Radial Nerve Decompression: An Intraoperative Masterclass for Posterior Interosseous Nerve Entrapment
Key Takeaway
Join us in the OR for a masterclass on radial nerve decompression, specifically targeting posterior interosseous nerve (PIN) entrapment. We'll meticulously dissect the anatomy, execute a precise posterior approach, and release critical compression points like the arcade of Frohse and the vascular leash of Henry. Learn invaluable pearls, avoid pitfalls, and ensure optimal patient outcomes through detailed surgical technique and comprehensive postoperative management.
Comprehensive Introduction and Patho-Epidemiology
Radial tunnel syndrome (RTS) and posterior interosseous nerve (PIN) syndrome represent a spectrum of compressive neuropathies affecting the radial nerve in the proximal forearm, presenting a formidable diagnostic and therapeutic challenge for the orthopedic surgeon. Initially described in the seminal literature as "radial pronator syndrome" by Michele and Krueger in 1956, and later refined by Roles and Maudsley, the pathophysiology of this condition hinges on the dynamic and static compression of the radial nerve as it traverses a complex fibro-osseous anatomical corridor. While lateral epicondylitis (tennis elbow) is exponentially more common, RTS frequently mimics, coexists with, or is misdiagnosed as recalcitrant lateral epicondylitis, leading to prolonged patient morbidity and frustration.
The clinical distinction between radial tunnel syndrome and PIN syndrome is paramount, though they share identical anatomical sites of compression. Posterior interosseous nerve syndrome is classically defined by objective motor weakness—specifically, a failure of finger and thumb extension at the metacarpophalangeal joints, and weakness of the extensor carpi ulnaris, resulting in radial deviation during wrist extension. Conversely, radial tunnel syndrome is characterized predominantly, and often exclusively, by pain. This presents a physiological paradox: how does compression of the PIN, historically classified as a pure motor nerve, generate such profound nociceptive symptoms? Contemporary neuroanatomical studies have definitively resolved this by demonstrating that the PIN contains a rich supply of unmyelinated and thinly myelinated afferent sensory fibers. These group IIA and group IV fibers, which innervate the wrist capsule and forearm musculature, are exquisitely sensitive to ischemia and mechanical deformation, mediating the deep, aching, cramp-like pain that is the hallmark of RTS.
The pathogenesis of radial tunnel syndrome is intrinsically linked to the biomechanics of the proximal forearm. The radial nerve is subjected to dynamic, intermittent compression during repetitive pronation and supination, as well as during forceful wrist extension. This repetitive microtrauma induces localized ischemia within the vasa nervorum, leading to intraneural edema, disruption of the blood-nerve barrier, and eventual perineural fibrosis. Unlike carpal or cubital tunnel syndromes, where a single, discrete anatomical structure is usually the culprit, the radial nerve faces a gauntlet of potential compressive structures within a span of merely five centimeters. Understanding the epidemiological profile—typically affecting patients in their fourth to sixth decades with a slight female predilection, often engaged in occupations or sports requiring repetitive forearm rotation—is crucial for maintaining a high index of suspicion.

Detailed Surgical Anatomy and Biomechanics
A masterful surgical decompression demands an encyclopedic understanding of the radial nerve's complex topography as it transitions from the arm into the forearm. The radial nerve pierces the lateral intermuscular septum approximately 10 to 12 centimeters proximal to the lateral epicondyle, transitioning from the posterior to the anterior compartment of the arm. It descends along the lateral border of the brachialis muscle, safely nestled in the interval between the brachialis medially and the brachioradialis (BR) and extensor carpi radialis longus (ECRL) laterally. At a highly variable distance—typically 3 to 5 centimeters distal to the radiocapitellar joint line—the radial nerve bifurcates into its two terminal branches: the predominantly motor posterior interosseous nerve (PIN) and the purely sensory superficial radial nerve (SRN).
The "radial tunnel" is a specialized, dynamic fibro-osseous conduit approximately 5 centimeters in length. Its floor is formed proximally by the anterior capsule of the radiocapitellar joint and distally by the deep head of the supinator muscle. The roof is a composite of muscular and fibrous structures, including the brachioradialis, the extensor carpi radialis brevis (ECRB), and the superficial head of the supinator. Within this confined space, there are six classically described, distinct anatomical sites where the PIN can be tethered or compressed. The most proximal potential site consists of inconstant fibrous bands traversing the interval between the brachialis and the brachioradialis. Moving distally, the second site is the "vascular leash of Henry," a fan-like array of recurrent radial arterial and venous branches that cross superficially over the radial nerve, tethering it during active pronation.
The third site is the tendinous medial border of the ECRB. During forceful wrist extension and forearm pronation, this medial edge becomes taut, acting as a rigid fulcrum against which the radial nerve is compressed. The fourth, less common site involves fibrous bands passing volar to the radiocapitellar joint capsule. The fifth, and unequivocally the most common and critical site of compression, is the Arcade of Frohse. This represents the proximal aponeurotic edge of the superficial head of the supinator muscle. Anatomical studies reveal that this arch is fibrous in approximately 30% of the adult population, and its rigidity tends to increase with age and repetitive mechanical loading. Finally, the sixth site is the distal margin of the supinator muscle, where the PIN exits the tunnel to arborize into the deep extensor compartment; fascial bands here can cause an hourglass constriction of the nerve, a phenomenon well-documented by Sponseller and colleagues.
Key Neurovascular Structures and Intervals
Beyond the primary compressive sites, several critical neurovascular structures must be meticulously managed during the approach. The posterior cutaneous nerve of the forearm, a sensory branch originating from the radial nerve proximal to the elbow, consistently courses superficially across the proximal aspect of the standard surgical incision. Iatrogenic injury to this nerve results in debilitating dysesthesias, numbness over the dorsal forearm, and the potential formation of painful neuromas. Furthermore, the motor branches to the ECRB exhibit significant anatomical variability; they may branch from the main radial nerve trunk proximal to the bifurcation, or directly from the PIN itself. These delicate branches often cross the surgical field transversely and are highly susceptible to traction injury or inadvertent transection during the mobilization of the ECRB or the release of the Arcade of Frohse.
Exhaustive Indications and Contraindications
The decision to proceed with surgical decompression of the radial nerve should never be made precipitously. Because the symptoms of radial tunnel syndrome are predominantly subjective (pain) and electrodiagnostic studies are notoriously unreliable for this specific condition, patient selection is the single most critical determinant of surgical success. A comprehensive, non-operative treatment paradigm must be exhausted before surgical intervention is entertained. This typically involves a minimum of 3 to 6 months of strict activity modification, ergonomic adjustments, non-steroidal anti-inflammatory drugs (NSAIDs), and targeted physical therapy focusing on stretching the supinator and ECRB while avoiding provocative repetitive pronation/supination.
Surgical intervention is indicated when a patient presents with persistent, debilitating lateral forearm pain that has completely failed this exhaustive conservative regimen. The clinical examination must yield specific, reproducible signs that localize the pathology to the radial tunnel rather than the lateral epicondyle. The pathognomonic triad includes: (1) maximal point tenderness located 4 to 5 centimeters distal to the lateral epicondyle over the mobile wad, (2) exacerbation of pain with resisted supination of the extended forearm, and (3) a positive "middle finger test," where resisted extension of the long finger with the elbow extended provokes severe pain in the proximal forearm due to the mechanical tension transmitted through the ECRB to the radial nerve. A diagnostic injection of local anesthetic into the radial tunnel (carefully avoiding the lateral epicondyle) that provides transient, complete relief of symptoms can serve as a powerful confirmatory test and a strong predictor of surgical success.
Indications and Contraindications Table
| Category | Specific Criteria | Clinical Rationale |
|---|---|---|
| Absolute Indications | Progressive PIN Syndrome | Objective motor weakness (loss of digit/thumb extension) mandates urgent decompression to prevent irreversible axonal loss. |
| Absolute Indications | Refractory Radial Tunnel Syndrome | Severe, debilitating pain failing >6 months of conservative care, with positive provocative tests (middle finger test, resisted supination). |
| Relative Indications | Space-Occupying Lesions | MRI-confirmed lipomas, ganglia, or radiocapitellar synovitis causing secondary nerve compression. |
| Absolute Contraindications | Active Local Infection | Cellulitis or deep space infection in the proximal forearm precludes elective nerve decompression. |
| Absolute Contraindications | Cervical Radiculopathy (Primary) | Symptoms entirely driven by C5-C6 root compression without a dual-crush component. |
| Relative Contraindications | Unresolved Worker's Compensation | Psychosocial factors and secondary gain can significantly cloud subjective outcomes in pain-predominant RTS. |
| Relative Contraindications | Concomitant Untreated Epicondylitis | Failure to address severe, co-existing lateral epicondylitis will result in persistent post-operative pain and perceived surgical failure. |
Pre-Operative Planning, Templating, and Patient Positioning
Pre-operative planning for radial nerve decompression relies heavily on a meticulous clinical examination and the strategic exclusion of confounding pathologies. While advanced imaging is not strictly required for the diagnosis of RTS, it plays a vital role in ruling out other etiologies. Standard orthogonal radiographs of the elbow are mandatory to evaluate for radiocapitellar arthritis, occult fractures, or anterior osteophytes that could impinge upon the radial nerve. Cervical spine radiographs should be considered to rule out significant spondylosis, as C5-C6 cervical radiculopathy can present with referred pain to the lateral forearm, mimicking or exacerbating RTS (the "double crush" phenomenon).
Magnetic Resonance Imaging (MRI) of the elbow and proximal forearm is highly recommended in cases of atypical presentation or when a palpable mass is present. MRI can elegantly delineate space-occupying lesions such as ganglion cysts arising from the radiocapitellar joint, lipomas, or severe focal synovitis. Furthermore, high-resolution MRI may occasionally demonstrate denervation edema within the supinator or extensor musculature in advanced cases, though its absence does not preclude the diagnosis. Electrodiagnostic studies (EMG/NCS) are routinely ordered but must be interpreted with caution. While they are highly sensitive for detecting the axonal loss characteristic of PIN syndrome, they are frequently normal in radial tunnel syndrome, where the pathology is driven by dynamic ischemia of sensory fibers rather than frank demyelination of motor axons. A normal EMG should never be used to definitively rule out RTS in the face of a compelling clinical picture.
Patient Positioning and Operating Room Setup
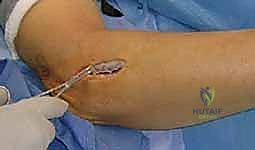
The patient is brought into the operating theater and placed in the supine position. General anesthesia or a regional brachial plexus block can be utilized, though if a block is chosen, it must be noted that intraoperative nerve stimulation will be rendered ineffective. The affected upper extremity is extended onto a radiolucent, well-padded hand table. The arm is abducted to approximately 60 degrees and externally rotated to allow comfortable access to the lateral and posterior aspects of the forearm. The forearm must be free to be fully pronated and supinated throughout the procedure, as dynamic positioning is essential for identifying anatomical intervals and confirming complete decompression.
A sterile pneumatic tourniquet is applied high on the brachium. The use of a tourniquet is absolutely critical; a bloodless surgical field is non-negotiable when performing intricate neurolysis, as even minor venous bleeding can obscure the delicate motor branches of the radial nerve and the surrounding microvascular structures. The arm is exsanguinated with an Esmarch bandage, and the tourniquet is inflated to 250 mmHg (or 100 mmHg above systolic pressure). The entire arm, from the axilla to the fingertips, is prepped and draped in a standard sterile fashion. This extensive draping allows for proximal extension of the incision into the distal arm if the compression extends proximally, or if a concomitant release of the lateral intermuscular septum is deemed necessary.
Step-by-Step Surgical Approach and Fixation Technique
Surgical decompression of the radial nerve can be achieved via an anterior (brachioradialis-ECRL interval) or a posterior (EDC-ECRB interval) approach. The posterior approach is heavily favored by most reconstructive microsurgeons and orthopedic specialists due to its direct, relatively avascular trajectory to the radial tunnel, avoiding the extensive muscle retraction required in the anterior approach.
1. Incision and Superficial Dissection
The procedure commences with the identification of Thompson’s cardinal line, an imaginary line drawn from the lateral epicondyle to Lister’s tubercle at the wrist. A 5 to 7-centimeter longitudinal incision is made centered over the proximal third of this line, beginning approximately 2 centimeters distal to the lateral epicondyle. The incision is carried sharply through the epidermis and dermis. Subcutaneous dissection is then performed meticulously using blunt-tipped Metzenbaum scissors or low-setting electrocautery.
During this superficial dissection, the surgeon must remain hyper-vigilant for the posterior cutaneous nerve of the forearm. This nerve consistently courses from anterior to posterior, crossing the proximal aspect of the operative field just superficial to the deep antebrachial fascia. Once identified, it must be gently mobilized with a vessel loop and retracted out of harm's way. Transection or aggressive traction on this nerve is a primary cause of post-operative dissatisfaction, leading to painful neuromas that are notoriously difficult to treat.

2. Identifying the EDC-ECRB Interval
With the deep antebrachial fascia exposed, the next critical step is identifying the intermuscular interval between the Extensor Digitorum Communis (EDC) and the Extensor Carpi Radialis Brevis (ECRB). This interval can be subtle proximally but becomes distinct distally. The surgeon should begin the fascial incision distally, where the muscle bellies begin to taper into their respective tendons, and carefully propagate the split proximally toward the lateral epicondyle.
To facilitate this, the assistant should fully pronate the forearm. This maneuver rotates the mobile wad anteriorly, opening the posterior interval and bringing the underlying supinator muscle into view. Blunt dissection with a periosteal elevator or the surgeon's finger is used to separate the EDC and ECRB. The fascia overlying the outcropping muscles (abductor pollicis longus and extensor pollicis brevis) is often encountered at the distal extent of this interval and serves as a reliable deep anatomical landmark.
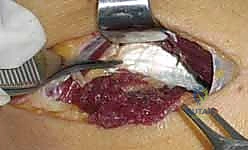
3. Deep Dissection and Nerve Exposure
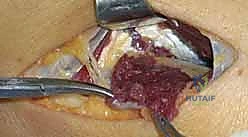
As the EDC and ECRB are retracted (EDC ulnarly, ECRB radially) using self-retaining retractors such as a Weitlaner or Gelpi, the superficial head of the supinator muscle is exposed in the depths of the wound. The muscle fibers of the supinator run obliquely, distal and radial, contrasting with the more longitudinal fibers of the overlying extensors. Carefully palpating the proximal edge of the supinator will reveal the Arcade of Frohse.
Just proximal to the Arcade, embedded in a layer of adipose tissue, the radial nerve and its bifurcation can be identified. Using delicate blunt dissection with a right-angle clamp or tenotomy scissors, the main trunk of the radial nerve, the superficial radial nerve (SRN), and the posterior interosseous nerve (PIN) are isolated. A vessel loop may be loosely placed around the main trunk for gentle manipulation, but excessive traction must be strictly avoided to prevent neurapraxia.
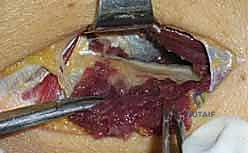
4. Systematic Release of Compressive Sites
The decompression must proceed systematically, addressing all six potential sites of compression.
First, inspect the area proximal to the radiocapitellar joint for any fibrous bands between the brachialis and brachioradialis; if present, they are sharply excised.
Next, locate the vascular leash of Henry crossing the nerve proximal to the supinator. These vessels should be meticulously isolated, ligated with fine silk ties or bipolar electrocautery, and divided to prevent dynamic tethering of the nerve during pronation.
Attention is then turned to the medial border of the ECRB. The deep fascial surface of the ECRB is inspected. If a thickened, tendinous leading edge is impinging upon the nerve, a fractional lengthening or limited fascial release of the ECRB origin is performed, taking extreme care to identify and preserve the small motor branches innervating the ECRB, which often arborize in this immediate vicinity.
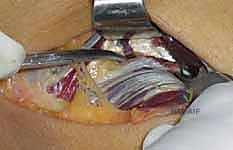
The crux of the procedure is the release of the Arcade of Frohse and the superficial head of the supinator. Using a right-angle clamp, a protective pathway is created deep to the Arcade, superficial to the PIN. The aponeurotic edge and the entire length of the superficial supinator muscle belly are then sharply divided using a scalpel or scissors. This release must extend completely to the distal margin of the supinator to ensure no hourglass constriction remains. As the muscle is divided, the surgeon will observe the PIN lying on the pristine deep head of the supinator.
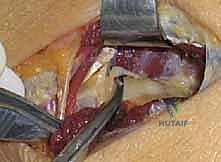
Finally, the assistant supinates and pronates the forearm through a full range of motion while the surgeon directly visualizes the nerve. The nerve should glide freely without any kinking, tethering, or localized blanching. The entire course of the radial tunnel is swept with a blunt instrument to confirm that no aberrant fascial bands or volar capsular structures remain. Once complete hemostasis is achieved after tourniquet deflation, the wound is irrigated, and only the subcutaneous tissue and skin are closed; the deep fascia is left open to prevent post-operative compartment syndrome or recurrent compression from hematoma.

Complications, Incidence Rates, and Salvage Management
While radial nerve decompression is generally highly successful in meticulously selected patients, complications can be devastating and technically demanding to salvage. The most feared complication is iatrogenic nerve injury. This can range from a transient neurapraxia due to excessive traction, to an axonotmesis from crush injury, or a complete neurotmesis (transection) of the PIN, SRN, or the motor branches to the ECRB.
Injury to the PIN results in a catastrophic loss of finger and thumb extension. If a transection is recognized intraoperatively, primary microsurgical epineural repair must be performed immediately. If recognized post-operatively and a complete laceration is confirmed via ultrasound or MRI, early exploration and repair, potentially utilizing nerve autografts (e.g., sural nerve) or allografts, is mandated. For delayed presentations or failed nerve reconstructions, salvage tendon transfers (such as Pronator Teres to ECRB, FCR to EDC, and Palmaris Longus to EPL) provide reliable restoration of motor function.
Incomplete decompression is a frequent cause of persistent post-operative pain and is usually the result of a failure to release the distal extent of the supinator or neglecting the medial edge of the ECRB. Hematoma formation is another significant risk, particularly if meticulous hemostasis is not achieved following tourniquet deflation. A large, expanding hematoma within the tight confines of the proximal forearm can cause acute, secondary compression of the radial nerve, necessitating emergent bedside evacuation or a return to the operating room for washout and hemostasis.
Complications and Management Table
| Complication | Estimated Incidence | Prevention Strategy | Salvage / Management Protocol |
|---|---|---|---|
| Transient Neurapraxia | 5 - 10% | Avoid aggressive retraction; handle nerve with vessel loops only; limit tourniquet time. | Observation; supportive splinting; typically resolves spontaneously within 6 to 12 weeks. |
| PIN Transection | < 1% | Meticulous dissection; visualize nerve prior to cutting the Arcade of Frohse. | Immediate microsurgical repair; late cases require nerve grafting or salvage tendon transfers. |
| Post-op Hematoma | 2 - 4% | Deflate tourniquet prior to closure; achieve pinpoint bipolar hemostasis; leave deep fascia open. | Emergent evacuation if causing neurological deficit; otherwise, warm compresses and observation. |
| Neuroma (Post. Cutaneous N.) | 3 - 5% | Identify and protect the nerve during the initial superficial subcutaneous dissection. | Gabapentinoids; localized steroid injections; surgical excision and burying of the proximal stump into muscle. |
| Incomplete Decompression | 5 - 8% | Systematically address all 6 sites; perform dynamic ROM testing under direct vision intraoperatively. | Re-evaluation with EMG/MRI; revision neurolysis ensuring complete release of the distal supinator. |
Phased Post-Operative Rehabilitation Protocols
The post-operative rehabilitation following radial nerve decompression is strategically phased to balance the protection of the healing soft tissues with the prevention of perineural adhesions. The protocol is divided into three distinct phases, requiring close collaboration between the surgeon and a specialized certified hand therapist (CHT).
Phase 1: Protection and Wound Healing (Weeks 0-2)
Immediately post-operatively, the patient is placed in a bulky, non-compressive soft dressing reinforced with a posterior plaster splint. The elbow is immobilized at 90 degrees of flexion, the forearm in neutral to slight supination, and the wrist in 20 degrees of extension. This specific posture minimizes tension on the newly decompressed radial nerve and prevents dynamic contraction of the supinator and ECRB. The patient is instructed to keep the extremity strictly elevated to minimize edema. Active range of motion of the digits and shoulder is encouraged immediately to prevent distal stiffness and promote venous return.
Phase 2: Early Mobilization and Nerve Gliding (Weeks 2-6)
At the two-week mark, the surgical sutures are removed, and the rigid splint is discontinued. The patient transitions to a removable wrist extension orthosis to be worn during strenuous activities and at night. The focus of this phase shifts to restoring the active and passive range of motion of the elbow and forearm. Gentle, progressive nerve gliding exercises are initiated by the therapist to prevent the radial nerve from adhering to the underlying supinator muscle bed or the overlying fascial scar. Aggressive stretching, forceful gripping, and resisted pronation/supination are strictly prohibited during this phase to avoid provoking inflammation in the surgical bed.
Phase 3: Strengthening and Return to Function (Weeks 6-12+)
Beginning at week six, assuming the patient is relatively pain-free with normal range of motion, a graduated strengthening program is introduced. This begins with isometric exercises for the wrist and forearm musculature, slowly progressing to isotonic and eventually eccentric loading. Work-specific or sport-specific functional rehabilitation is integrated into the therapy sessions. Patients can typically expect to return to light duty or non-manual labor by weeks 4 to 6, while heavy manual labor, repetitive occupational tasks, or racquet sports may require 10 to 12 weeks of recovery to ensure the musculotendinous units have fully healed and the nerve has adapted to the restored anatomical space.
Summary of Landmark Literature and Clinical Guidelines
The evolution of our understanding and surgical management of radial nerve entrapment is deeply rooted in several landmark publications. Michele and Krueger’s 1956 paper was revolutionary, first conceptualizing "radial pronator syndrome" and shifting the paradigm away from the assumption that all lateral elbow pain was epicondylar in origin. This was significantly expanded upon by Roles and Maudsley in 1972, who coined the term "radial tunnel syndrome" and elegantly mapped the anatomical sites of compression, emphasizing the role of the radial nerve in mediating pain despite its motor classification.
More recently, anatomical studies by Spinner and physiological investigations into the microanatomy of the PIN have validated the presence of nociceptive afferent fibers, providing the definitive biological rationale for the pain experienced in RTS. Sponseller's contributions highlighted the critical importance of releasing the distal aspect of the supinator, noting that failure to do so is a primary cause of revision surgery.
Current clinical guidelines and systematic reviews suggest that surgical decompression for appropriately selected patients with radial tunnel syndrome yields good to excellent results in 70% to 85% of cases. However, the literature consistently underscores a critical caveat: the outcomes for radial nerve decompression are historically less predictable and less uniformly successful than those for carpal or cubital tunnel syndromes. This discrepancy reinforces the absolute necessity of an exhaustive preoperative diagnostic workup, a prolonged trial of conservative management, and meticulous intraoperative execution to ensure every potential tethering point is liberated.
