Masterclass in Distal Upper Extremity Skin Grafting & Substitutes: An Intraoperative Guide
Key Takeaway
Welcome, fellows, to an immersive masterclass on skin grafting and substitutes for distal upper extremity wounds. We'll meticulously cover patient positioning, comprehensive anatomy, advanced wound bed preparation, precise graft harvesting, and application techniques. Learn critical pearls and pitfalls for both split-thickness and full-thickness grafts, ensuring optimal functional and aesthetic outcomes in complex cases.
Comprehensive Introduction and Patho-Epidemiology
The reconstruction of soft tissue defects in the distal upper extremity represents one of the most intellectually and technically demanding challenges in orthopedic and plastic surgery. The hand and wrist are anatomically unforgiving; they possess a paucity of redundant soft tissue and house a dense concentration of critical neurovascular and musculotendinous structures immediately deep to the dermis. Consequently, the judicious application of skin grafting and modern skin substitutes is not merely an exercise in achieving wound closure, but a fundamental prerequisite for the restoration of complex biomechanical function, the protection of vital gliding planes, and the optimization of aesthetic outcomes. As reconstructive surgeons, we must approach these defects with a profound understanding of tissue physiology and a meticulous surgical technique.
The epidemiology of distal upper extremity soft tissue loss is broad and encompasses high-energy trauma, thermal and chemical burns, oncologic extirpation, and severe necrotizing infections. Regardless of the etiology, the reconstructive elevator dictates that we employ the simplest, most reliable method of coverage that fulfills the functional requirements of the specific anatomic zone. Before advancing to local, regional, or free tissue transfer, the surgeon must exhaust the potential of skin grafting. To do so effectively, a rigorous standardization of our operative vocabulary is essential. We must delineate between the biologic behaviors of various graft sources to predict their clinical trajectory accurately.

An autograft, representing the absolute gold standard in reconstructive surgery, is harvested from the patient’s own body, ensuring perfect histocompatibility and the highest probability of permanent integration. Conversely, an isograft (harvested from a monozygotic twin) is genetically identical but rarely encountered in standard practice. Allografts, typically derived from cadaveric human skin, and xenografts, most frequently of porcine origin, provide critical but strictly temporary biologic coverage. Due to inevitable immunologic rejection, these grafts will eventually slough or require excision; however, they remain invaluable in our armamentarium for managing massive burn injuries, temporizing critically ill patients, or preparing a hostile wound bed for definitive closure.
The architectural classification of autografts dictates their clinical utility. Split-thickness skin grafts (STSGs) harvest the entire epidermis and a variable portion of the underlying dermis. Their relatively low metabolic demand allows them to survive on less robustly vascularized wound beds, making them highly reliable for large surface area coverage. However, their reduced dermal component predisposes them to significant secondary contraction, diminished durability, and poor sensory recovery. Full-thickness skin grafts (FTSGs) incorporate the entire epidermis and dermis, offering superior elasticity, resistance to shear forces, and a vastly improved cosmetic and functional match, particularly over joints and glabrous surfaces. The inherent trade-off is their absolute requirement for a pristine, highly vascularized recipient bed and the necessity for primary closure of the donor site.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of surgical anatomy is the cornerstone of safe and effective graft harvesting. The surgeon must navigate the delicate balance between securing an adequate volume of high-quality tissue and minimizing iatrogenic donor site morbidity. The selection of a donor site is never arbitrary; it is dictated by the specific requirements of the recipient bed, the desired thickness of the graft, and the patient's individual anatomic variations.
Split-Thickness Skin Graft Donor Sites
The anterior and anterolateral thigh represents the workhorse donor site for STSGs due to its expansive, relatively flat surface area and the presence of thick, high-quality skin. When harvesting from this region, the surgeon must maintain a strict superficial plane to avoid injury to the lateral femoral cutaneous nerve. This nerve typically emerges beneath the inguinal ligament, medial to the anterior superior iliac spine, and courses superficially over the sartorius muscle before arborizing across the anterolateral thigh. Deep passes with the dermatome or aggressive underlying tissue manipulation can result in meralgia paresthetica, a deeply distressing neuropathic complication for the patient. The harvest must remain strictly superficial to the fascia lata, preserving the underlying vastus lateralis and rectus femoris musculature.
The scalp is an exceptionally robust, yet frequently underutilized, donor site for STSGs. Its unique anatomic layered structure—comprising Skin, dense Connective tissue, the Galea Aponeurotica, Loose areolar tissue, and Pericranium (SCALP)—provides a highly vascularized dermal bed with an immense density of epidermal appendages (hair follicles and sebaceous glands). This rich appendageal network drives astonishingly rapid re-epithelialization of the donor site, often within 5 to 7 days. Vascular supply is profusely derived from the external carotid (superficial temporal, posterior auricular, occipital arteries) and internal carotid (supraorbital, supratrochlear arteries) systems. Surgeons must be cognizant of the superficial temporal artery and vein running anterior to the auricle, ensuring harvest parameters avoid these major conduits. A critical technical pearl involves the subgaleal injection of an epinephrine-containing tumescent solution; this hydro-dissection minimizes hemorrhagic oozing and creates a firm, convex surface that facilitates smooth dermatome passes.
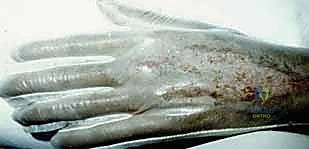
The dorsal and volar aspects of the proximal forearm offer a convenient STSG donor site, particularly when operating exclusively on the upper extremity under a regional block. The skin here is thinner than that of the thigh, making it suitable for delicate recipient sites. However, the surgeon must meticulously avoid the superficial venous network (cephalic, basilic, and median cubital veins) and the medial and lateral antebrachial cutaneous nerves. Furthermore, the cosmetic implications of a forearm donor site must be carefully discussed with the patient, as the resulting scar is highly visible and may be aesthetically displeasing compared to the easily concealed thigh.
Full-Thickness Skin Graft Donor Sites
When reconstructing the specialized glabrous (hairless) skin of the palmar hand and volar digits, matching the unique microanatomy is paramount. The sole of the foot, specifically the non-weight-bearing arch starting at the junction of glabrous and non-glabrous skin, yields an FTSG that perfectly mimics palmar tissue in thickness, durability, and sensory potential. However, harvesting from the plantar arch carries the risk of injuring the medial plantar nerve and artery. Furthermore, the resulting donor defect often cannot be closed primarily and may paradoxically require an STSG for closure, creating a secondary site of morbidity. Alternatively, the hypothenar eminence along the ulnar border of the hand can provide a small, excellent color- and texture-matched FTSG, though the available surface area is highly restricted.

For larger FTSG requirements where glabrous match is not critical (e.g., dorsal hand defects), redundant skin folds are targeted to allow for primary closure. The lower abdomen, extending in an elliptical fashion from the anterior superior iliac spines, provides an abundant source of FTSG, particularly in multiparous or massive weight-loss patients. While harvesting, the surgeon must be aware of the superficial epigastric vessels. The upper inner arm, located at the medial bicipital groove, offers thin, pliable, and generally hairless skin ideal for dorsal digital reconstruction. Care must be taken to avoid the medial brachial cutaneous nerve and the basilic vein during harvest, and the ellipse must be oriented longitudinally to allow tension-free primary closure without restricting shoulder abduction.
Recipient Site Considerations in the Distal Upper Extremity
The distal upper extremity demands a recipient site capable of supporting not just skin, but the dynamic excursion of underlying tendons and joints. The survival of any graft depends entirely on a process called plasmatic imbibition (the initial absorption of nutrients from the wound bed) followed by inosculation and true angiogenesis. This physiological cascade absolutely requires a vascularized substrate. Healthy muscle, fat, paratenon, and periosteum possess the requisite capillary density to support a graft.
Conversely, denuded cortical bone lacking periosteum, exposed articular cartilage, and bare tendon devoid of paratenon are strictly avascular. Placing a skin graft directly onto these structures is biologically futile and will universally result in graft necrosis. In the hand, where the preservation of tendon gliding is paramount, grafting directly over a joint or a flexor tendon without an intervening layer of vascularized gliding tissue (such as a dermal substitute or a local fascial flap) will lead to severe, debilitating contractures and loss of digital excursion.
Exhaustive Indications and Contraindications
The decision to utilize a skin graft, and the subsequent selection between a split-thickness and full-thickness graft, requires a nuanced understanding of the patient's functional demands, the anatomic location of the defect, and the physiological status of the wound bed.
Graft Type Selection Dynamics
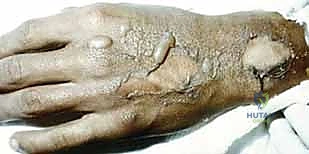
Split-thickness skin grafts are indicated for the coverage of large, broad, and relatively flat surface areas where the primary goal is rapid, reliable wound closure. In the upper extremity, STSGs are frequently employed for extensive dorsal forearm defects, massive burn excisions, and fasciotomy wound closures. Because they require less revascularization energy, STSGs demonstrate a higher "take" rate on sub-optimal (yet still vascularized) beds compared to FTSGs. However, the surgeon must anticipate the profound secondary contraction inherent to STSGs. As the wound heals, the myofibroblasts within the limited dermal component contract aggressively. If an STSG is placed across a joint line (e.g., the dorsal metacarpophalangeal joints or the volar wrist crease), this secondary contraction will inevitably lead to severe functional contractures.
Full-thickness skin grafts are the reconstructive gold standard for defects involving the specialized glabrous skin of the palm and volar digits, and for any defect crossing a joint crease. Because FTSGs contain the entire dermal architecture, they exhibit minimal secondary contraction, preserving joint mobility. Furthermore, they provide superior resistance to the immense shear forces experienced by the palmar hand during grip and prehension. FTSGs also offer a significantly better cosmetic match in terms of color, texture, and contour, and they possess a higher capacity for sensory reinnervation—a critical factor for tactile function in the digits.
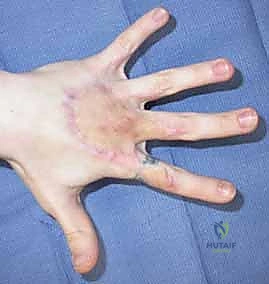
Contraindications to Grafting
The absolute contraindications to skin grafting primarily revolve around the biological inadequacy of the recipient bed. As previously stated, grafting onto avascular structures (bare bone, bare tendon, bare cartilage, or synthetic implants) is contraindicated. Furthermore, grafting onto a wound bed with active, uncontrolled clinical infection or a bacterial bioburden exceeding $10^5$ colony-forming units per gram of tissue will almost certainly result in graft lysis due to bacterial proteases and purulent exudate separating the graft from the bed.
| Parameter | Split-Thickness Skin Graft (STSG) | Full-Thickness Skin Graft (FTSG) |
|---|---|---|
| Primary Indications | Large surface area defects, fasciotomy closures, burns, suboptimal (but vascular) wound beds. | Small to medium defects, glabrous skin (palm/volar digits), over joint creases, cosmetically sensitive areas. |
| Contraindications | Placement over joint creases (due to contracture risk), areas requiring high shear resistance (palms), avascular beds. | Large defects precluding primary donor closure, avascular beds, heavily contaminated/exudative wounds. |
| Secondary Contraction | High (Significant risk of joint contracture). | Low (Maintains original dimensions well). |
| Primary Contraction | Low (Expands slightly upon harvest). | High (Shrinks immediately upon harvest). |
| Durability & Shear | Poor to Moderate. | Excellent. |
| Sensory Recovery | Poor and unpredictable. | Good, approaches normal tactile sensation over time. |
| Donor Site Morbidity | Painful, heals by secondary intention (re-epithelialization), potential for hypertrophic scarring. | Less painful, requires primary surgical closure, leaves a linear scar. |
Pre-Operative Planning, Templating, and Patient Positioning
Meticulous preoperative planning is the invisible framework upon which surgical success is built. The reconstructive surgeon must anticipate the spatial requirements of the operative field, ensuring simultaneous, unimpeded access to both the recipient wound bed and the selected donor site.
Wound Bed Assessment and Preparation
The preparation of the recipient bed is arguably the most critical determinant of graft survival. The surgeon must transition a contaminated, necrotic, or chronic wound into a clean, highly vascularized, and biologically active bed. This begins with aggressive, meticulous sharp débridement. Using a combination of scalpel blades (typically #15 or #10), fine Metzenbaum scissors, and tissue forceps, the surgeon systematically excises all nonviable tissue, slough, eschar, and fibrotic debris until healthy, punctate bleeding is encountered across the entire surface. The surgical aphorism holds true: never graft onto necrotic tissue.
Following sharp débridement, the bacterial bioburden must be aggressively managed. High-volume pulse lavage using sterile saline serves to mechanically disrupt bacterial biofilms and clear microscopic debris. In cases of chronic or heavily contaminated wounds, a period of wound bed maturation is often required before grafting. The application of Negative Pressure Wound Therapy (NPWT), or Vacuum-Assisted Closure (VAC), has revolutionized this phase. By applying continuous sub-atmospheric pressure, the VAC device removes inhibitory wound exudate, reduces interstitial edema, and mechanically stimulates the rapid proliferation of robust, "beefy red" granulation tissue. This granulation tissue provides the optimal hypervascular substrate for subsequent graft inosculation.

Patient Positioning and Operating Room Setup
Positioning must facilitate seamless ergonomic flow for the surgical team. When addressing the distal upper extremity, the patient is typically positioned supine with the affected arm extended onto a radiolucent hand table. A proximal pneumatic tourniquet is routinely applied to provide a bloodless field during the precise débridement phase, though it must be deflated prior to final hemostasis and graft application to ensure the viability of the wound bed is accurately assessed.
If an STSG is to be harvested from the anterior thigh, the ipsilateral leg is prepped and draped freely to allow the surgeon to maneuver the dermatome across the broad expanse of the quadriceps. If an FTSG from the lower abdomen or groin is planned, the patient's torso must be prepped widely, and the table may be slightly flexed to reduce tension during the primary closure of the donor defect. For scalp harvests, the head is shaved in the designated area, and the patient's head is rotated laterally on a gel donut to provide optimal exposure.
Step-by-Step Surgical Approach and Fixation Technique
The intraoperative execution of skin grafting demands a synthesis of delicate tissue handling, precise instrumentation, and rigid fixation. The methodology diverges significantly depending on whether an STSG or an FTSG is being utilized.
Split-Thickness Skin Graft Harvest and Application
The harvest of an STSG is most commonly performed using a powered air or electric dermatome (e.g., Zimmer dermatome). The surgeon must meticulously select the desired thickness; for the upper extremity, a medium-thickness graft ranging from 0.012 to 0.015 inches is typically ideal, balancing graft durability with reliable donor site healing. The donor site is generously lubricated with sterile mineral oil to ensure the dermatome glides smoothly without catching or tearing the skin. The surgeon applies firm, downward pressure and advances the dermatome at a steady, continuous speed, maintaining a consistent 45-degree angle of attack. An assistant simultaneously applies counter-traction to the skin using a tongue depressor or edge of a towel to maintain a taut, flat surface.
Once harvested, the STSG may be applied as a solid sheet or meshed. Sheet grafts provide superior cosmetic outcomes and are preferred for visible areas like the dorsal hand. Meshing the graft (typically at a 1.5:1 ratio) involves passing it through a mechanical device that cuts multiple staggered slits. This serves two critical functions: it allows the graft to expand to cover a larger surface area, and, more importantly, it creates fenestrations that permit the egress of underlying hematoma or seroma, which are the leading causes of graft failure. The graft is then meticulously inset into the recipient bed, ensuring absolute contact without tension or redundant folds. Fixation is achieved using surgical staples or fine absorbable sutures (e.g., 4-0 or 5-0 chromic gut).
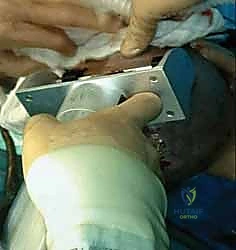
Full-Thickness Skin Graft Harvest and Application
The harvest of an FTSG requires precise templating. The surgeon presses a piece of sterile foil, Esmarch bandage, or specialized template material onto the recipient defect to create an exact structural map. This template is then transferred to the chosen donor site and outlined with a surgical marker. Because FTSGs undergo immediate primary contraction upon harvest due to the high elastin content in the intact dermis, the surgeon must harvest the graft slightly larger (approximately 10-15%) than the template.
The graft is sharply excised using a scalpel, taking care to include the full thickness of the dermis while minimizing the inclusion of underlying subcutaneous adipose tissue. The most critical step follows: defatting the graft. Any residual subcutaneous fat left on the deep surface of the FTSG acts as an impenetrable barrier to revascularization, preventing plasmatic imbibition and guaranteeing graft necrosis. The surgeon drapes the graft over their non-dominant index finger, dermal side up, and meticulously trims away all yellow adipose tissue using sharp curved Iris or tenotomy scissors until the gleaming white undersurface of the dermis is uniformly exposed. The donor site is then widely undermined and closed primarily in layers. The FTSG is inset into the recipient bed and sutured meticulously, often utilizing a tie-over bolster dressing to provide uniform compressive force.
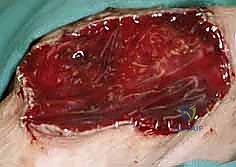
The Role of Skin Substitutes and Biologic Dressings
In modern reconstructive upper extremity surgery, the advent of advanced dermal substitutes has fundamentally altered our treatment algorithms, particularly for wounds with exposed avascular structures. Acellular dermal matrices (e.g., Integra®) and biodegradable temporizing matrices (e.g., NovoSorb® BTM) serve as highly engineered scaffolds. When applied to a complex wound bed (even over bare tendon or bone), these matrices are gradually infiltrated by the patient's own fibroblasts and endothelial cells, generating a robust, neovascularized "neodermis."
This process, which typically takes 3 to 4 weeks, effectively converts a previously avascular, non-graftable wound into a highly vascularized bed capable of supporting an ultra-thin STSG. This two-stage approach allows for the preservation of vital anatomic structures, prevents tendon desiccation, and provides a pliable, gliding soft-tissue envelope that closely mimics native tissue, significantly reducing the need for complex, morbid flap coverage in selected patients.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, skin grafting in the distal upper extremity carries a distinct profile of potential complications. The surgeon must be vigilant in identifying and managing these issues promptly to salvage the reconstruction and optimize functional recovery.
Early Postoperative Complications
The absolute most common cause of early graft failure is the accumulation of fluid—specifically a hematoma or seroma—between the graft and the recipient bed. This fluid collection acts as a physical barrier, instantly halting plasmatic imbibition and preventing the critical ingrowth of capillary buds. Prevention relies on meticulous intraoperative hemostasis, the judicious use of meshed grafts to allow fluid egress, and the application of rigid, compressive tie-over bolster dressings or NPWT over the graft. If a hematoma is identified early (within 24-48 hours), it must be immediately evacuated by rolling a sterile swab over the graft or making a small incision in the graft to express the clot.
Infection is another devastating early complication. Beta-hemolytic Streptococcus can rapidly lyse a skin graft within 24 hours via the production of potent proteolytic enzymes. Pseudomonas aeruginosa, often identified by its characteristic green exudate and sweet odor, is also highly toxic to graft integration. Management requires aggressive systemic antibiotic therapy guided by deep tissue cultures, and often necessitates the complete removal of the necrotic graft, serial débridement, and eventual re-grafting once the bioburden is eradicated. Shear forces, caused by inadequate immobilization of the extremity, can also disrupt the fragile capillary connections forming during inosculation, leading to graft avulsion and failure.

Late Complications and Functional Deficits
Late complications are frequently related to the biomechanical properties of the grafted tissue. Secondary contracture is the most significant long-term issue, particularly with STSGs placed over joints or web spaces. As the graft contracts, it can tether underlying tendons and restrict joint excursion, leading to severe functional impairment of the hand. Management of established contractures often requires surgical release (incisional or excisional), followed by the application of an FTSG, a local flap (e.g., Z-plasty), or a dermal substitute to reconstruct the resulting defect.
Dyspigmentation is nearly universal; STSGs frequently become hyperpigmented or hypopigmented relative to the surrounding native skin, presenting a significant cosmetic concern for the patient. Hypertrophic scarring can occur at both the recipient and donor sites, particularly in patients with darker Fitzpatrick skin types or in areas subjected to high tension. Donor site morbidity, particularly the severe, burning pain associated with STSG harvest sites on the thigh, can often overshadow the pain of the primary injury and requires proactive multimodal pain management and advanced occlusive dressings.
| Complication | Incidence Rate | Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Hematoma / Seroma | 10 - 15% | Inadequate hemostasis, lack of fenestration, insufficient bolster compression. | Early evacuation via rolling/incision; if late/necrotic, requires debridement and re-grafting. |
| Graft Infection | 2 - 5% | High preoperative bioburden (Streptococcus, Pseudomonas), necrotic debris. | Systemic antibiotics, removal of sloughed graft, VAC therapy, delayed re-grafting. |
| Shear / Mechanical Failure | 5 - 10% | Poor splinting, patient non-compliance, placement over highly mobile joints. | Strict immobilization (intrinsic plus splint), revision grafting if completely avulsed. |
| Joint Contracture | 15 - 30% (STSG) | Use of STSG over joint creases, inadequate post-op splinting/rehab. | Aggressive hand therapy, serial splinting; surgical release and FTSG/flap reconstruction if refractory. |
| Hypertrophic Scarring | 10 - 20% | Genetic predisposition, prolonged inflammation, high-tension areas. | Silicone gel sheeting, customized compression garments, intralesional corticosteroid injections. |
Phased Post-Operative Rehabilitation Protocols
The surgical application of a skin graft in the hand is only the first phase of reconstruction; the ultimate functional outcome is heavily dictated by the rigor and precision of the postoperative rehabilitation protocol. The hand therapist is an indispensable partner in this phase.
Acute Phase Immobilization and Protection
Immediately following surgery, the paramount goal is the absolute protection of the graft to allow for uninterrupted inosculation and angiogenesis. The distal upper extremity must be rigidly immobilized in a custom orthosis. For dorsal hand or general wrist grafts, the hand is placed in the "safe position" (intrinsic plus position): the wrist is extended 20-30 degrees, the metacarpophalangeal (MCP) joints are flexed 70-90 degrees, and the interphalangeal (IP) joints are fully extended. This position maintains the collateral ligaments of the MCP and IP joints at their maximum length, preventing devastating joint stiffness while simultaneously minimizing shear forces across the graft.
The initial dressing, whether a tie-over bolster or a VAC device, is typically left entirely undisturbed for 5 to 7 days. Premature dressing changes risk avulsing the delicate, newly formed capillary networks, resulting in iatrogenic graft failure. The extremity must be strictly elevated above the level of the heart to minimize interstitial edema, which can increase the diffusion distance for nutrients and impair graft take.
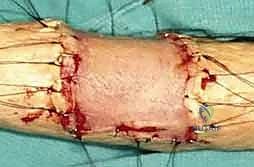
Subacute Phase Mobilization and Scar Management
Upon the first dressing change (typically day 5-7), the graft is inspected for take, hematoma, and infection. If the graft demonstrates robust pink coloration and adherence, the rehabilitation protocol transitions to the subacute phase. The primary objective shifts from absolute protection to the restoration of gliding planes and the prevention of contractures. Gentle, controlled Active Range of Motion (AROM) is initiated. Passive Range of Motion (PROM) is generally avoided in the early subacute phase to prevent excessive tension on the maturing graft.
As the graft fully integrates and the epidermis stabilizes (typically weeks 3-4), aggressive scar management becomes critical. The patient is fitted with custom, continuous compression garments (e.g., Isotoner gloves or custom Jobst garments) to provide counter-pressure, which helps organize collagen deposition and mitigate hypertrophic scarring. Silicone gel sheeting is applied directly over the graft to maintain hydration and further suppress excessive scar formation. Deep friction massage is instituted by the therapist to mobilize the graft over the underlying fascia and tendons, ensuring the restoration of independent tendon excursion and preventing the graft from tethering to deeper structures.
Summary of Landmark Literature and Clinical Guidelines
The modern practice of skin grafting is built upon over a century of meticulous clinical observation and evolving scientific paradigms. A firm grasp of the landmark literature provides the reconstructive surgeon with the necessary context to make
