Cervical Laminoplasty: A Masterclass in Posterior Spinal Decompression
Key Takeaway
Welcome, fellows, to a comprehensive masterclass on Cervical Laminoplasty. We'll meticulously dissect the techniques for posterior spinal cord decompression, addressing multilevel cervical myelopathy. From precise patient positioning and meticulous subperiosteal dissection to the nuanced creation of open-door and French-door troughs, we'll cover every critical step. Learn to preserve motion, manage complications, and achieve optimal outcomes for your patients.
Comprehensive Introduction and Patho-Epidemiology
Cervical laminoplasty represents a paradigm shift in the surgical management of multilevel cervical myelopathy, evolving from its conceptual origins in Japan to a globally recognized, motion-preserving standard of care. Originally pioneered in the 1970s to address the high prevalence of ossification of the posterior longitudinal ligament (OPLL) in Asian populations, this technique was ingeniously designed to expand the spinal canal without the inherent morbidities associated with traditional laminectomy. By artfully reconstructing the posterior neural arch rather than ablating it, laminoplasty achieves indirect decompression of the spinal cord through dorsal migration, meticulously avoiding the post-laminectomy kyphosis and dense epidural fibrosis (post-laminectomy membrane) that historically plagued posterior cervical surgery. The procedure serves as a masterclass in balancing aggressive neural decompression with the preservation of spinal biomechanics.
To master this procedure, the operating surgeon must first possess a profound understanding of the underlying pathogenesis dictating the need for intervention. The vast majority of patients present with cervical spondylotic myelopathy (CSM), an insidious, progressive condition driven by a degenerative cascade. The inciting event is typically intervertebral disc desiccation and proteoglycan depletion, leading to a loss of hydrostatic pressure and subsequent disc space narrowing. This mechanical failure transfers aberrant loads to the uncovertebral joints (joints of Luschka) and the posterior zygapophyseal (facet) joints, stimulating osteophytic hypertrophy as a physiological, albeit pathological, stabilizing response.
Concurrently, the loss of disc height results in radial bulging of the annulus fibrosus and infolding or buckling of the ligamentum flavum. This combination of ventral osteochondral bars and dorsal ligamentous hypertrophy creates a circumferential stricture around the spinal cord, transforming the normally capacious cervical canal into a rigid, stenotic cylinder. This mechanical compression is not merely a static phenomenon; dynamic compression occurs during cervical flexion and extension, repeatedly micro-traumatizing the spinal cord. The resulting "pincer effect" between the anterior osteophytes and the posterior ligamentum flavum leads to profound neurological compromise.
The pathophysiology of the resultant myelopathy extends far beyond simple mechanical deformation. Chronic compression induces a devastating ischemic cascade within the spinal cord parenchyma. The extrinsic pressure compromises the delicate microvasculature, particularly the terminal penetrating branches of the anterior spinal artery, leading to watershed ischemia in the central gray matter and adjacent white matter tracts. This chronic ischemia triggers a cascade of excitotoxicity, free radical generation, and localized inflammation, ultimately culminating in neuronal and oligodendroglial apoptosis. The clinical manifestations—ranging from subtle loss of manual dexterity and hyperreflexia to profound spastic paraparesis and sphincter dysfunction—are direct reflections of this irreversible cellular death and demyelination. Laminoplasty aims to arrest this cascade by restoring the physiological cerebrospinal fluid (CSF) buffer around the cord, facilitating reperfusion and preventing further mechanical trauma.
Detailed Surgical Anatomy and Biomechanics
A flawless laminoplasty demands an encyclopedic command of posterior cervical anatomy and an appreciation for the intricate biomechanics governing the subaxial spine. The cervical spine is a highly mobile, lordotic structure comprising seven vertebrae, with the subaxial segment (C3-C7) being the primary domain for laminoplasty. Unlike the highly specialized atlantoaxial complex, which facilitates the majority of cervical rotation, the subaxial vertebrae are designed for a coupled motion of lateral bending and rotation, dictated by the 45-degree coronal inclination of their facet joints. Understanding this biomechanical reality is crucial, as aggressive lateral dissection during surgery can destabilize these joints, leading to iatrogenic deformity.

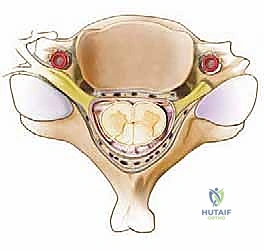
The osseous boundaries of the spinal canal dictate our surgical margins. Anteriorly, the canal is bounded by the vertebral bodies, intervertebral discs, and the posterior longitudinal ligament (PLL). Laterally, the pedicles and the medial aspect of the facet joints form the walls, while the laminae and the ligamentum flavum create the dorsal roof. The transition zone between the lamina and the lateral mass (the "valley") is the critical landmark for our osteotomies. The hinge must be placed precisely at the medial aspect of the lateral mass to ensure a sufficient opening angle without violating the facet capsule or entering the lateral recess prematurely.
Neurovascular preservation is the paramount directive during this procedure. The spinal cord itself is highly vulnerable to contusion, particularly in the stenotic, myelopathic patient where the protective subarachnoid space is obliterated. The exiting nerve roots, particularly the C5 root, are at risk of traction injury as the cord drifts dorsally following decompression. Furthermore, the vertebral arteries, ascending through the transverse foramina (V2 segment) from C6 to C1, lie perilously close to the lateral masses. While a standard laminoplasty approach remains medial to the vertebral artery, any aberrant lateral plunge with a high-speed burr or excessive retraction can result in catastrophic, life-threatening hemorrhage.
The muscular anatomy of the posterior cervical spine is equally critical, particularly regarding the preservation of sagittal alignment. The posterior tension band is primarily composed of the semispinalis cervicis, multifidus, and splenius capitis muscles. The attachment of the semispinalis cervicis to the massive spinous process of C2 is the keystone of cervical lordosis. Detachment or denervation of this muscle complex inevitably leads to progressive kyphosis, effectively negating the benefits of the laminoplasty by tethering the spinal cord over the anterior compressive lesions. Meticulous, tissue-sparing dissection utilizing avascular internervous planes is mandatory to preserve this dynamic muscular envelope.
Exhaustive Indications and Contraindications
Patient selection is the ultimate determinant of success in cervical laminoplasty. The procedure is not a panacea for all cervical pathology; rather, it is a highly specific tool designed for a distinct subset of patients. The ideal candidate presents with multilevel (typically three or more levels) cervical spondylotic myelopathy or OPLL, with preserved cervical lordosis and an absence of gross instability. In these patients, laminoplasty offers a motion-preserving alternative to multilevel anterior cervical discectomy and fusion (ACDF) or posterior laminectomy and fusion, significantly reducing the risk of adjacent segment disease and avoiding the pseudarthrosis complications inherent to long-segment fusions.
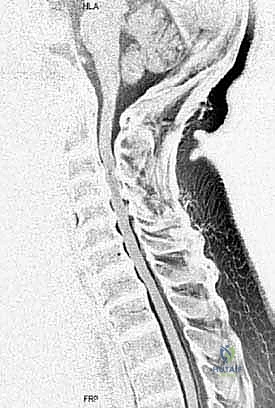
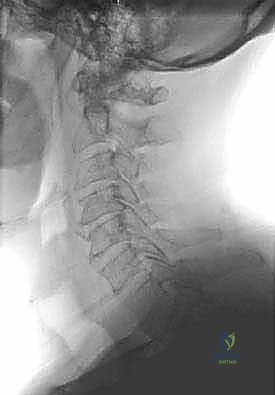
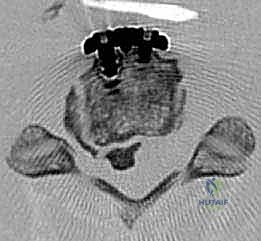

Conversely, recognizing the absolute and relative contraindications is vital to prevent catastrophic outcomes. The most critical absolute contraindication is a fixed kyphotic deformity. Laminoplasty relies on the "bowstring effect"—the dorsal migration of the spinal cord away from anterior pathology. In a kyphotic spine, the cord remains tethered anteriorly regardless of the posterior space created, rendering the decompression entirely ineffective. A general consensus suggests that a kyphosis exceeding 10 to 14 degrees precludes the use of isolated laminoplasty. Furthermore, the "K-line" concept is crucial in OPLL; if the anterior compressive mass crosses the K-line (a line connecting the midpoints of the spinal canal at C2 and C7), the cord will not drift posteriorly enough, necessitating an anterior approach or a combined anterior-posterior procedure.
Significant segmental instability is another absolute contraindication. Because laminoplasty does not fuse the spine, any preexisting hypermobility or translation (e.g., >3.5 mm of translation or >11 degrees of angular change on dynamic radiographs) will persist or worsen postoperatively, potentially exacerbating the myelopathy. Relative contraindications include severe, predominant axial neck pain, as laminoplasty does not address the arthritic facet joints or degenerative discs that typically generate this pain. In fact, posterior cervical surgery can occasionally exacerbate axial neck pain due to muscular disruption.
| Parameter | Indications for Laminoplasty | Contraindications for Laminoplasty |
|---|---|---|
| Pathology | Multilevel CSM (≥ 3 levels), OPLL, Congenital stenosis, Dorsal epidural tumors | Single or two-level radiculopathy, Isolated anterior focal compression |
| Sagittal Alignment | Lordotic or neutral alignment (Kyphosis < 10 degrees) | Fixed kyphotic deformity (> 10-14 degrees) |
| Stability | Stable subaxial spine | Segmental instability (>3.5mm translation, >11° angulation) |
| Symptomatology | Progressive myelopathy, Myeloradiculopathy | Severe, predominant axial neck pain without myelopathy |
| Anatomy | K-line positive (OPLL) | K-line negative (OPLL crossing the K-line) |
Pre-Operative Planning, Templating, and Patient Positioning
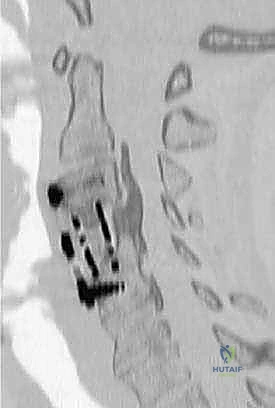
Surgical execution is merely the final act of a process that begins with exhaustive preoperative planning. Advanced neuroimaging is non-negotiable. Magnetic Resonance Imaging (MRI) is the gold standard for evaluating the soft tissues, allowing precise quantification of spinal cord compression, intrinsic cord signal changes (myelomalacia or edema), and the extent of ligamentum flavum hypertrophy. However, MRI often overestimates osseous stenosis and fails to adequately characterize OPLL. Therefore, a high-resolution Computed Tomography (CT) scan is mandatory, particularly in Asian populations or when OPLL is suspected on plain radiographs. CT provides exquisite detail of the bony anatomy, allowing the surgeon to template the precise location of the lateral mass-lamina junction for hinge placement and to identify complex OPLL morphologies that might complicate the decompression.
Patient positioning is a critical phase of the operation, fraught with potential hazards. The myelopathic spinal cord is profoundly sensitive to any hyperextension or hyperflexion during intubation and positioning. Close communication with the anesthesia team is vital. Fiberoptic awake intubation or video laryngoscopy with strict in-line manual stabilization should be strongly considered for patients with severe stenosis. Baseline intraoperative neuromonitoring (IONM)—including Somatosensory Evoked Potentials (SSEPs) and Motor Evoked Potentials (MEPs)—must be obtained prior to positioning to establish a reliable baseline and detect any positioning-induced neural compromise. Furthermore, mean arterial pressure (MAP) must be strictly maintained above 80-85 mmHg throughout the procedure to ensure adequate spinal cord perfusion, particularly as the cord is decompressed and reperfusion injury becomes a risk.

Once intubated, the patient is secured in a Mayfield rigid cranial fixator. This device is indispensable, providing absolute control over head positioning and preventing inadvertent ocular or facial pressure necrosis. The patient is then carefully log-rolled onto a Jackson spinal table or similar specialized frame equipped with chest and pelvic bolsters. The abdomen must hang completely free to minimize intra-abdominal pressure, which directly correlates with epidural venous engorgement and excessive intraoperative hemorrhage.
The neck is positioned in a neutral to slightly flexed posture (the "military tuck"). Hyperextension must be rigorously avoided, as it buckles the ligamentum flavum and acutely worsens canal stenosis. Reverse Trendelenburg positioning (approximately 15 degrees) is employed to facilitate venous drainage from the head and neck, further minimizing epidural bleeding. Finally, the shoulders are gently taped down to allow radiographic visualization of the lower cervical spine, taking great care to avoid excessive traction that could precipitate a brachial plexus palsy.
Step-by-Step Surgical Approach and Fixation Technique
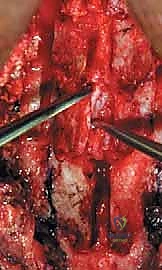
The surgical approach requires meticulous, bloodless dissection to preserve the posterior tension band. A standard midline longitudinal incision is made, typically spanning from the spinous process of C2 to C7 or T1, depending on the planned levels of decompression. The dissection proceeds through the avascular ligamentum nuchae. It is imperative to identify and strictly adhere to this midline raphe to minimize muscle bleeding and denervation. Subperiosteal dissection of the paraspinal musculature is carried out laterally off the spinous processes and laminae.
A critical technical nuance is the preservation of the muscular attachments to C2 (semispinalis cervicis) and C7. If C3 requires decompression, the lamina can often be undercut without detaching the C2 musculature. The lateral dissection must stop precisely at the midpoint of the lateral masses. Exposing the lateral aspect of the lateral mass or violating the facet joint capsule invites postoperative instability and severe axial neck pain. Self-retaining retractors are placed carefully, ensuring the blades do not crush the muscle against the lateral masses.

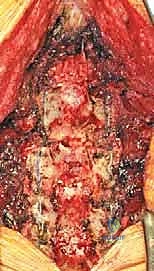
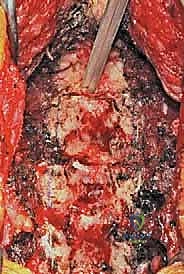
The core of the laminoplasty is the creation of the bilateral bony troughs. In the classic Hirabayashi "open-door" technique, one side acts as a hinge, while the contralateral side is completely osteotomized. Using a high-speed matchstick or acorn burr (typically 3mm), a longitudinal trough is drilled at the junction of the lamina and the lateral mass. On the planned "hinge" side (often the side with less severe radicular symptoms), the dorsal cortex and cancellous bone are removed, leaving the ventral cortex intact but thinned enough to allow a greenstick fracture. On the "open" side, the trough is carried completely through the ventral cortex.
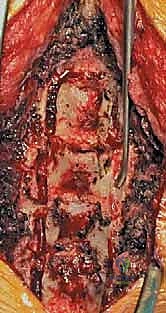
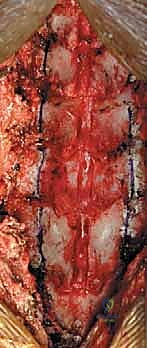
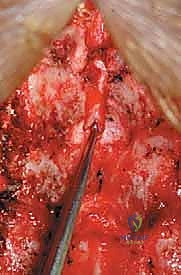
Once the troughs are prepared, the ligamentum flavum at the cranial and caudal extents of the construct (e.g., C2-C3 and C6-C7 interlaminar spaces) is carefully resected using Kerrison rongeurs to mobilize the entire laminar arch. The spinous processes are then gently pushed toward the hinge side. The surgeon will feel a tactile "give" as the ventral cortex of the hinge side undergoes a greenstick fracture, allowing the "door" to swing open. This maneuver must be performed slowly and deliberately to avoid a complete fracture of the hinge, which would effectively convert the procedure into a laminectomy. As the door opens, epidural veins may bleed profusely; these are managed with bipolar electrocautery, hemostatic matrices (e.g., Floseal, Surgiflo), and gentle cottonoid tamponade.
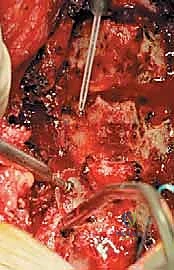
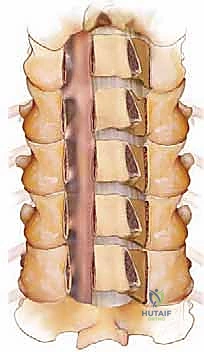
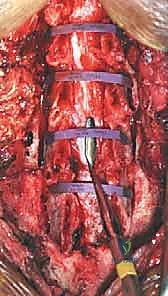
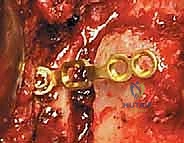
Rigid fixation of the open door is mandatory to prevent postoperative closure ("spring-back"), which would result in catastrophic re-compression of the spinal cord. Historically, this was achieved with complex suture techniques, but modern practice relies heavily on titanium mini-plates. These specialized laminoplasty plates are secured with small, self-drilling screws to the lateral mass on one side and the elevated lamina on the other. The plates maintain the expanded canal volume precisely and securely. Bone allograft or autograft can be placed in the hinge side to promote structural healing, though the rigid plating often provides sufficient stability. The wound is closed in meticulous layers over a subfascial drain to prevent epidural hematoma formation.
Complications, Incidence Rates, and Salvage Management
While laminoplasty is generally safer than multilevel laminectomy and fusion, it is not without significant risks. The surgeon must be acutely aware of potential complications, their pathomechanisms, and appropriate salvage strategies. The most notorious complication specific to this procedure is post-operative C5 palsy. Occurring in roughly 5% to 15% of cases, this presents as a unilateral or bilateral deltoid and biceps weakness, typically manifesting 24 to 72 hours postoperatively.
The etiology of C5 palsy is multifactorial and heavily debated. The dominant theory involves the "tethering effect": as the spinal cord drifts dorsally into the newly expanded canal, the relatively short and horizontally oriented C5 nerve roots are placed under immense traction against the unyielding pedicles and lateral masses. An alternative theory posits a localized reperfusion injury to the anterior horn cells following sudden decompression. Management is overwhelmingly conservative, relying on physical therapy and time; the vast majority of patients recover full or near-full function within 6 to 12 months. Prophylactic bilateral C4-C5 foraminotomies at the time of laminoplasty have been advocated by some to reduce this risk, though this remains controversial due to the increased risk of bleeding and instability.
Another prevalent complication is severe postoperative axial neck pain, reported in up to 60% of patients. This is primarily attributed to the extensive stripping and ischemic necrosis of the paraspinal extensor musculature, as well as potential violation of the facet joint capsules. Meticulous surgical technique, sparing the C2 muscle attachments, and utilizing muscle-splitting approaches can mitigate this risk. If pain is refractory to conservative measures (NSAIDs, physical therapy, muscle relaxants), facet injections or ultimately, posterior cervical fusion may be required as a salvage procedure.
Intraoperative complications such as hinge fracture and dural tears require immediate, decisive action. A complete fracture of the hinge side destabilizes the lamina, risking migration into the canal. If this occurs, the floating lamina must be secured with bilateral plates, or the procedure must be converted to a laminectomy and instrumented fusion. Dural tears, particularly common when operating on OPLL with dural ossification, must be repaired primarily if possible, augmented with synthetic dural substitutes and tissue sealants, and managed with a lumbar drain if a high-flow CSF leak is present.
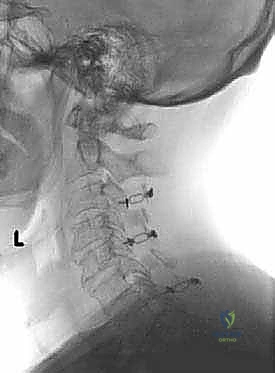
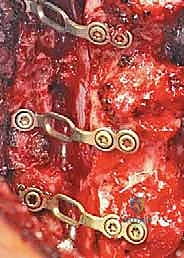
| Complication | Estimated Incidence | Primary Pathomechanism | Prevention / Salvage Management |
|---|---|---|---|
| C5 Nerve Root Palsy | 5% - 15% | Cord drift causing root tethering; Reperfusion injury | Prophylactic foraminotomy (controversial); Conservative management (PT, time) |
| Axial Neck Pain | 20% - 60% | Disruption of extensor musculature; Facet capsule violation | Preserve C2 attachments; Limit lateral dissection; Post-op NSAIDs and PT |
| Hinge Fracture | 3% - 10% | Over-drilling the hinge trough; Aggressive door opening | Slow, controlled opening; Salvage with bilateral plating or convert to fusion |
| Post-op Kyphosis | 5% - 10% | Loss of posterior tension band; Pre-existing poor alignment | Strict adherence to alignment contraindications; Convert to anterior/posterior fusion |
| Dural Tear / CSF Leak | 2% - 5% | Adhesions (OPLL); Errant burr usage | Primary repair, dural sealants, subfascial drain, potential lumbar drain |
Phased Post-Operative Rehabilitation Protocols
The success of a cervical laminoplasty is heavily dependent on a structured, phased postoperative rehabilitation protocol. The immediate postoperative phase (0-2 weeks) focuses on wound healing, pain control, and the prevention of medical complications such as deep vein thrombosis and atelectasis. Patients are typically mobilized on postoperative day one. The use of a cervical collar is highly variable among surgeons; however, a rigid or semi-rigid orthosis (e.g., Aspen or Miami J collar) is frequently utilized for the first 2 to 4 weeks to provide comfort, reduce muscular spasm, and protect the delicate hinge osteotomy while early bony healing occurs.
The subacute phase (2-6 weeks) marks the transition from protection to active rehabilitation. The cervical collar is gradually weaned. The primary focus shifts to restoring the function of the posterior cervical musculature, which has suffered unavoidable surgical trauma. Physical therapy is initiated, emphasizing gentle, pain-free isometric neck strengthening exercises. Scapular stabilization and postural training are critical during this phase to prevent the development of a forward head posture, which increases the biomechanical load on the healing cervical spine and can contribute to late kyphotic deformity.
During the intermediate phase (6-12 weeks), the rehabilitation program becomes more aggressive. Isotonic strengthening exercises are introduced, and active range of motion (ROM) is progressively restored. Patients are encouraged to resume normal activities of daily living, though heavy lifting and high-impact activities remain restricted. The physical therapist will closely monitor for any signs of neurological regression or the onset of severe axial neck pain, which may indicate
