Masterclass: Closed Reduction & Percutaneous Pinning for Supracondylar Humerus Fractures
Key Takeaway
Step into the operating room for an in-depth masterclass on closed reduction and percutaneous pinning of pediatric supracondylar humerus fractures. This guide covers essential surgical anatomy, meticulous preoperative planning, and granular, real-time intraoperative execution for both lateral-entry and crossed-pin techniques. Learn critical pearls, pitfalls, and comprehensive postoperative management to achieve optimal outcomes for these common injuries.
Welcome, colleagues, to the operating theater. Today, we are addressing a ubiquitous and often highly challenging pediatric injury: the supracondylar fracture of the humerus. This is a foundational, critical skill for any orthopedic surgeon. Mastery of this procedure requires not only technical dexterity but also a profound understanding of pediatric anatomy, fracture biomechanics, and spatial reasoning. In this masterclass, we will systematically walk through every phase of management, from the initial emergency department assessment to the final percutaneous pin placement, with an uncompromising emphasis on precision, anatomic restoration, and patient safety.
Comprehensive Introduction and Patho-Epidemiology
Supracondylar fractures of the humerus represent a massive proportion of pediatric elbow injuries, accounting for up to 67% of all children hospitalized with elbow trauma and approximately 17% of all childhood fractures overall. The epidemiological peak for these injuries is sharply defined, typically occurring in children between the ages of 5 and 7 years. This specific age distribution is not coincidental; it correlates directly with a period of rapid skeletal growth, increased physical activity, and a relatively high degree of physiologic ligamentous laxity. During this developmental window, the metaphyseal bone of the distal humerus is undergoing significant remodeling, rendering it structurally vulnerable to the massive bending moments generated during a fall.
The vast majority of these injuries—approximately 97%—are classified as extension-type fractures. The classic mechanism of injury involves a fall onto an outstretched hand (FOOSH) with the elbow locked in full extension. Biomechanically, in this position, the olecranon process of the ulna is driven rigidly into the olecranon fossa of the distal humerus. The olecranon effectively acts as a solid bony fulcrum, concentrating immense bending and shear forces directly across the remarkably thin supracondylar region. This anatomical choke point, combined with the transmission of force through the robust anterior joint capsule (which inserts distal to the olecranon fossa but proximal to the physis), leads to a highly predictable and consistent fracture pattern characterized by posterior displacement of the distal fragment.
While extension-type injuries dominate the clinical landscape, flexion-type fractures represent the remaining 3% and warrant careful consideration. These typically result from direct, high-energy trauma to the posterior aspect of the flexed elbow, driving the distal fragment anteriorly. The pathomechanics here are entirely distinct, often lacking the predictable periosteal hinges seen in extension injuries, which makes closed reduction maneuvers fundamentally different and frequently more challenging. Furthermore, the baseline ligamentous laxity inherent to the pediatric population frequently permits transient hyperextension of the elbow joint beyond neutral, exacerbating the fulcrum effect of the olecranon and predisposing these children to the classic extension-type failure cascade.
Detailed Surgical Anatomy and Biomechanics
Before a surgeon even considers manipulating a supracondylar fracture or driving a percutaneous wire, an absolute, three-dimensional mastery of the regional anatomy is mandatory. The distal humerus is a complex, highly contoured region, and we must be acutely aware of its osteology, the surrounding muscular intervals, and, most critically, the neurovascular structures that are placed at immediate risk by both the injury and our interventions.
Osteology and Biomechanical Vulnerabilities
The distal humerus is characterized by its broad, articular condylar mass—comprising the capitellum laterally and the trochlea medially—which abruptly transitions into the narrow, flattened supracondylar metaphysis. This region is the anatomical "weak link" of the pediatric elbow. It is exceptionally thin in the anteroposterior plane, essentially forming a bony wafer between the olecranon fossa posteriorly and the coronoid fossa anteriorly. These fossae, designed to accommodate the ulnar processes during the extremes of motion, create a significant structural stress riser. When subjected to the hyperextension bending moment, the bone predictably fails through this thinnest cross-section.
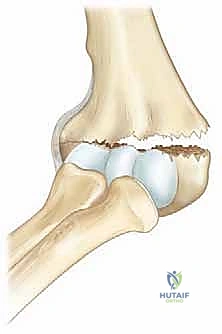
Critically important to the long-term outcome is the growth dynamic of this region. The distal humeral physis contributes only about 20% to the overall longitudinal growth of the humerus. Because the remodeling potential of any fracture is directly proportional to the proximity and activity of the adjacent physis, supracondylar fractures possess very limited capacity to correct angular deformities over time. While sagittal plane deformities (flexion/extension) may remodel slightly in very young children due to the plane of joint motion, coronal plane deformities (varus/valgus) and rotational malalignments will not remodel. This biological limitation mandates near-anatomic reduction in the operating room to prevent permanent, unsightly, and potentially dysfunctional deformities such as cubitus varus (the classic "gunstock" deformity).
Neurovascular Topography and Structures at Risk
The intimate, unyielding relationship of major nerves and vessels to the distal humerus makes meticulous neurovascular assessment and protection the absolute cornerstone of this procedure. The displacement pattern of the fracture dictates which structures are in immediate peril.
The Brachial Artery courses directly anterior to the elbow joint, tethered by the bicipital aponeurosis (lacertus fibrosus). It is at extreme risk in posterolaterally displaced fractures. In this pattern, the sharp, jagged proximal metaphyseal spike is driven anteromedially, directly into the neurovascular bundle. Compression, intimal tear, or frank transection can occur, leading to devastating ischemic consequences, including compartment syndrome or the dreaded Volkmann's ischemic contracture if not rapidly recognized and addressed.
The Median Nerve runs intimately with the brachial artery, located just medial to it. It shares the same vulnerability profile in posterolaterally displaced fractures. The proximal fragment can spear or stretch the nerve over its bony edge.
* The Anterior Interosseous Nerve (AIN), a major motor branch of the median nerve, is the most frequently injured nerve overall in supracondylar fractures. Because it is tethered posteriorly as it branches, it is highly susceptible to traction. Clinically, AIN neuropraxia manifests as an inability to flex the interphalangeal joint of the thumb and the distal interphalangeal joint of the index finger, resulting in an absent "OK" sign.
The Radial Nerve courses laterally in the distal arm, piercing the lateral intermuscular septum to enter the anterior compartment. It is primarily at risk with posteromedially displaced fractures, where the proximal fragment penetrates anterolaterally. Injury here results in deficits in wrist, finger, and thumb extension, as well as sensory loss in the dorsal first web space.
The Ulnar Nerve is located posteriorly, tightly constrained within the cubital tunnel behind the medial epicondyle. While rarely injured by the initial trauma in extension-type fractures (it is more at risk in flexion-type), it is highly vulnerable to iatrogenic injury during medial percutaneous pin placement. As the elbow is flexed to facilitate reduction, the ulnar nerve frequently subluxates anteriorly, draping directly over the medial epicondyle where the pin entry site is targeted.
Muscular Intervals and Periosteal Hinge Dynamics
The soft tissue envelope, particularly the periosteum and the brachialis muscle, plays a dual role: it can be a formidable obstacle to reduction, or it can be the surgeon's greatest ally. The brachialis muscle lies directly anterior to the distal humerus. In severely displaced (Gartland Type III) fractures, the sharp proximal spike frequently penetrates the brachialis muscle and the overlying anterior brachial fascia. The bone becomes "buttonholed" or entrapped within this muscular rent. Clinically, this manifests as the pathognomonic "pucker sign" (or dimple sign) on the anterior skin, indicating severe soft tissue interposition that will make closed reduction significantly more challenging, often requiring specific milking maneuvers to extract the bone.
Understanding the integrity of the periosteal sleeve is the key to successful closed reduction. In typical extension fractures, the anterior periosteum tears completely. However, a posterior periosteal hinge often remains intact.
* With posteromedial displacement, the periosteum tears laterally, leaving an intact medial hinge. To utilize this hinge, the surgeon must pronate the forearm during reduction to tension the medial periosteum and close the lateral gap.
* Conversely, with posterolateral displacement, the periosteum tears medially, leaving an intact lateral hinge. Here, the surgeon must supinate the forearm to tension the lateral periosteum and close the medial gap. Recognizing these hinges allows the surgeon to use the soft tissues to guide the bone fragments back into anatomical alignment.
Exhaustive Indications and Contraindications
Decision-making in supracondylar humerus fractures relies heavily on the modified Gartland classification system, which categorizes fractures based on the degree of displacement and the integrity of the periosteal hinges. Proper classification directly dictates the surgical indications.
The classification is as follows: Type I fractures are nondisplaced or minimally displaced (<2mm) with an intact anterior humeral line. Type II fractures feature a displaced capitellum posterior to the anterior humeral line, but an intact posterior cortex (a functional posterior hinge). Type III fractures are completely displaced with no cortical contact, further subdivided into IIIA (posteromedial) and IIIB (posterolateral). Finally, Leitch introduced the Type IV fracture, defined by complete multidirectional instability (incompetent periosteum circumferentially), which is often only diagnosed intraoperatively under fluoroscopy when the fracture falls into both flexion and extension.
Operative intervention via Closed Reduction and Percutaneous Pinning (CRPP) is the gold standard for virtually all displaced supracondylar fractures to prevent malunion and minimize the risk of compartment syndrome.
| Parameter | Indications for CRPP | Indications for Open Reduction | Contraindications to CRPP |
|---|---|---|---|
| Fracture Type | Gartland Type II, III, and IV fractures. | Irreducible Type II/III/IV via closed means. | Truly nondisplaced Gartland Type I fractures (non-op). |
| Soft Tissue | Significant swelling requiring stabilization. | Open fractures requiring formal debridement. | Active, untreated severe infection at pin entry sites. |
| Vascular Status | "Pink, pulseless" hand that remains well-perfused. | "White, pulseless" hand not resolving with reduction. | Unexplored, persistent vascular ischemia. |
| Associated Injuries | Ipsilateral forearm fractures (floating elbow). | Entrapped neurovascular bundle (e.g., in the fracture site). | Severe medical comorbidities precluding anesthesia (relative). |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning and meticulous operating room setup are not mere formalities; they are critical steps that separate a smooth, efficient 20-minute case from a frustrating, prolonged ordeal. The preparation begins the moment the patient arrives in the emergency department.
Clinical and Neurovascular Assessment
A complete, rigorously documented neurovascular examination of the involved extremity is absolutely critical. This establishes the baseline and dictates the urgency of the intervention. The examination must be specific and deliberate:
* Anterior Interosseous Nerve (AIN): Test motor function by asking the child to make an "OK" sign. Inability to flex the IP joint of the thumb (flexor pollicis longus) and the DIP joint of the index finger (flexor digitorum profundus) indicates AIN palsy.
* Radial Nerve: Assess motor function via wrist extension and thumb interphalangeal extension (extensor pollicis longus). Test sensation in the dorsal first web space.
* Ulnar Nerve: Assess motor function by having the child cross their fingers or abduct/adduct against resistance (interossei). Test sensation on the volar aspect of the little finger.
* Vascular Status: Palpate the radial and ulnar pulses. Assess capillary refill, skin color, and temperature. A "pink, pulseless" hand is a known entity where collateral circulation maintains viability despite brachial artery spasm or kinking; this usually resolves with reduction. A "white, pulseless" hand is a surgical emergency demanding immediate reduction and potentially vascular exploration if perfusion does not return. Always maintain a high index of suspicion for compartment syndrome (pain out of proportion, tense compartments, pain on passive extension of the fingers).
Radiographic Evaluation and Templating
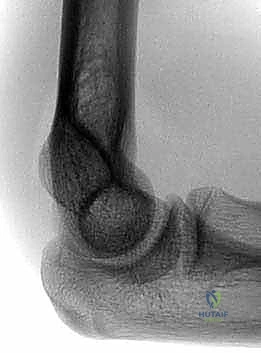
High-quality orthogonal imaging is mandatory. Review true Anteroposterior (AP) and lateral radiographs of the elbow. If the child is in severe pain, a "trauma AP" (beam perpendicular to the humerus) and a "trauma lateral" (beam perpendicular to the forearm) are acceptable initial substitutes.
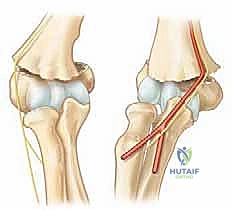
On the lateral view, meticulously trace the Anterior Humeral Line (AHL). Drawn along the anterior cortex of the humeral shaft, this line must intersect the middle third of the capitellum in a normal pediatric elbow. If the capitellum sits entirely posterior to this line, it confirms an extension-type displacement (Gartland II or III). Additionally, look for the posterior fat-pad sign, which represents joint effusion displacing the extracapsular fat; its presence is highly sensitive for an occult intra-articular fracture, even if a fracture line is not overtly visible.
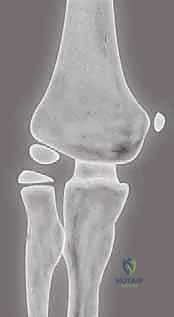
On the AP radiograph, assess the Baumann angle. This is the angle formed by the intersection of a line drawn down the longitudinal axis of the humeral shaft and a line drawn along the physeal line of the lateral condyle. A normal Baumann angle is typically between 70 and 78 degrees. It is a reliable surrogate for the carrying angle of the elbow. A difference of more than 5 to 10 degrees compared to the contralateral, uninjured side indicates coronal plane malalignment (varus or valgus) that must be corrected during surgery.
Operating Room Setup and Patient Positioning
The timing of surgery has evolved. While historic protocols demanded immediate, middle-of-the-night surgery for all supracondylar fractures, modern literature supports delaying surgery until daylight hours for isolated, well-perfused fractures without neurological deficit, provided the child is appropriately splinted and fasted. However, a dysvascular limb, compartment syndrome, or an irreducible fracture with skin tenting remain absolute surgical emergencies.
The patient is positioned supine on the operating table. The fractured extremity is carefully supported and positioned on a radiolucent hand table or armboard. The arm must be placed far enough laterally to allow the C-arm to easily capture both AP and lateral views without obstruction from the table pedestal. For infants and toddlers, the child's entire torso may need to be shifted to the edge of the bed to facilitate this.
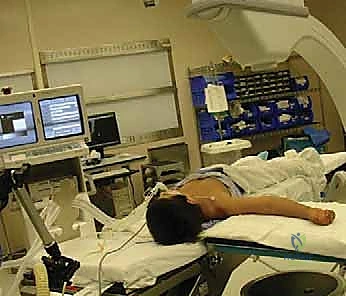
The fluoroscopy C-arm is brought in either parallel to the bed or from the foot of the bed, depending on surgeon preference, but it must be highly maneuverable. The monitor is positioned directly across from the surgeon to prevent neck strain and ensure continuous visual feedback.
* Crucial Technique Note: For highly unstable Gartland Type III or IV fractures, manipulating the arm to obtain orthogonal views will inevitably cause loss of the reduction. In these cases, the surgeon must hold the arm perfectly still in the reduced position, and the radiology technician must rotate the C-arm 90 degrees around the arm to obtain the AP and lateral views.
Step-by-Step Surgical Approach and Fixation Technique
The execution of Closed Reduction and Percutaneous Pinning is a choreographed sequence of precise maneuvers. It requires an understanding of the fracture's personality and the methodical reversal of the deforming forces.
Closed Reduction Maneuvers
Reduction is performed under general anesthesia to ensure complete muscle relaxation. The reduction sequence must follow a strict, logical order: unlock the fragments, correct coronal alignment, correct rotational alignment, and finally, correct sagittal alignment.
- Traction and Unlocking: The surgeon grasps the patient's forearm and applies steady, sustained longitudinal traction with the elbow in slight flexion (20-30 degrees). An assistant provides counter-traction at the axilla. If the proximal spike is buttonholed through the brachialis (pucker sign), simple traction will fail. The surgeon must perform a "milking" maneuver, using the thumb to physically push the proximal fragment posteriorly while simultaneously pulling the distal fragment anteriorly to disengage the bone from the muscle.
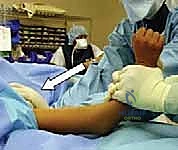
- Coronal Plane Correction: While maintaining traction, the surgeon corrects any medial or lateral translation and varus or valgus angulation. This is done by applying direct, translating pressure to the epicondyles. Varus/valgus alignment is the most critical to restore to prevent long-term cosmetic deformity.
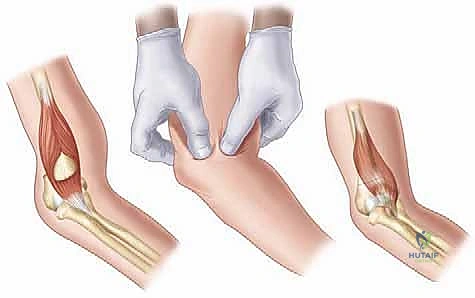
- Sagittal Plane Correction and Hinge Utilization: Once length and coronal alignment are restored, the surgeon places their thumb firmly on the posterior aspect of the olecranon. The fingers wrap around the anterior aspect of the proximal humerus. The surgeon then smoothly hyperflexes the elbow while pushing the olecranon (and thus the distal fragment) anteriorly.
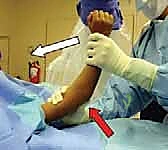
- If the fracture was posteromedially displaced, the forearm is hyperflexed in pronation to tighten the intact medial periosteal hinge.
- If the fracture was posterolaterally displaced, the forearm is hyperflexed in supination to tighten the intact lateral periosteal hinge.
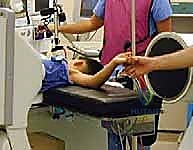
Once hyperflexed, the intact periosteal hinge usually locks the fracture into a stable reduction. The surgeon holds the forearm tightly against the upper arm to maintain this position.
Fluoroscopic Verification and Stability Testing
Before passing any wires, the reduction must be rigorously verified on fluoroscopy.
* Lateral View: With the elbow hyperflexed, obtaining a standard lateral is straightforward. The C-arm is positioned parallel to the floor. Confirm that the Anterior Humeral Line bisects the capitellum and that the posterior cortex is flush.

* AP View (Jones View): Because the elbow is hyperflexed, a standard AP view is impossible. Instead, the C-arm beam is angled to shoot directly through the flexed forearm, perpendicular to the distal humerus. This is the Jones view. Here, you must meticulously check the Baumann angle and ensure the medial and lateral columns are anatomically aligned without step-offs.
Percutaneous Pinning Strategies
Once anatomic reduction is confirmed, fixation is achieved using smooth Kirschner wires (K-wires), typically 1.6 mm or 2.0 mm depending on the child's size. The goal is to maximize biomechanical stability while absolutely minimizing iatrogenic nerve injury.
Lateral Entry Pinning: The safest and most widely accepted configuration is the use of two or three divergent lateral pins.
1. The first pin is placed at the center of the lateral epicondyle. The surgeon uses the thumb to palpate the epicondyle and walks the wire onto the bone to avoid slipping.
2. The wire is driven across the fracture site, aiming medially and proximally to engage the medial cortex of the humeral shaft.
3. A second pin is placed parallel or slightly divergent to the first, starting slightly more anterior or distal on the lateral condyle. To achieve maximal rotational stability, the pins should be maximally separated at the fracture site.
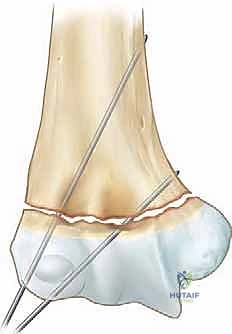
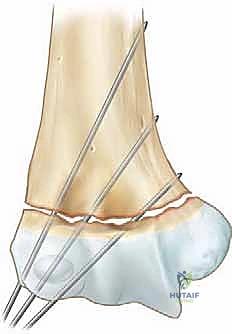
4. If the fracture remains rotationally unstable, a third lateral pin can be added. Biomechanical studies have shown that three widely spaced lateral pins provide stability equivalent to crossed medial-lateral pins.
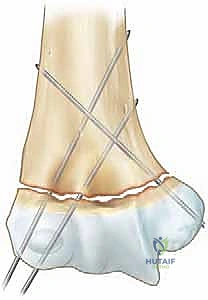
Crossed Pinning (Medial and Lateral): If acceptable stability cannot be achieved with lateral pins alone (often in very high or highly comminuted fractures), a medial pin may be required. However, this carries a significant risk of iatrogenic ulnar nerve injury.
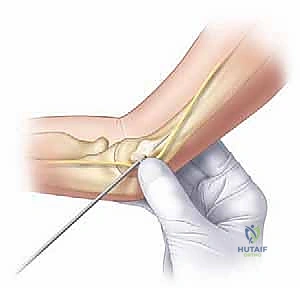
1. If a medial pin is deemed necessary, the elbow must be extended to approximately 45-60 degrees. Hyperflexion subluxates the ulnar nerve anteriorly, placing it directly in the path of the wire.
2. A "mini-open" technique is mandatory. A small incision is made over the medial epicondyle, and blunt dissection is carried down to the bone using a hemostat to physically protect the ulnar nerve before placing the drill guide and wire.
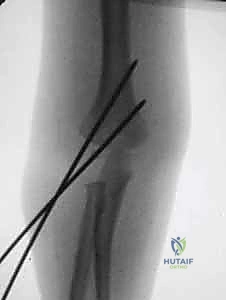
After pin placement, the elbow is extended, and final AP and lateral fluoroscopic images are taken to confirm that the reduction has not been lost during pinning and that the pins have bicortical purchase without entering the olecranon fossa.
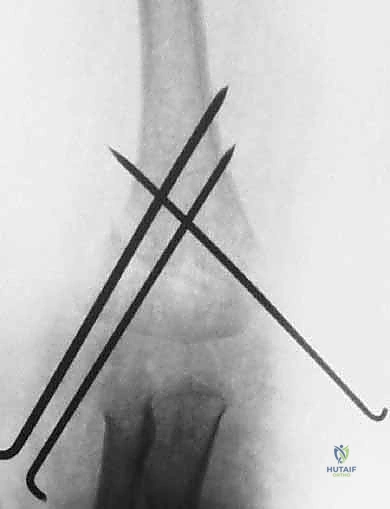
The pins are then bent outside the skin, cut, and capped to prevent migration and facilitate easy removal in the clinic.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, supracondylar humerus fractures are associated with a distinct set of complications. Rapid recognition and appropriate management are essential to prevent permanent disability.
| Complication | Incidence Rate | Etiology / Risk Factors | Salvage & Management Strategy |
|---|---|---|---|
| Neurologic Injury (Traumatic) | 10 - 20% | AIN most common in extension injuries. Radial nerve in posteromedial displacement. | Usually neuropraxia. Observation is key; >90% resolve spontaneously within 3-6 months. |
| Iatrogenic Ulnar Nerve Palsy | 2 - 4% (with medial pin) | Blind medial pinning, pinning in hyperflexion, failure to use mini-open technique. | If noted post-op, immediate removal of the medial pin is mandatory. |
| Vascular Compromise | 1 - 3% | Brachial artery entrapment, kinking, or spasm. | "Pink, pulseless" -> observe. "White, pulseless" post-reduction -> emergent |
