Rectus Femoris Transfer for Stiff-Knee Gait in Cerebral Palsy: An Intraoperative Masterclass
Key Takeaway
This masterclass guides fellows through the rectus femoris transfer, a crucial procedure for stiff-knee gait in cerebral palsy. We'll cover in-depth anatomy, meticulous surgical technique, optimal patient positioning, and critical intraoperative decision-making. Learn to identify and mitigate risks, ensuring precise tendon transfer and effective postoperative rehabilitation for improved patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
The management of ambulatory children with cerebral palsy (CP) represents one of the most intellectually demanding and technically intricate domains within pediatric orthopedic surgery. Central to this challenge is the mitigation of the "stiff-knee gait," a pervasive kinematic abnormality characterized by delayed and severely diminished peak knee flexion during the swing phase of the gait cycle. The Rectus Femoris Transfer (RFT) has emerged as a cornerstone intervention, typically executed within the broader framework of Single-Event Multilevel Surgery (SEMLS). This operative masterclass is designed to elucidate the profound biomechanical rationale, rigorous patient selection criteria, and meticulous intraoperative execution required to optimize outcomes for these complex patients.
To truly master this procedure, one must first deconstruct the pathogenesis of the stiff-knee gait. From a functional and biomechanical perspective, the quadriceps femoris must not be viewed as a monolithic extensor mechanism. Rather, it operates as two distinct functional units: the biarticular rectus femoris muscle, and the uniarticular "triceps femoris" (comprising the vastus lateralis, vastus medialis, and vastus intermedius). In the neurologically intact individual, the rectus femoris remains largely silent during the initial swing phase, allowing the knee to passively flex secondary to the momentum generated by hip flexion and ankle plantarflexion (the "pull-off" and "push-off" phases).
In the child with spastic diplegic or hemiplegic cerebral palsy, an upper motor neuron lesion disrupts this elegant orchestration. The hallmark of this disruption is dynamic overactivity and inappropriate firing of the rectus femoris during the swing phase. Clinically, this manifests as a profound resistance to passive knee flexion just as the limb attempts to clear the ground. The resulting stiff-knee gait is far more than an aesthetic deviation; it is a biomechanical catastrophe that forces the child to adopt highly energy-inefficient compensatory mechanisms. To achieve foot clearance and prevent toe-dragging or tripping, the patient must resort to circumduction of the swing limb, vaulting on the contralateral stance limb, or exaggerated ipsilateral hip hiking. Over time, these compensations lead to premature fatigue, abnormal joint loading, and a progressive decline in ambulatory endurance.
Furthermore, the natural history of spasticity in the growing child dictates a transition from dynamic dysfunction to fixed structural pathology. In the early developmental years (typically up to age 6), the rectus femoris exhibits a purely dynamic dysfunction—the muscle possesses a normal resting sarcomere length but demonstrates an exaggerated, velocity-dependent response to stretch. However, as the child's skeletal growth outpaces the spastic muscle's ability to elongate, this dynamic state inexorably progresses to fixed myostatic shortening between the ages of 6 and 10. The sarcomeres undergo structural remodeling, resulting in a permanent contracture. The surgical transfer of the rectus femoris is specifically designed to address both the dynamic spasticity and the fixed contracture, effectively converting a pathological knee extensor moment into a functional knee flexor moment, thereby restoring swing-phase kinematics and preserving vital hip flexion power.
Detailed Surgical Anatomy and Biomechanics
A profound mastery of the anterior thigh's surgical anatomy is non-negotiable for the safe and effective execution of the rectus femoris transfer. The rectus femoris is unique among the quadriceps musculature due to its biarticular nature, crossing both the hip and the knee joints. Its proximal origin is dual and complex: the direct head arises from the anterior inferior iliac spine (AIIS), while the reflected head originates from the innominate bone just proximal to the superior margin of the acetabulum, blending with the anterior hip capsule. This dual origin provides the muscle with a significant mechanical advantage as a hip flexor, particularly when the knee is flexed.
Distally, the rectus femoris transitions into a robust aponeurotic tendon that fuses with the underlying vastus intermedius several centimeters proximal to the superior pole of the patella. Together with the vastus lateralis and vastus medialis, these structures converge to form the common quadriceps tendon, which inserts onto the patella and continues distally as the patellar ligament to insert on the tibial tubercle. Biomechanically, the rectus femoris possesses a relatively small physiologic cross-sectional area (PCSA) but a large ratio of tendon length to muscle fiber length. This architectural design indicates that the muscle is optimized for maximal excursion and rapid velocity rather than the generation of massive force, making it highly susceptible to the velocity-dependent stretch reflexes characteristic of spasticity.
The neurovascular anatomy of the anterior thigh dictates the limits of our proximal mobilization during the transfer. The rectus femoris is innervated by the femoral nerve (L2-L4). The specific motor branches to the rectus femoris enter the muscle at its proximal third, deep to its posterior fascia. Aggressive or blind proximal dissection during the mobilization phase can easily result in catastrophic denervation of the muscle, rendering the transfer completely useless.
Similarly, the vascular supply must be meticulously preserved. The primary blood supply to the rectus femoris is derived from the descending branch of the lateral circumflex femoral artery, which enters the muscle proximally in close proximity to the motor nerve branches. While the distal two-thirds of the muscle can be safely mobilized from the underlying vastus intermedius, the surgeon must remain acutely aware of small perforating vessels traversing the intermuscular plane. The lateral femoral cutaneous nerve, providing sensation to the anterolateral thigh, courses superficially after piercing the fascia lata near the anterior superior iliac spine (ASIS). Although generally proximal and lateral to our primary surgical field, aberrant branches can be encountered during subcutaneous dissection, necessitating a careful, layered approach.
Exhaustive Indications and Contraindications
The decision to proceed with a rectus femoris transfer must never be made based on observational gait analysis alone. A rigorous, multimodal diagnostic matrix is required to isolate the specific kinematic deficits and ensure the patient possesses the prerequisite biomechanical foundation to benefit from the procedure. Meticulous patient selection is the primary determinant of surgical success.
Clinical Examination and the Diagnostic Matrix
The physical examination begins with the prone rectus femoris test, commonly known as the Duncan-Ely test. With the patient prone and the pelvis stabilized, the examiner rapidly flexes the knee. A positive "fast" rectus test—characterized by a sudden catch and spontaneous elevation of the ipsilateral hip off the examining table—indicates severe dynamic spasticity. Conversely, a positive "slow" rectus test, where hip elevation occurs even with slow, controlled knee flexion, confirms the presence of a fixed myostatic contracture.
However, clinical examination alone is insufficient. Three-dimensional Instrumented Gait Analysis (IGA) is the gold standard and an absolute prerequisite for this procedure. IGA provides objective, quantifiable data regarding temporospatial parameters, sagittal-plane kinematics, and dynamic electromyography (EMG).

Kinematic and Kinetic Prerequisites
The primary kinematic indicators for transfer include a decreased range and velocity of knee flexion during the stance-to-swing transition (pre-swing), followed by a delayed and severely diminished peak knee flexion angle during the mid-swing phase. The dynamic range of motion is often blunted, presenting as a flattened or "mounded" waveform on the kinematic graph.
Crucially, the surgeon must evaluate the prerequisite mechanics required for a successful transfer. Gait velocity is a paramount consideration; it must be greater than 60% of age-matched normative values. The swing phase of gait is largely a passive phenomenon driven by momentum. If a child's gait velocity is profoundly diminished, they lack the kinetic energy necessary to propel the limb forward, and transferring the rectus femoris will not miraculously restore knee flexion. Furthermore, robust hip flexor power at terminal stance is mandatory. A poor transition at the hip, characterized by inadequate hip flexion velocity, will result in poor "pull-off" mechanics. In this scenario, the stiff knee is secondary to proximal weakness, and a rectus transfer is strictly contraindicated.
Dynamic Electromyography (EMG)
Dynamic EMG is indispensable for confirming the pathological firing patterns of the rectus femoris. In a normal gait cycle, the rectus femoris demonstrates a brief burst of activity at initial contact to stabilize the knee, followed by silence through mid-stance and swing. In the stiff-knee CP patient, surface or fine-wire EMG will reveal prolonged, intense, and entirely inappropriate electrical activity extending well into the middle subphase of swing. This objective confirmation of inappropriate firing is the definitive indication that the muscle is actively resisting knee flexion.
| Parameter | Indications for Rectus Femoris Transfer | Contraindications for Rectus Femoris Transfer |
|---|---|---|
| Clinical Exam | Positive Duncan-Ely Test (Fast or Slow) | Negative Duncan-Ely Test; Severe knee flexion contracture |
| Gait Velocity | > 60% of age-matched normative values | < 60% of age-matched normative values |
| Knee Kinematics | Delayed/diminished peak knee flexion in swing | Normal peak knee flexion timing and amplitude |
| Hip Kinematics | Adequate hip flexion velocity at pre-swing | Poor hip flexor power/velocity at stance-to-swing transition |
| Dynamic EMG | Continuous, inappropriate firing during swing phase | Silent rectus femoris during mid-to-late swing phase |
| Leg Length | Ipsilateral limb is equal or longer than contralateral | Ipsilateral limb is significantly shorter (needs functional lengthening) |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough preoperative planning sets the stage for flawless intraoperative execution. Because the rectus femoris transfer is rarely performed in isolation—often accompanying hamstring lengthenings, calf procedures, or derotational osteotomies within a SEMLS framework—the sequence of procedures must be carefully choreographed. Generally, bony procedures are addressed prior to soft tissue transfers to ensure accurate tensioning of the muscle-tendon units over the newly established skeletal alignment.
Examination Under Anesthesia (EUA)
Upon induction of general anesthesia, a comprehensive EUA is mandatory. This critical step eliminates the influence of volitional guarding and dynamic spasticity, allowing the surgeon to precisely quantify the degree of fixed myostatic contracture. The Duncan-Ely test is repeated; if the previously noted "catch" disappears, the pathology was purely dynamic. If a restriction remains, a structural contracture is present, dictating the necessity of a formal surgical release and transfer rather than a simple recession. Range of motion at the hip, knee, and ankle is meticulously documented to serve as a baseline for postoperative rehabilitation.
Positioning and Sterile Setup
The patient is positioned supine on a fully radiolucent operating table, essential if concurrent bony osteotomies require fluoroscopic guidance. A small, radiolucent bump may be placed under the ipsilateral hemipelvis to control excessive external rotation of the limb, ensuring the patella points directly toward the ceiling.
A sterile pneumatic tourniquet is applied to the most proximal aspect of the thigh. The use of a tourniquet is highly recommended; a bloodless surgical field is paramount for the meticulous identification of intermuscular planes and the safe mobilization of the rectus femoris without compromising its delicate proximal neurovascular pedicle. The limb is then subjected to a rigorous surgical prep, typically extending from the iliac crest to the toes.
Draping must be executed to allow the limb to be entirely free-floating. The surgeon must have the ability to passively flex the hip and knee through their absolute maximum arcs of motion. Restrictive draping that prevents full knee flexion will catastrophically impair the surgeon's ability to appropriately tension the transfer later in the procedure. The foot is enveloped in a sterile stockinette or impervious drape but remains accessible for manipulation.
Step-by-Step Surgical Approach and Fixation Technique
The execution of the rectus femoris transfer requires a meticulous balance of aggressive mobilization and delicate tissue handling. The objective is to completely isolate the muscle-tendon unit, reroute it to a flexor insertion, and secure it with appropriate tension to convert its biomechanical vector.
Surgical Exposure and Fascial Incision
The procedure commences with an 8 to 12-cm longitudinal incision centered over the anterior aspect of the distal third of the thigh. The incision is carried distally, terminating approximately one to two fingerbreadths proximal to the superior pole of the patella. Subcutaneous tissues are divided sharply, maintaining a pristine plane superficial to the deep fascia to minimize the risk of postoperative fat necrosis and seroma formation.
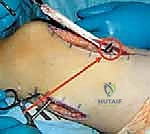
Upon exposing the glistening fascia lata, an incision is made along the entire length of the surgical wound. This fascial release immediately exposes the underlying quadriceps musculature. The rectus femoris is identified centrally, distinguished by its bipennate muscle fibers converging onto a robust central tendon. It is flanked laterally by the vastus lateralis and medially by the vastus medialis.
Isolation and Distal Release
Using precise blunt dissection, utilizing a combination of Metzenbaum scissors and the surgeon's index finger, the areolar tissue between the rectus femoris and the adjacent vasti is swept away. This defines the medial and lateral borders of the muscle.
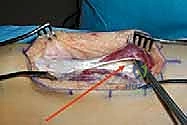
Attention is then directed distally to the tendinous insertion. The dissection is carried directly onto the superior pole of the patella. Using electrocautery or a sharp scalpel, the rectus femoris tendon is meticulously detached from the patella. It is critical to ensure that only the rectus femoris is released; the underlying vastus intermedius tendon must remain perfectly intact to preserve the primary extensor mechanism of the knee.
Proximal Mobilization: The Critical Step
Once the distal tendon is liberated, a locking Krackow or whipstitch using a heavy, non-absorbable suture (e.g., #2 FiberWire) is immediately placed into the free end of the tendon. This suture serves as a secure handle for traction.
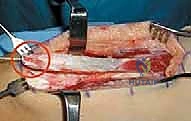
The surgeon now begins the most critical phase of the procedure: proximal mobilization. Applying distal traction via the suture, the surgeon develops the avascular plane between the deep surface of the rectus femoris and the superficial surface of the vastus intermedius. This mobilization must extend proximally to the junction of the proximal and middle thirds of the thigh.

Inadequate proximal mobilization is the leading cause of transfer failure. If the muscle is not freed sufficiently, it will act merely as a tether, failing to provide the necessary excursion for knee flexion. During this proximal dissection, the surgeon must remain hyper-vigilant for the neurovascular pedicle entering the deep, proximal aspect of the muscle. Gentle blunt dissection is mandatory here; aggressive tearing will avulse the motor branches of the femoral nerve. Small perforating vessels crossing the intermuscular plane are identified and coagulated.
Tunneling and Routing the Transfer
With the rectus femoris fully mobilized, the surgeon must select the transfer site. The most common and biomechanically sound target is the sartorius or the gracilis/semitendinosus complex medially. Lateral transfer to the iliotibial band is an alternative but is generally reserved for specific rotational deformities.
For a medial transfer, a subcutaneous tunnel is created. Using a long Kelly clamp or a specialized tendon passer, a generous tunnel is blunted from the primary anterior incision, coursing medially and distally around the medial aspect of the distal femur, aiming toward the medial hamstrings or sartorius insertion. The tunnel must be exceptionally wide to accommodate the bulky muscle belly of the rectus femoris without any fascial constriction.

The traction sutures are passed through the tunnel, and the rectus femoris tendon is gently pulled into its new medial position. The surgeon must verify that the line of pull is straight and unimpeded by fascial bands.
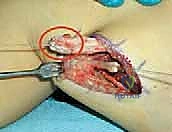
Fixation and Tensioning
The recipient tendon (e.g., sartorius) is identified and isolated. The fixation technique typically involves a side-to-side anastomosis or a modified Pulvertaft weave if tendon length permits.
Tensioning the transfer is an art form requiring precise clinical judgment. The hip is placed in neutral extension (0 degrees), and the knee is flexed to approximately 30 to 45 degrees.
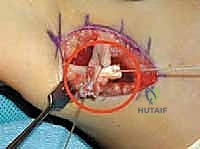
While maintaining this specific joint alignment, the rectus femoris tendon is pulled distally to remove all slack without overstretching the muscle spindle fibers. The tendon is then sutured securely to the recipient tendon using multiple interrupted figure-of-eight stitches with heavy non-absorbable suture.

Following fixation, the knee is passively taken through a full range of motion. The surgeon must confirm that full extension is achievable without excessive tension on the anastomosis, and that the knee easily flexes past 90 degrees. The tourniquet is deflated, meticulous hemostasis is achieved, and the fascial layer is closed loosely over a closed-suction drain if necessary, followed by standard subcutaneous and subcuticular skin closure.
Complications, Incidence Rates, and Salvage Management
While the rectus femoris transfer is highly effective, it is not without risk. Complications can arise from technical errors during execution or from poor patient selection. A comprehensive understanding of these pitfalls is essential for the operating surgeon.
Inadequate proximal mobilization is the most frequent technical failure, resulting in a persistent stiff-knee gait. If the muscle remains tethered to the underlying vastus intermedius by fascial adhesions, it cannot glide, effectively acting as a static tenodesis rather than a dynamic transfer. Conversely, over-tensioning the transfer—suturing it tightly with the knee in excessive flexion—can lead to a postoperative extensor lag, significantly impairing the patient's stance-phase stability.
Iatrogenic nerve injury, specifically to the motor branches of the femoral nerve supplying the rectus femoris, is a catastrophic complication that completely negates the purpose of the transfer. This occurs during overly aggressive or blind proximal dissection. Hematoma formation is also a notable risk due to the extensive dead space created between the rectus and vastus intermedius.
| Complication | Estimated Incidence | Etiology / Risk Factors | Salvage / Management Strategy |
|---|---|---|---|
| Persistent Stiff Knee | 10 - 15% | Inadequate proximal mobilization; Poor patient selection (low velocity) | Revision surgery for further mobilization; Re-evaluation of proximal hip mechanics |
| Extensor Lag | 5 - 8% | Over-tensioning of the transfer; Inadvertent damage to vastus intermedius | Aggressive physical therapy; Rarely requires surgical lengthening of the transfer |
| Hematoma / Seroma | 3 - 5% | Inadequate hemostasis; Failure to use a drain in large dead spaces | Observation for small hematomas; Surgical evacuation and washout for large/expanding lesions |
| Iatrogenic Denervation | < 1% | Aggressive proximal dissection avulsing femoral nerve branches | Irreversible. Focus shifts to maximizing triceps femoris function via therapy |
| Superficial Infection | 1 - 2% | Poor soft tissue handling; Contamination | Oral or intravenous antibiotics; Local wound care |
Phased Post-Operative Rehabilitation Protocols
The surgical intervention represents only half of the therapeutic equation; rigorous, phased postoperative rehabilitation is critical to integrating the transferred muscle into the child's central motor programming. The rehabilitation protocol must be tailored to the specific combination of procedures performed during the SEMLS.
Phase I: Protection and Healing (Weeks 0-4)
Immediately postoperatively, the limb is immobilized. If the rectus transfer was performed in isolation or with minor soft-tissue procedures, a removable knee immobilizer locked in full extension is utilized. If concurrent bony osteotomies were performed, a short-leg or long-leg cast may be required.
Weight-bearing status is dictated by the concurrent procedures. For isolated soft-tissue transfers, weight-bearing as tolerated with assistive devices (crutches or a walker) is encouraged immediately to stimulate proprioception and maintain bone density. The primary goal during this phase is the protection of the surgical anastomosis and the management of edema and pain. Passive range of motion (PROM) of the knee is initiated cautiously, typically restricted to 0-60 degrees to prevent excessive tension on the healing transfer.
Phase II: Active Mobilization and Motor Retraining (Weeks 4-8)
At four weeks, formal immobilization is gradually weaned. The focus shifts to active-assisted and active range of motion. The physical therapist plays a crucial role in motor retraining. Because the rectus femoris has been anatomically converted from a knee extensor to a knee flexor, the child's motor cortex must adapt to this new biomechanical reality.
Biofeedback and neuromuscular electrical stimulation (NMES) are highly effective adjunctive therapies during this phase. Therapists focus on initiating active knee flexion during the swing phase of gait using parallel bars or a body-weight-supported treadmill system. Ankle-foot orthoses (AFOs) are meticulously adjusted to ensure optimal alignment of the foot and ankle, providing a stable platform for the knee to operate.
Phase III: Strengthening and Functional Integration (Weeks 8+)
By the eighth postoperative week, the tendon anastomosis is robustly healed. Rehabilitation transitions to aggressive strengthening of the remaining triceps femoris to prevent extensor lags, alongside strengthening of the newly transferred rectus femoris in its flexor role.
Dynamic activities, including stair climbing, cycling, and advanced gait training, are incorporated. The ultimate goal is to increase the child's self-selected walking speed and endurance. It is vital to educate parents that the maximum functional benefit of a rectus femoris transfer may not be fully realized until 12 to 18 months postoperatively, as the central nervous system slowly integrates the altered biomechanics into spontaneous gait patterns.
Summary of Landmark Literature and Clinical Guidelines
The evolution of the rectus femoris transfer is deeply rooted in the pioneering work of orthopedic surgeons and biomechanists who championed the use of 3D Instrumented Gait Analysis. Historically, the treatment for a spastic rectus femoris was a simple distal release or excision. However, landmark studies in the late 20th century demonstrated that simple release often resulted in an unacceptable loss of hip flexion power, leading to a precipitous decline in overall gait velocity.
The seminal work by Gage and Novacheck established the rectus femoris transfer as the gold standard. Their research utilized dynamic EMG and kinematic data to prove that transferring the distal tendon posterior to the axis of knee rotation not only removed the pathological extensor moment during swing phase but also preserved the muscle's critical function as a hip flexor.
Further investigations by Ounpuu and colleagues evaluated the long-term kinematic outcomes of the procedure. They demonstrated statistically significant improvements in peak knee flexion timing and amplitude, directly correlating with reduced tripping and improved energy efficiency. Interestingly, there remains ongoing academic debate regarding the true mechanism of action. While originally conceived as an active phase-conversion transfer (where the muscle actively fires to flex the knee), contemporary dynamic MRI and fine-wire EMG studies suggest that the primary benefit may be a "dynamic tenodesis" effect. Regardless of whether the muscle actively pulls or simply acts as a passive tether in its new position, the clinical consensus remains unequivocal: the rectus femoris
