. Exquisite point tenderness at the anatomic snuffbox is the hallmark clinical finding of scaphoid nonunion. B. The radial styloid-scaphoid articulation is the epicenter of early degenerative change (SNAC Stage I); localized tenderness here suggests early arthropathy. C. The distal pole of the scaphoid is palpable at the base of the thenar eminence. Tenderness here can indicate nonunion or concurrent scaphotrapezial-trapezoidal (STT) joint pathology.
A meticulous clinical examination is mandatory. This includes quantifying resting and active pain levels, measuring bilateral wrist range of motion (using a goniometer), and assessing grip strength (using a Jamar dynamometer). The surgeon must palpate for specific areas of tenderness (snuffbox, volar scaphoid tuberosity, scapholunate interval). A thorough neurological examination is required to document any pre-existing median or radial nerve compression neuropathies, which may be exacerbated by post-traumatic edema or surgical positioning.
Preoperative Surgical Planning
Advanced surgical planning requires a multidimensional approach:
1. Graft Selection Algorithm: The choice between a pedicled graft (e.g., 1,2-ICASRA for smaller dorsal defects with AVN) and a free graft (e.g., MFC for massive defects, severe humpback, or revision cases) must be finalized based on CT and MRI findings.
2. Surgical Approach: A dorsal approach is generally utilized for proximal pole AVN and 1,2-ICASRA harvest. A volar (palmar) approach is preferred for correcting severe humpback deformities (allowing for volar wedge placement) or when utilizing palmar-based pedicles.
3. Fixation Strategy: The surgeon must anticipate the required fixation construct. Headless compression screws (e.g., Acutrak, Herbert) are standard, but the specific diameter (mini vs. micro) and length must be templated. In cases of extreme proximal pole fragmentation, K-wire fixation may be the only viable option to avoid shattering the remaining osseous shell.
Anesthesia and Patient Positioning
- Anesthesia: A regional anesthetic block (e.g., ultrasound-guided supraclavicular or axillary brachial plexus block) combined with general anesthesia or deep sedation is optimal. The regional block provides superior intraoperative muscle relaxation, profound postoperative analgesia, and induces a sympathectomy, resulting in peripheral vasodilation which theoretically optimizes flow through the microvascular anastomosis or pedicle.
- Patient Positioning: The patient is positioned supine. The operative arm is placed on a radiolucent hand table. A pneumatic tourniquet is applied to the proximal brachium. The limb is exsanguinated via elevation or an Esmarch bandage prior to tourniquet inflation (tourniquet pressure typically set at 250 mmHg).
- Two-Team Approach (Free Grafts): For MFC free flap procedures, a simultaneous two-team approach is utilized. The ipsilateral lower extremity is prepped and draped from the hip to the ankle. A sterile tourniquet is applied to the proximal thigh. This allows one team to prepare the recipient wrist bed while the second team simultaneously harvests the MFC graft, significantly reducing total operative and ischemia times.
Detailed Surgical Approach and Technique
The execution of a vascularized bone graft requires meticulous tissue handling, precise osteotomies, and, in the case of free grafts, microsurgical expertise. The use of surgical loupes (minimum 3.5x magnification) is mandatory for pedicled grafts, while an operating microscope is required for free tissue transfer.
Surgical Exposure of Scaphoid Nonunion
Dorsal Approach
This approach is the standard for accessing proximal pole nonunions and is seamlessly integrated with the harvest of the dorsal 1,2-ICASRA graft.
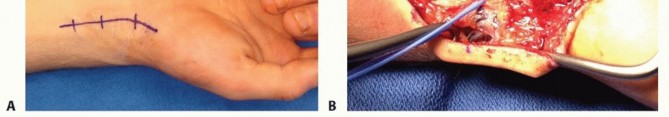
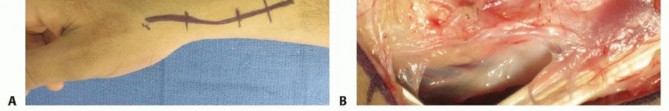
1. Incision: A dorsal longitudinal or slightly curvilinear incision is made, centered over Lister's tubercle and the scapholunate interval, extending from 3 cm proximal to the radiocarpal joint down to the base of the third metacarpal.
2. Superficial Dissection: The skin and subcutaneous tissues are carefully elevated. The dorsal sensory branches of the radial nerve are meticulously identified, mobilized, and protected with vessel loops. Dorsal venous continuity should be preserved whenever possible to minimize postoperative edema.
3. Retinacular Incision: The extensor retinaculum is sharply incised longitudinally precisely between the first dorsal compartment (APL, EPB) and the second dorsal compartment (ECRL, ECRB).
4. Capsular Exposure: The first compartment is retracted radially, and the second is retracted ulnarly. The dorsal radiocarpal capsule is exposed. A ligament-sparing capsulotomy (often a T-shaped or longitudinal incision between the dorsal radiocarpal and dorsal intercarpal ligaments) is performed. The nonunion site is identified. Extreme care is taken during capsular elevation to avoid inadvertent transection of the dorsal carpal branch of the radial artery, which arborizes in this region.
Palmar (Volar) Approach
This approach is utilized for waist nonunions, correction of severe humpback deformities, and harvest of volar pedicled grafts.
1. Incision: A modified Henry approach is utilized, featuring a curvilinear incision along the radial border of the FCR tendon, extending from the distal radius into the proximal palm across the wrist crease.
2. Dissection: The FCR tendon sheath is incised, and the tendon is retracted ulnarly. The radial artery is identified radially and protected. The deep fascial layers are incised.
3. Capsular Exposure: The pronator quadratus is elevated if necessary. The palmar wrist capsule (radioscaphocapitate and long radiolunate ligaments) is incised longitudinally over the scaphoid. This approach provides direct visualization of the scaphoid waist and allows for powerful mechanical correction of the flexed distal fragment.
Debridement and Preparation of Nonunion Site
The preparation of the recipient bed is as critical as the graft harvest itself.
1. Identification and Excision: The fibrous pseudarthrosis tissue filling the nonunion gap is meticulously excised using small curettes and rongeurs.
2. Sclerosis Resection: Dense, avascular sclerotic bone on both the proximal and distal fragments must be aggressively resected back to healthy, punctate bleeding bone. This is typically achieved using a high-speed burr under constant saline irrigation to prevent thermal necrosis. Any cystic cavities are thoroughly curetted.
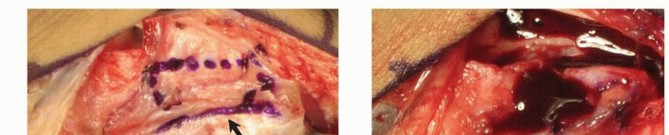
3. Deformity Correction: If a humpback deformity exists, the distal scaphoid fragment is extended. This often creates a volar wedge-shaped defect. The scaphoid length and alignment are provisionally restored and held with a 0.045-inch K-wire serving as a "joystick." Fluoroscopy confirms the restoration of the intrascaphoid angle and the correction of the DISI deformity (restoration of the normal radiolunate angle). The resulting defect is precisely measured to template the required graft dimensions.
Harvesting of Vascularized Bone Graft
1,2-ICASRA Pedicle Graft
* FIG 2 • The anatomical landmarks and incision for the dorsal approach to the scaphoid nonunion and the simultaneous harvesting of the 1,2-ICASRA pedicled vascularized bone graft.
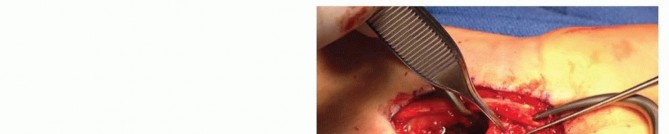
* FIG 3 • High-magnification view of the dissection of the 1,2-ICASRA pedicle. Note its origin from the radial artery and its predictable course within the retinacular tissue between the first and second extensor compartments.
The 1,2-ICASRA graft is harvested from the dorsal metaphysis of the distal radius.
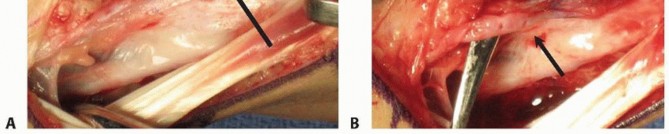
1. Pedicle Identification: Utilizing the proximal extent of the dorsal incision, the radial artery is identified in the anatomical snuffbox. The 1,2-ICASRA pedicle is visualized coursing superficially on the extensor retinaculum.
2. Pedicle Mobilization: Using tenotomy scissors and bipolar electrocautery, a cuff of retinacular tissue (approximately 3-5 mm wide) is left attached to the pedicle (artery and venae comitantes) to prevent vasospasm and protect the delicate vessels. The pedicle is dissected proximally to its origin on the radial artery to maximize arc of rotation.
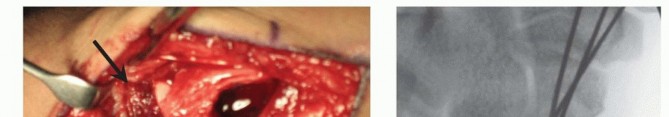
3. Bone Harvest: A rectangular or trapezoidal cortical bone graft (typically 1.5 x 1.0 cm) is marked on the dorsal radius, incorporating the distal insertion of the pedicle. Using a thin osteotome or an oscillating microsaw, the corticocancellous block is harvested. The cut is made 4-5 mm deep to include highly vascular cancellous bone.
4. Confirmation of Perfusion: The tourniquet is temporarily deflated. The surgeon must visually confirm active, pulsatile bleeding from the cancellous surface of the harvested graft before proceeding with insertion.
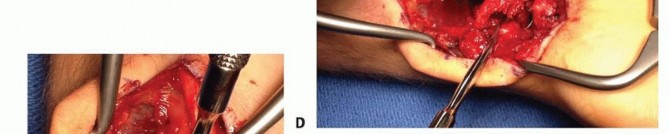
* FIG 4 • The completely harvested 1,2-ICASRA graft. The corticocancellous bone block is clearly visible, suspended by its robust, intact vascular pedicle, ready for transposition.
* FIG 5 • Insertion of the 1,2-ICASRA graft into the prepared dorsal scaphoid nonunion site. The pedicle is carefully routed to avoid kinking or tension.
Distal Radius Palmar Pedicle Grafts (e.g., Palmar ICASRA, AIA)
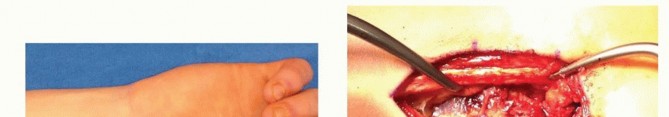
* FIG 6 • The extended volar (Henry) incision utilized for a palmar approach to the scaphoid and the harvesting of a palmar-based pedicled vascularized bone graft.

* FIG 7 • Meticulous microsurgical dissection of a palmar pedicle from the distal radius. These vessels, often based on transverse branches of the radial artery near the FCR tendon, require delicate handling.
- Vessel Identification: Through the volar approach, transverse vascular branches originating from the radial artery and coursing across the volar metaphysis of the radius are identified using loupe magnification.
- Bone Harvest: Once a suitable pedicle is isolated, a corticocancellous block is outlined on the volar radius. The graft is harvested with an osteotome, ensuring the pedicle remains securely attached to the periosteum of the graft.
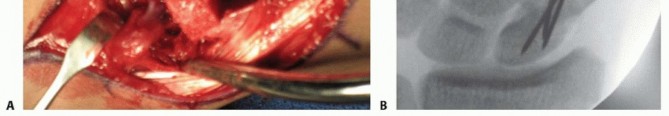
* FIG 8 • A successfully harvested palmar pedicled bone graft, demonstrating the vascular leash attached to the cortical surface.
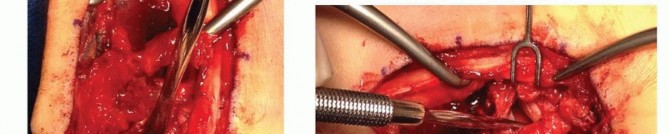
* FIG 9 • The palmar-based pedicled vascularized bone graft is press-fit into the volar scaphoid defect, acting as a structural wedge to correct the humpback deformity while providing revascularization.
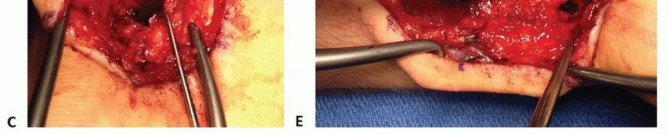
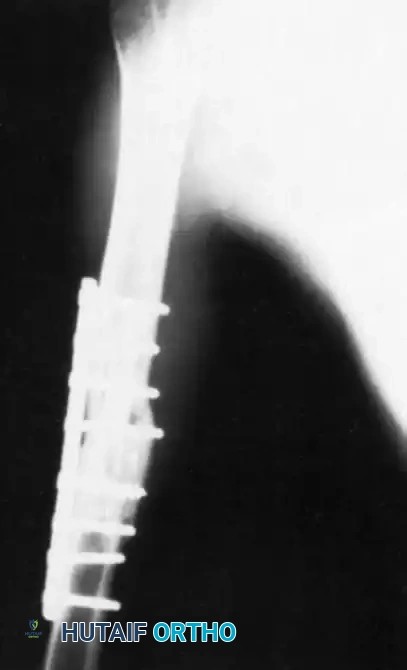
* FIG 10 • Intraoperative fluoroscopic imaging confirming proper placement of the volar graft, restoration of scaphoid length, and stable internal fixation.
Medial Femoral Condyle (MFC) Free Vascularized Graft
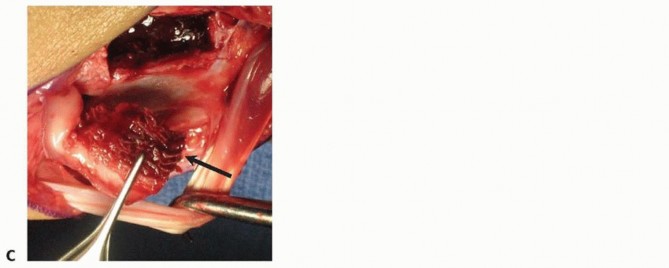
* FIG 11 • Detailed anatomical illustration depicting the vascular supply to the medial femoral condyle, highlighting the descending genicular artery (DGA
Clinical & Radiographic Imaging
You Might Also Like