Masterclass: Advanced Rotational and Pedicle Flaps for Distal Upper Extremity Reconstruction
Key Takeaway
Join us in the OR for a deep dive into rotational and pedicle flaps for distal upper extremity injuries. Learn critical anatomy, meticulous surgical steps, and advanced techniques for radial forearm, groin, kite, and posterior interosseous flaps, ensuring optimal patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Alright, fellows, gather around the table. Today, we are delving into the nuanced, high-stakes world of soft tissue coverage for distal upper extremity injuries. This is the precise intersection where the art of spatial geometry meets the rigorous science of microvascular physiology, where meticulous preoperative planning and flawless intraoperative execution dictate ultimate functional outcomes. Our overarching goal as reconstructive orthopedic surgeons is never merely to close a wound; it is to meticulously restore form, function, and sensibility to the human hand. The distal upper extremity presents a unique reconstructive challenge due to its paucity of redundant soft tissue, the superficial location of critical gliding structures (tendons, nerves, and vessels), and the paramount importance of preserving highly specialized glabrous skin.
The epidemiology of distal upper extremity soft tissue defects is predominantly driven by high-energy trauma, including industrial crush injuries, motor vehicle collisions, and agricultural accidents. Furthermore, oncologic resections for soft tissue sarcomas or aggressive cutaneous malignancies, as well as severe necrotizing soft tissue infections, frequently leave profound soft tissue voids. When vital structures are denuded of their vascularized enveloping tissues, secondary intention healing or simple skin grafting is not just suboptimal; it is functionally catastrophic. The resulting desiccation of tendons and bone leads to necrosis, profound scarring, and intractable stiffness. Therefore, the reconstructive surgeon must be intimately familiar with the "reconstructive ladder"—and more contemporarily, the "reconstructive elevator"—to select the most appropriate vascularized tissue transfer.
Let us begin with foundational definitions. This is not mere semantics; it is the absolute crux of our surgical approach. A precise understanding of tissue transfer mechanics is essential. A flap is a composite collection of tissue—skin, fascia, muscle, or bone—that is mobilized from its native anatomical bed to a recipient site while critically maintaining its intrinsic blood supply. This vascular autonomy is the definitive differentiator from a graft. Grafts, conversely, are completely detached from their native blood supply and rely entirely on the recipient bed for survival. A skin graft initially survives via passive diffusion of nutrients (plasmatic imbibition) from the recipient bed, followed days later by capillary ingrowth (inosculation). This delicate biological process mandates a highly vascularized recipient bed; grafts will unequivocally fail over exposed bare bone, denuded tendon lacking paratenon, or exposed hardware.
When we classify flaps, we must categorize them by their inherent vascular supply and their geometry of movement. Pedicled Flaps remain attached to their original blood supply (the pedicle) as they are transposed. Free Flaps represent the zenith of reconstructive complexity, requiring the complete division of the flap's dominant artery and vein, followed by microscopic reanastomosis to recipient vessels at the defect site. Among local pedicled flaps, we define them by their vector of movement:
* Advancement Flaps: These are elevated and advanced in a strictly linear vector away from the base of their pedicle. The inherent elasticity of the skin is utilized to stretch the tissue directly forward over the defect.
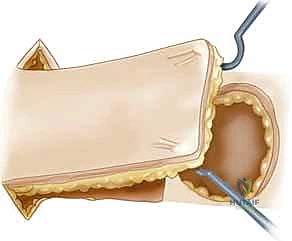
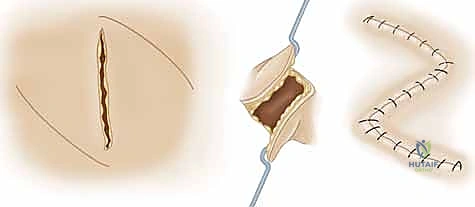
* Rotational Flaps: These are semicircular flaps elevated adjacent to the defect and rotated around a pivot point into the defect within the same tissue plane. Crucially, the effective radius of the flap decreases as the angle of rotation increases. A strategic backcut or Burow's triangle excision can extend the arc of coverage, but one must remain exquisitely mindful not to compromise the base pedicle.
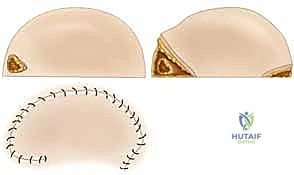
* Transpositional Flaps: Geometrically similar to rotational flaps, these are elevated and moved laterally across an intervening bridge of normal, intact tissue to reach a new, adjacent defect site.

* Axial Flaps: These robust flaps depend on the continuous blood supply from a single, consistent, anatomically named blood vessel running along the longitudinal axis of the flap. The radial forearm flap and dorsal metacarpal artery flaps are classic examples, providing highly predictable and reliable perfusion.
* Random Flaps: These lack a named axial vessel and rely entirely on preserving a sufficient network of the subcutaneous and subdermal vascular plexus for survival. Z-plasties, V-Y advancements, and traditional cross-finger flaps fall into this category. Their length-to-width ratio is strictly limited to ensure distal perfusion.
Detailed Surgical Anatomy and Biomechanics
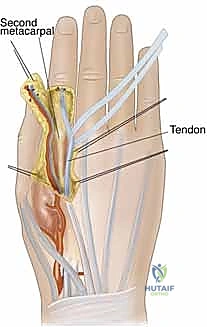
A profound, three-dimensional understanding of upper extremity vascular anatomy is the non-negotiable prerequisite for safe flap elevation. The vascular architecture of the forearm and hand is characterized by a high degree of redundancy, primarily supplied by the radial and ulnar arteries, which terminate in the superficial and deep palmar arches. This redundancy is the physiological basis that allows us to sacrifice one major axial vessel (e.g., the radial artery in a radial forearm flap) without compromising distal limb perfusion, provided the collateral circulation is intact. The concept of the "angiosome," introduced by Taylor and Palmer, dictates that specific three-dimensional blocks of tissue are supplied by distinct source arteries. Understanding the interconnecting "choke vessels" between these angiosomes allows the surgeon to design extended flaps that capture adjacent vascular territories.
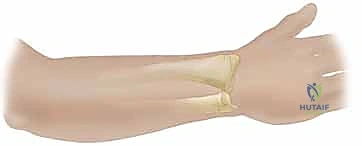
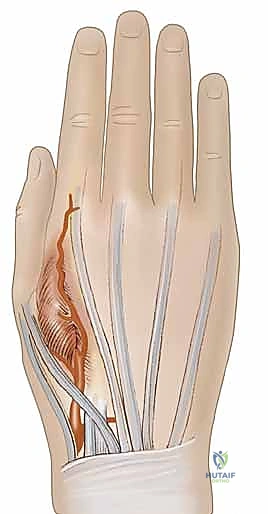
The Radial Forearm Flap (RFF), a true workhorse in our reconstructive armamentarium, perfectly illustrates these anatomical principles. It is a fasciocutaneous flap based on the radial artery and its paired venae comitantes. The radial artery courses distally through the forearm in the intermuscular septum between the brachioradialis (BR) and the flexor carpi radialis (FCR) muscles.
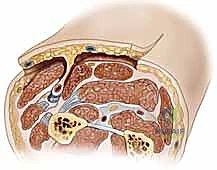
Within this loose areolar septum, numerous critical septocutaneous perforating branches arise from the radial artery to supply the overlying antebrachial fascia and skin. During flap elevation, the surgeon must meticulously preserve this fascial plexus; stripping the fascia from the artery will inevitably devascularize the cutaneous paddle.
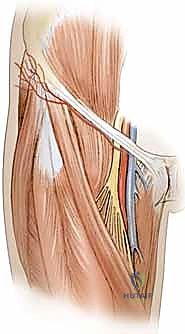
Venous drainage of the distal upper extremity is dual-layered, comprising the deep venae comitantes accompanying the major arteries and a robust superficial venous system (the cephalic and basilic veins). When a pedicled flap is designed for antegrade flow, venous drainage is physiological. However, when designing a reverse-flow pedicled flap (e.g., a reverse radial forearm flap to cover a dorsal hand defect), the arterial inflow is retrograde via the palmar arches. More importantly, the venous outflow must also occur retrogradely through the venae comitantes. This retrograde venous flow must overcome the resistance of venous valves. Fortunately, the rich network of interconnecting bypass channels (cross-connections) between the paired venae comitantes allows blood to circumvent these valves, making reverse-flow flaps viable, albeit more susceptible to venous congestion.
Neurological anatomy is equally critical, primarily to avoid crippling donor site morbidity. In the distal third of the forearm, the superficial branch of the radial nerve emerges from deep to the brachioradialis to become subcutaneous, providing critical sensation to the dorsal-radial aspect of the hand. During the elevation of a radial forearm flap, this nerve lies in extreme proximity to the radial artery. It must be definitively identified, meticulously neurolyzed, and protected unless a sensate flap is specifically planned, in which case a portion of the nerve or the lateral antebrachial cutaneous nerve is harvested with the flap. Iatrogenic injury to the superficial radial nerve results in a painful neuroma, a complication that often overshadows the success of the reconstruction.
Biomechanically, the skin of the upper extremity exhibits significant regional variance in thickness, elasticity, and adherence to underlying structures. The volar palmar skin is highly specialized: thick, glabrous, lacking hair follicles, and firmly anchored to the underlying palmar aponeurosis by robust vertical fascial septa. This architecture prevents shear during grip. Conversely, the dorsal skin is thin, highly pliable, and loosely attached via an areolar layer to accommodate the extreme excursion of the extensor tendons during digital flexion. When reconstructing defects, the surgeon must adhere to the principle of "like with like." A bulky fasciocutaneous flap placed on the dorsum of the hand will mechanically impede tendon glide and require subsequent secondary debulking procedures.
Exhaustive Indications and Contraindications
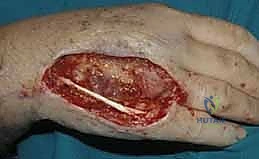
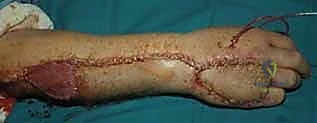
The decision-making process regarding when to employ a flap versus a graft is fundamental to reconstructive success. While many superficial wounds can heal via secondary intention or accept a split-thickness skin graft, these modalities are entirely inappropriate for complex defects. Exposed vital structures unequivocally mandate vascularized flap coverage. The primary indications for advanced rotational and pedicle flaps in the distal upper extremity include traumatic avulsions, massive crush injuries, thermal or electrical burns resulting in full-thickness necrosis, and oncologic defects following tumor extirpation where bone, tendon (devoid of paratenon), major nerves, or vascular bundles are exposed.
In the context of tendon exposure, a skin graft placed directly over bare tendon will fail, leading to tendon desiccation and rupture. Even if a graft were to "take" on a marginally vascularized bed, the resulting cicatrix would firmly tether the tendon to the overlying skin, completely obliterating the gliding mechanism and resulting in a stiff, non-functional digit. A fasciocutaneous or adipofascial flap provides a pliable, well-vascularized gliding surface that prevents this restrictive adhesion formation, facilitating early postoperative mobilization and optimal functional recovery. Similarly, exposed bone devoid of periosteum will undergo desiccation and avascular necrosis if not promptly covered with robust vascularized tissue.
However, the application of these complex flaps is not without significant risk, and rigorous patient selection is critical. Contraindications must be carefully weighed.
| Category | Specific Factors | Rationale / Clinical Consequence |
|---|---|---|
| Absolute Contraindications | Inadequate vascular inflow/outflow | A reverse-flow flap (e.g., reverse RFF) is absolutely contraindicated if the ulnar artery is occluded or the palmar arch is incomplete (failed Allen's test). Ischemia of the hand will result. |
| Active, uncontrolled infection | Flapping over gross purulence or necrotic tissue guarantees flap failure and deep space infection. Aggressive, staged serial débridement is mandatory prior to coverage. | |
| Unreconstructable vascular injury | If the entire limb is non-viable or requires amputation due to irreversible ischemia, local flap coverage is futile. | |
| Relative Contraindications | Severe systemic comorbidities (e.g., uncontrolled diabetes, ESRD) | Microvascular disease impairs collateral flow and wound healing, significantly increasing the risk of partial or total flap necrosis. |
| Active tobacco use | Nicotine causes profound peripheral vasoconstriction, increasing thrombotic risk and diminishing microcirculatory flow. Patients should ideally cease smoking 4 weeks pre- and post-op. | |
| Prior trauma to the donor site | Previous scarring or surgery in the planned flap territory may have disrupted the critical perforating vessels or axial pedicle, rendering the flap unreliable. | |
| Patient non-compliance | Complex reconstructions require rigorous postoperative rehabilitation and strict adherence to immobilization protocols. Non-compliant patients risk avulsing the pedicle or stiffening the hand. |
When evaluating a patient with a severe crush injury, the surgeon must be highly suspicious of the "zone of injury." The visible cutaneous defect often drastically underestimates the true extent of deep tissue trauma. Crush injuries induce profound microvascular thrombosis and progressive tissue necrosis over several days. Attempting definitive flap coverage on day one in a severe crush injury is a recipe for disaster. These wounds require serial, meticulous débridements until a definitively stable, healthy, bleeding wound bed is established. Only then should flap coverage be executed.
Pre-Operative Planning, Templating, and Patient Positioning
Gentlemen, I cannot overstate this: the most critical phase of any complex microvascular or pedicled reconstruction occurs entirely outside the operating room. Meticulous preoperative planning is the bedrock of surgical success. It begins with a comprehensive history and physical examination. The mechanism of injury dictates the surgical timeline. A sharp, clean guillotine amputation or laceration may be amenable to immediate flap coverage. Conversely, an abrasive avulsion from a motor vehicle accident implies high contamination and extensive microvascular trauma, necessitating a staged approach with temporizing negative pressure wound therapy (NPWT).
Vascular assessment is the absolute cornerstone of preoperative planning for upper extremity flaps. Before considering any flap that sacrifices a major axial vessel (such as the radial or ulnar artery), the patency and dominance of the collateral circulation must be definitively proven. The Modified Allen's Test is mandatory. The examiner manually occludes both the radial and ulnar arteries at the wrist while the patient repeatedly clenches their fist to exsanguinate the hand. The patient then opens their hand (which should appear blanched). The examiner releases the pressure on the ulnar artery while maintaining pressure on the radial artery. The hand should flush pink within 3 to 5 seconds. If capillary refill is delayed or absent, the ulnar collateral circulation via the palmar arches is insufficient, and harvesting the radial artery is absolutely contraindicated, as it would precipitate catastrophic hand ischemia. We routinely supplement the clinical Allen's test with handheld Doppler ultrasound mapping of the palmar arches and digital vessels, and in complex trauma, formal CT angiography is utilized to delineate the precise vascular architecture and identify any occult zones of injury.
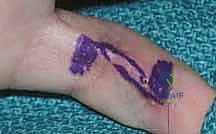
Once vascular patency is confirmed, precise templating of the defect is performed. We typically use a sterile Esmarch bandage wrapper or the paper from a sterile glove package to create an exact two-dimensional template of the recipient defect.
This template is then transferred to the donor site. It is a fundamental reconstructive axiom to "plan in reverse." The template is placed on the donor site, and the pivot point of the pedicle is marked. The flap is then simulated by rotating the template from the donor site to the recipient site to ensure that the pedicle length is adequate to reach the most distal aspect of the defect without undue tension. Always design the flap 10-15% larger than the templated defect to account for primary tissue contraction upon elevation and to ensure a tension-free inset.
Proper patient positioning and operating room setup are essential for facilitating meticulous microsurgical technique and minimizing surgeon fatigue during prolonged cases. The patient is positioned supine. The affected upper extremity is placed on a radiolucent, well-padded arm board, abducted to no more than 90 degrees to prevent traction injury to the brachial plexus. A pneumatic tourniquet is applied high on the brachium over generous cast padding. Standard sterile draping must expose the entire upper extremity from the axilla down to the fingertips to allow for assessment of limb perfusion, access to proximal vessels, and the ability to intraoperatively alter the reconstructive plan if necessary. If a free tissue transfer or complex microvascular dissection is anticipated, the operating microscope must be draped and balanced prior to the start of the case, and the room geometry arranged so that both the primary surgeon and the assistant can sit comfortably with their elbows supported.
Step-by-Step Surgical Approach and Fixation Technique
Let us walk through the intraoperative execution of several workhorse flaps, beginning with the definitive elevation of the Radial Forearm Flap (RFF).
The Reverse Radial Forearm Flap
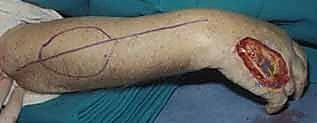
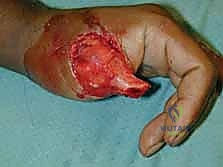
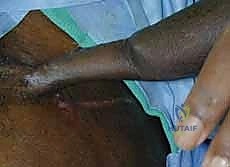
The reverse RFF is the gold standard for robust coverage of complex dorsal hand and wrist defects. Following exsanguination of the limb and tourniquet inflation, the templated flap design is incised on the volar forearm.
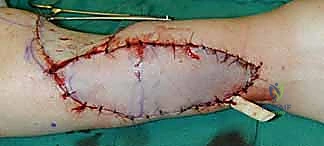
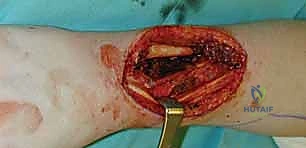
Dissection begins distally and proceeds proximally. The skin and subcutaneous tissues are incised down to the antebrachial fascia. It is imperative to elevate the flap in a subfascial plane to capture the delicate septocutaneous perforators.
As the dissection approaches the radial border of the flexor carpi radialis (FCR), extreme caution is exercised to preserve the thin paratenon overlying the FCR tendon. Stripping the paratenon will preclude successful subsequent skin grafting of the donor site.
The radial artery and its venae comitantes are identified in the distal forearm.
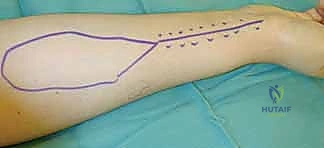
The pedicle is ligated proximally (for a reverse-flow flap) and meticulously dissected free from its muscular bed between the FCR and brachioradialis, sweeping the perforators up into the overlying fascia of the flap.
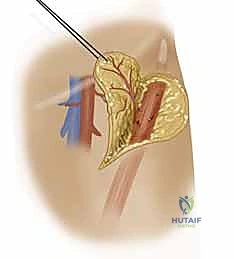
Simultaneously, the superficial branch of the radial nerve is identified deep to the brachioradialis and carefully neurolyzed away from the pedicle to ensure it remains in the donor bed.
Once the flap is completely islanded on its distal pedicle, the tourniquet is deflated to confirm retrograde perfusion via the palmar arch. The flap should exhibit brisk capillary refill and robust dermal bleeding.
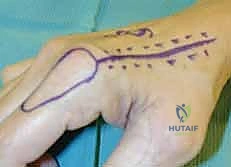
A wide subcutaneous tunnel is then bluntly dissected from the pivot point at the wrist to the dorsal recipient defect. The tunnel must be exceptionally capacious; any compression of the pedicle within the tunnel will lead to catastrophic venous congestion.
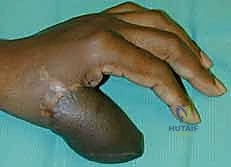
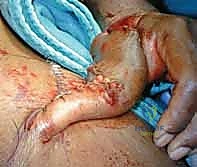
The flap is gently passed through the tunnel and inset into the defect using interrupted half-buried mattress sutures to minimize tension on the flap edges.
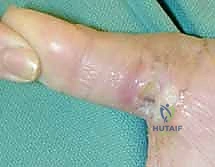
The donor site is typically resurfaced with a full-thickness or thick split-thickness skin graft, meticulously quilted to the muscle bellies and the preserved paratenon of the FCR and BR.
Local Digital Flaps: The Cross-Finger and V-Y Advancement
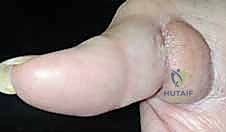
For complex volar distal phalangeal amputations with exposed bone, where secondary healing is inappropriate, local random or axial flaps are utilized. The V-Y Advancement Flap (Atasoy flap) is an excellent choice for transverse or dorsal-oblique fingertip amputations.
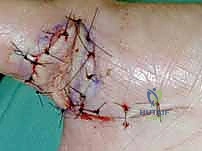
A V-shaped incision is made on the volar pad, with the apex at the distal interphalangeal (DIP) joint crease.