Mastering Operative Treatment of Radius & Ulna Nonunions
Key Takeaway
This topic focuses on Mastering Operative Treatment of Radius & Ulna Nonunions, A forearm nonunion is a diaphyseal fracture showing no healing progression or likelihood of union. The incidence in the radius is 2%, often linked to bone deficits or inadequate fixation. The treatment of radius fractures is typically operative to restore the vital radial bow, essential for forearm rotation. This addresses issues like hypertrophic nonunion and ensures proper function.
Introduction and Epidemiology
A diaphyseal forearm fracture must be clinically and radiographically evaluated for nonunion when there is either no likelihood that the fracture will progress to union, such as in the presence of a large segmental bone defect, or if the fracture has ceased to demonstrate any progression of healing over a period of consecutive months. The forearm is a complex functional unit, and the failure of diaphyseal healing severely compromises upper extremity kinematics. Secondary to the advent of dynamic compression plating and modern principles of absolute stability, the incidence of forearm nonunions in the primary setting is low. Historical and contemporary literature cites nonunion rates in the radius of approximately 2 percent and the ulna of 4 percent following appropriate open reduction and internal fixation.
Despite these low primary rates, when nonunions do occur, they present a formidable reconstructive challenge. Forearm nonunions do not heal without surgical intervention. The resultant loss of stability of one or both bones unhinges the entire mechanical axis of the upper extremity, leading to progressive deformity, weakness, and loss of pronosupination. The etiology of these nonunions is multifactorial, encompassing mechanical failure, biological compromise, and infection. In the case of a single-bone injury, whether radius or ulna, any bone deficit at the fracture site significantly increases the risk of nonunion. In this scenario, the intact, uninjured bone acts as a rigid distracting force, preventing compressive loading across the fracture site of the injured bone.
Diaphyseal comminution at the fracture site increases the incidence of nonunion to up to 12 percent despite plate fixation. High-energy mechanisms, particularly ballistic injuries and gunshot blasts, frequently result in extensive comminution, periosteal stripping, and soft tissue compromise. Open both-bone forearm fractures are frequently associated with bone loss at the fracture site, loss of the fracture hematoma, and an increased infection rate, all of which precipitate nonunion. Consequently, comminuted open fractures with significant bone loss exhibit the highest rates of nonunion. Furthermore, inadequate internal fixation that fails to withstand the high torsional stresses involved in forearm rotation, coupled with poor surgical technique, remains a frequent iatrogenic cause of hypertrophic nonunion.
Surgical Anatomy and Biomechanics
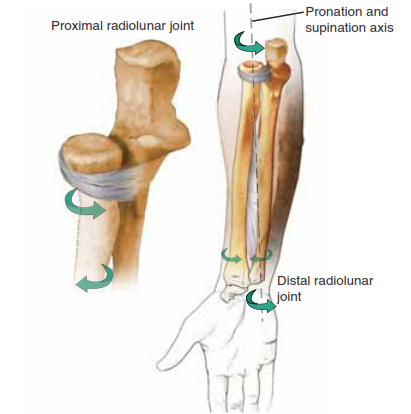
The forearm functions biomechanically as a complex, two-bone joint. The ulna serves as a straight, stable axis around which the bowed radius rotates to facilitate pronation and supination. This relationship is critical for the spatial positioning of the hand.

The anatomical bow of the radius is characterized by an apex radial and apex dorsal curvature. The maximum radial bow typically occurs at approximately 60 percent of the length of the radius measured from the bicipital tuberosity to the radiocarpal joint. Restoration of this precise curvature during nonunion surgery is non-negotiable; failure to restore the radial bow results in a mechanical block to pronosupination and alters the load transmission across the radiocapitellar and ulnocarpal joints.

The distal radioulnar joint, the proximal radioulnar joint, and the interosseous membrane are the primary stabilizers that bind the two bones together. The interosseous membrane, particularly its central band, is a stout ligamentous structure that transfers axial loads from the radius to the ulna. There is inherent length variability built into the kinematic relationship between the radius and ulna: the radius is at its relative longest in full supination and relative shortest during full pronation. Despite this dynamic variance, there is a very close coordination of length between the two bones that is paramount to normal forearm function.
Extrinsic and intrinsic hand extensors and flexors originate in the forearm, as do the primary wrist flexors and extensors. Additionally, the forearm provides passage to critical neural and vascular elements, including the median, ulnar, and radial nerves, along with their respective vascular bundles. Forearm nonunions, depending on their etiology and the number of previous surgical interventions, typically result in a considerable amount of dense, fibrotic scarring. This scar tissue obliterates normal internervous tissue planes, tethering neurovascular structures and significantly complicating surgical dissection during nonunion takedown.
Pathogenesis and Classification of Nonunions
Forearm diaphyseal nonunions are broadly classified based on their biological activity into hypertrophic, oligotrophic, and atrophic variants. Understanding the biological and mechanical environment of the nonunion is essential for preoperative planning.

Hypertrophic Nonunions
Hypertrophic nonunions possess excellent biological healing potential but lack adequate mechanical stability. They are characterized radiographically by abundant, "elephant foot" or "horse hoof" callus formation that fails to bridge the fracture gap. The primary problem is excessive strain at the fracture site, usually secondary to inadequate plate fixation, premature weight-bearing, or hardware failure. The treatment principle for hypertrophic nonunions is the provision of absolute mechanical stability, typically through rigid compression plating, which allows the existing biological potential to consolidate the fracture. Bone grafting is rarely necessary.
Atrophic Nonunions
Atrophic nonunions lack both mechanical stability and biological healing potential. Radiographically, there is a complete absence of callus formation, and the bone ends are typically sclerotic, rounded, or osteopenic. Because many of the high-energy injuries that result in nonunion involve a segmental bone defect or severe periosteal stripping, the vast majority of diaphyseal nonunions of the forearm are atrophic in nature. The problem in an atrophic nonunion is a fundamental lack of biological activity. Treatment requires excision of the sclerotic bone ends, opening of the medullary canals, rigid internal fixation, and the introduction of osteoinductive and osteoconductive materials, most commonly autologous iliac crest bone graft.
Infected Nonunions
Infected nonunions represent the most challenging subset. They may present with active drainage, sinus tracts, or subtle signs such as chronic pain and elevated inflammatory markers. Eradication of the infection through radical debridement of dead bone and soft tissue, hardware removal, and targeted systemic or local antibiotic therapy (e.g., antibiotic-impregnated cement spacers) must precede or accompany any definitive stabilization and bone grafting efforts.
Indications and Contraindications
The management of forearm nonunions is overwhelmingly operative. Nonoperative management is reserved for a highly select, medically fragile patient population.
| Parameter | Operative Management | Non Operative Management |
|---|---|---|
| Indications | Atrophic nonunion with or without bone defect | Extreme medical comorbidities precluding anesthesia |
| Hypertrophic nonunion with hardware failure | Asymptomatic nonunion in a low-demand elderly patient (rare) | |
| Infected nonunion requiring debridement | Patient refusal of surgical intervention | |
| Symptomatic deformity or loss of pronosupination | Severe, untreatable active systemic infection | |
| Impending hardware failure or catastrophic pullout | ||
| Contraindications | Active untreated infection (for definitive grafting) | High-demand patient requiring upper extremity weight-bearing |
| Inadequate soft tissue coverage (requires flap first) | Painful, mobile nonunion | |
| Medically unstable patient | Progressive angular deformity |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is the cornerstone of successful forearm nonunion surgery. The surgeon must evaluate the soft tissue envelope, the status of the neurovascular structures, and the exact geometry of the nonunion.

Clinical and Radiographic Evaluation
Physical examination must document the current range of motion of the elbow, wrist, and forearm pronosupination. A meticulous neurological exam is mandatory, particularly assessing the posterior interosseous nerve and the superficial sensory branch of the radial nerve, which are at high risk during revision exposures.
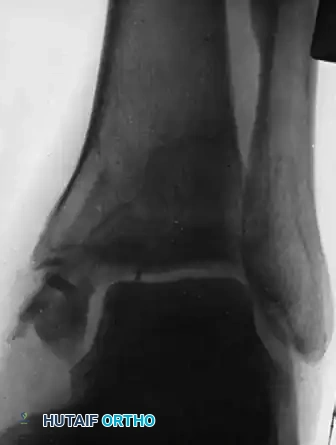
Standard orthogonal anteroposterior and lateral radiographs of the entire forearm, including the elbow and wrist, are required. Computed tomography is highly recommended to assess the true extent of bone loss, identify occult areas of bridging synostosis, and evaluate the precise geometry of the sclerotic bone ends. Contralateral, uninjured forearm radiographs should be obtained to serve as a template for restoring the native radial bow and ulnar variance. Inflammatory markers (C-reactive protein and erythrocyte sedimentation rate) should be drawn to rule out indolent infection.
Patient Positioning and Preparation
The patient is positioned supine on the operating table with the affected extremity extended on a radiolucent hand table. A non-sterile pneumatic tourniquet is placed high on the brachium. The entire upper extremity, including the iliac crest (if autogenous bone grafting is planned), is prepped and draped in a standard sterile fashion. Fluoroscopy must be positioned to enter from the head or the contralateral side of the table to allow unhindered orthogonal imaging of the forearm.
Detailed Surgical Approach and Technique
Surgical exposure in the setting of a nonunion is fraught with difficulty due to altered anatomy and dense scar tissue. The surgeon must meticulously identify and develop the native internervous planes proximal and distal to the zone of injury before navigating into the scarred nonunion site.
Surgical Approaches to the Radius
For the radius, the two primary approaches are the volar (Henry) approach and the dorsal (Thompson) approach. The choice depends on the location of the nonunion, the position of prior incisions, and the planned position of the plate.

The Volar Henry Approach
The volar approach utilizes the internervous plane between the brachioradialis (innervated by the radial nerve) and the flexor carpi radialis (innervated by the median nerve). It is highly versatile and allows exposure of the entire volar surface of the radius.
1. Incision: A longitudinal incision is made along the line connecting the biceps tendon proximally to the radial styloid distally.
2. Superficial Dissection: The fascia between the brachioradialis and flexor carpi radialis is incised. The superficial sensory branch of the radial nerve lies on the undersurface of the brachioradialis and must be protected.
3. Deep Dissection: Proximally, the radial artery and its recurrent branches (the "leash of Henry") are identified, ligated, and divided to allow lateral retraction of the brachioradialis. The supinator muscle is identified. To protect the posterior interosseous nerve, the supinator is sharply elevated off its ulnar insertion and reflected radially. Distally, the pronator teres and pronator quadratus are elevated subperiosteally to expose the nonunion site.

The Dorsal Thompson Approach
The dorsal approach is often preferred for proximal and middle third radius nonunions, utilizing the internervous plane between the extensor carpi radialis brevis (radial nerve) and the extensor digitorum communis (posterior interosseous nerve).
1. Incision: A straight line from the lateral epicondyle to the dorsal radial tubercle (Lister's tubercle).
2. Dissection: The fascia is incised, and the plane between the extensor carpi radialis brevis and extensor digitorum communis is developed. The outcropping muscles (abductor pollicis longus and extensor pollicis brevis) are mobilized. The supinator is exposed, and the posterior interosseous nerve is identified as it emerges from the supinator. The muscle is split meticulously to expose the radial shaft.
Surgical Approach to the Ulna

The ulna is approached via a direct subcutaneous route along its subcutaneous border. This utilizes the internervous plane between the extensor carpi ulnaris (posterior interosseous nerve) and the flexor carpi ulnaris (ulnar nerve). The incision is made directly over the palpable border of the ulna, excising previous scar tissue as necessary. Subperiosteal elevation exposes the nonunion.
Nonunion Takedown and Preparation
Once the nonunion site is exposed, any broken or loose hardware is removed. In atrophic nonunions, the fibrous nonunion tissue is radically excised using a rongeur and curettes until healthy, bleeding cortical bone is encountered (the "paprika sign"). The sclerotic medullary canals of both the proximal and distal fragments must be aggressively opened using a drill or a small burr. This step is critical to restore endosteal blood supply and allow the migration of osteoprogenitor cells into the fracture gap.
In hypertrophic nonunions, the callus is preserved as much as possible, as it represents a highly vascularized, osteogenic environment. The nonunion is mobilized just enough to achieve anatomic alignment.
Reduction and Plate Fixation
Restoration of length, alignment, and rotation is mandatory. The radial bow must be meticulously recreated.

Fixation is achieved using a 3.5-millimeter limited-contact dynamic compression plate or a locking compression plate. Biomechanical principles dictate the use of a plate long enough to achieve a minimum of six, but preferably eight, cortices of fixation on either side of the nonunion.
1. Plate Contouring: The plate must be perfectly contoured to the native anatomy. For the radius, this means matching the apex radial and dorsal bow. Under-contouring or over-contouring will result in malreduction and subsequent loss of pronosupination.
2. Compression: If the nonunion is hypertrophic or if the atrophic ends have been resected to create a flush interface, absolute stability is achieved via axial compression. Articulated tensioning devices or eccentric drilling in the dynamic compression holes are utilized.
3. Defect Management: If a segmental defect exists after debridement, acute shortening is generally contraindicated as it alters the radioulnar variance and disrupts the distal radioulnar joint. The defect must be bridged with a longer plate, maintaining length, and the void filled with structural or cancellous graft.
Bone Grafting Techniques

For atrophic nonunions, autologous cancellous bone graft from the anterior or posterior iliac crest remains the gold standard due to its unmatched osteogenic, osteoinductive, and osteoconductive properties. The graft is tightly packed into and around the nonunion site, taking care not to spill graft into the interosseous space, which could precipitate a radioulnar synostosis.
For segmental defects greater than 2 to 3 centimeters, a structural autograft (e.g., tricortical iliac crest) or the induced membrane technique (Masquelet) is required. The Masquelet technique involves a first stage of radical debridement, hardware placement, and insertion of a polymethylmethacrylate cement spacer. Six to eight weeks later, the spacer is removed, leaving a bioactive pseudomembrane into which copious cancellous autograft is packed. For massive defects (>6 centimeters), a free vascularized fibular graft is the reconstructive procedure of choice.
Complications and Management
The surgical management of forearm nonunions is technically demanding and carries a significant complication profile due to the compromised baseline state of the limb.

Hardware failure is a devastating complication that typically results from an ongoing lack of biological healing, leading to fatigue failure of the plate or screws. It necessitates a complete revision of the nonunion, often requiring a longer plate, orthogonal plating, and renewed bone grafting.

Infection following nonunion surgery requires aggressive surgical debridement. Suppressive antibiotics alone are insufficient in the presence of hardware and dead bone. If the hardware remains stable, it may be retained while the infection is treated with serial debridements and targeted intravenous antibiotics. If the hardware is loose, it must be removed, the canal reamed, and stabilization achieved via external fixation or antibiotic-impregnated intramedullary nails until the infection is eradicated.
| Complication | Estimated Incidence | Management and Salvage Strategies |
|---|---|---|
| Hardware Failure | 5-10% | Revision open reduction internal fixation, longer plate, autogenous bone grafting, consider dual orthogonal plating. |
| Deep Infection | 3-8% | Radical debridement, hardware removal if loose, antibiotic cement spacer, delayed reconstruction (Masquelet technique). |
| Radioulnar Synostosis | 2-6% | Observation if asymptomatic. Surgical excision of the bony bridge and placement of interpositional material (e.g., fat pad, synthetic membrane) delayed until mature (usually >6 months). |
| Nerve Palsy (PIN or SRN) | 1-5% | Most are neurapraxias due to traction. Observation and supportive splinting. Tendon transfers if no recovery at 6-12 months. |
| Persistent Nonunion | 5-15% | Re-evaluation of mechanical stability and biological vitality. Revision plating with enhanced bone grafting (e.g., BMP-2, ICBG). |
| Stiffness / Loss of ROM | 20-40% | Aggressive physical therapy. Surgical release (capsulectomy) rarely indicated unless functional deficit is severe. |
Post Operative Rehabilitation Protocols
The postoperative rehabilitation protocol must balance the need for mechanical protection of the healing nonunion with the necessity of early motion to prevent debilitating stiffness.

In cases where rigid, absolute stability is achieved (e.g., hypertrophic nonunion treated with compression plating), the patient may be placed in a soft dressing and allowed early active range of motion of the elbow, wrist, and forearm within the first week. Weight-bearing and lifting are strictly prohibited until radiographic evidence of bridging callus or obliteration of the fracture lines is observed.
Conversely, in atrophic nonunions with segmental defects requiring structural grafting or in cases of tenuous fixation due to poor bone quality, a period of immobilization is warranted. The extremity is placed in a long-arm cast or a rigid Muenster splint for 4 to 6 weeks. Following this period of protection, progressive active and active-assisted range of motion exercises are initiated under the strict guidance of an occupational or physical therapist.

Radiographic surveillance is conducted at 2, 6, 12, and 24 weeks postoperatively. Clinical union is defined as the absence of pain with palpation and torsional stressing of the forearm. Radiographic union is defined as the presence of bridging trabecular bone across at least three of four cortices on orthogonal views. Full return to heavy labor or high-impact activities is typically delayed until 6 to 9 months postoperatively, contingent upon complete radiographic consolidation.
Summary of Key Literature and Guidelines
The academic foundation for the operative treatment of forearm nonunions is built upon decades of biomechanical and clinical research. The principles of absolute stability, pioneered by the AO Foundation (Arbeitsgemeinschaft für Osteosynthesefragen), remain the gold standard.
Classic studies by Anderson et al. established the efficacy of dynamic compression plating in diaphyseal forearm fractures, dramatically reducing the historical nonunion rates associated with cast immobilization or inadequate fixation. Ring and colleagues extensively reviewed the management of atrophic and hypertrophic forearm nonunions, reinforcing that hypertrophic nonunions require only mechanical stabilization via compression plating, whereas atrophic nonunions mandate aggressive debridement, rigid fixation, and the addition of autologous bone graft.
Contemporary guidelines emphasize the critical importance of restoring the radial bow. Schemitsch and Richards demonstrated a direct correlation between the accurate restoration of the radial bow and the functional recovery of forearm rotation. Furthermore, the evolution of the induced membrane technique by Masquelet has revolutionized the management of critical-sized diaphyseal defects, providing a reliable alternative to complex microvascular free tissue transfers in selected patients.
In summary, mastering the operative treatment of radius and ulna nonunions demands a profound understanding of forearm kinematics, meticulous preoperative planning, advanced surgical dissection skills to navigate altered anatomy, and strict adherence to the biomechanical principles of rigid internal fixation and biological augmentation.
Clinical & Radiographic Imaging













You Might Also Like