Partial Selective Fasciectomy for Dupuytren's Contracture: Surgical Masterclass
Key Takeaway
Partial selective fasciectomy remains the gold standard surgical intervention for advanced Dupuytren's contracture. This technique involves the meticulous excision of pathological fascial cords while preserving the overlying skin and neurovascular structures. Proper execution requires a thorough understanding of palmar fascial anatomy, precise surgical dissection, and rigorous postoperative rehabilitation to optimize functional outcomes, minimize flare reactions, and prevent recurrence in the affected digits.
Introduction to Partial (Selective) Fasciectomy
Partial (selective) fasciectomy is the most widely performed and arguably the most reliable surgical intervention for the management of advanced Dupuytren’s disease. Unlike radical fasciectomy, which attempts to remove all palmar fascia (both diseased and macroscopically normal) and carries an unacceptably high complication rate, partial fasciectomy targets only the macroscopically diseased fascial cords responsible for the contracture.
The primary objective of this procedure is to restore functional digital extension by excising the pathological myofibroblast-rich cords while preserving the overlying skin, the neurovascular bundles, and the uninvolved fascial structures. This approach strikes an optimal balance between minimizing surgical morbidity and maximizing long-term functional outcomes.
Pathoanatomy and Biomechanical Considerations
A profound understanding of the normal and pathological anatomy of the palmar fascia is mandatory for safe execution. Dupuytren’s disease is characterized by fibroproliferation, driven by transforming growth factor-beta (TGF-β) and the differentiation of fibroblasts into contractile myofibroblasts expressing alpha-smooth muscle actin (α-SMA).
Normal fascial bands hypertrophy and contract to form pathological cords:
* Pretendinous Cord: Arises from the pretendinous band; primarily responsible for metacarpophalangeal (MCP) joint contractures.
* Central Cord: An extension of the pretendinous cord into the digit, causing proximal interphalangeal (PIP) joint contractures.
* Spiral Cord: The most surgically treacherous structure. Formed by the amalgamation of the pretendinous band, spiral band, lateral digital sheet, and Grayson’s ligament. As it contracts, it displaces the neurovascular bundle proximally, centrally, and superficially, placing it at high risk during surgical dissection.
* Natatory Cord: Causes web space contractures and restricts digital abduction.
* Lateral Cord: Contributes to PIP and distal interphalangeal (DIP) joint contractures.
Clinical Pearl: The neurovascular bundle is most vulnerable at the level of the distal palmar crease and the proximal phalanx, particularly when a spiral cord is present. Always identify the nerve in normal tissue proximally before tracing it into the diseased area.
Preoperative Assessment and Indications
Surgical intervention is generally indicated when the patient demonstrates a positive "tabletop test" (inability to place the palm flat on a table) accompanied by functional impairment. Specific surgical thresholds include:
* MCP joint contracture ≥ 30 degrees.
* PIP joint contracture ≥ 15 to 20 degrees (PIP contractures are notoriously difficult to correct and tend to worsen rapidly).
* Severe web space contractures.
Evaluating the PIP Joint: The Tenodesis Test
Chronic PIP joint contractures of more than 60 degrees may result in secondary attenuation of the extensor mechanism's central slip.
Surgical Warning: If a tenodesis test is positive—defined as the failure of the PIP joint to extend fully when the wrist and MCP joints are placed in full passive flexion—the surgeon must suspect central slip attenuation. This dictates specific postoperative splinting protocols to prevent a secondary boutonnière deformity.
Surgical Technique: Step-by-Step
1. Patient Positioning and Preparation
The procedure is performed under regional anesthesia (brachial plexus block) or general anesthesia. The patient is positioned supine with the operative arm extended on a hand table. A well-padded pneumatic tourniquet is applied to the upper arm. Exsanguination is performed using an Esmarch bandage, and the tourniquet is inflated to 250 mm Hg (or 100 mm Hg above systolic blood pressure). Loupe magnification (2.5x to 4.5x) is essential.
2. Incision Planning
Incision design is critical to prevent postoperative scar contracture. Longitudinal incisions converted to Z-plasties are preferred, especially when concomitant digital joint contractures are present. Alternatively, multiple V-Y advancements or Bruner (zigzag) incisions can be utilized.

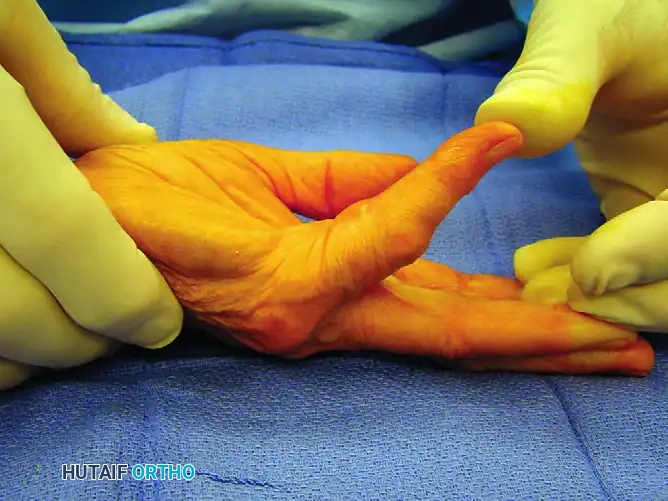
Figure 75-9 A: Preoperative presentation demonstrating a 60-degree metacarpophalangeal joint contracture.
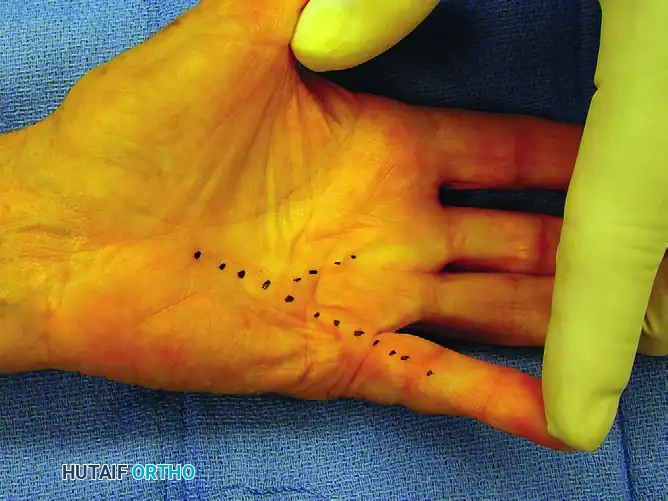
Figure 75-9 B: Dotted lines indicating the underlying pathological cords to be released at surgery.
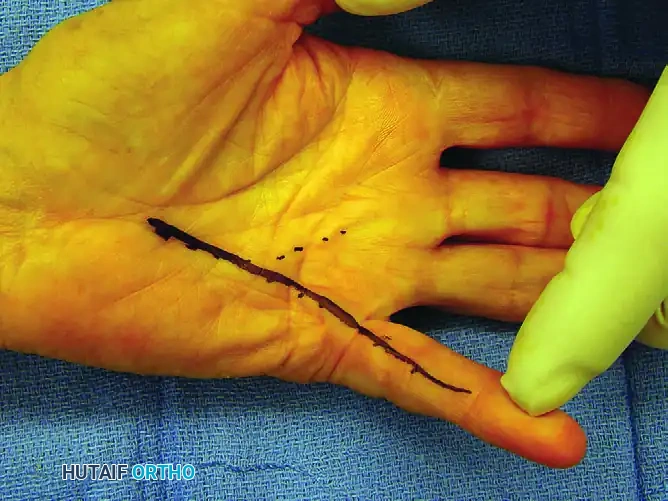
Figure 75-9 C: Solid line outlining the intended longitudinal incision.
For more complex, multi-digit involvement, incisions must be meticulously planned to allow adequate exposure of all involved cords without compromising skin flap viability.
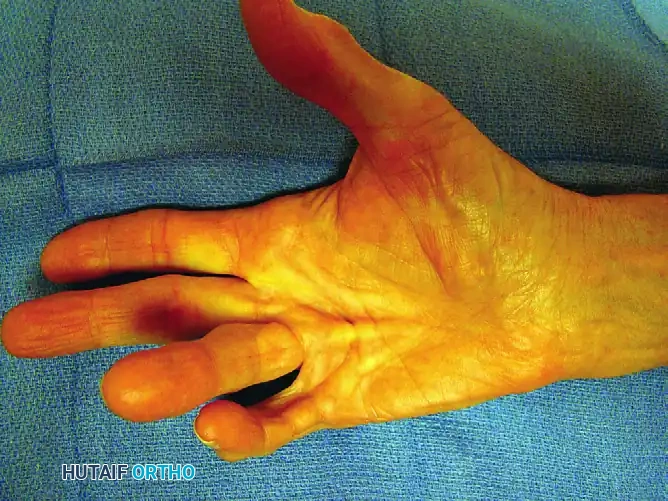
Figure 75-10 A: Complex disease involving the thumb, middle, ring, and small fingers.
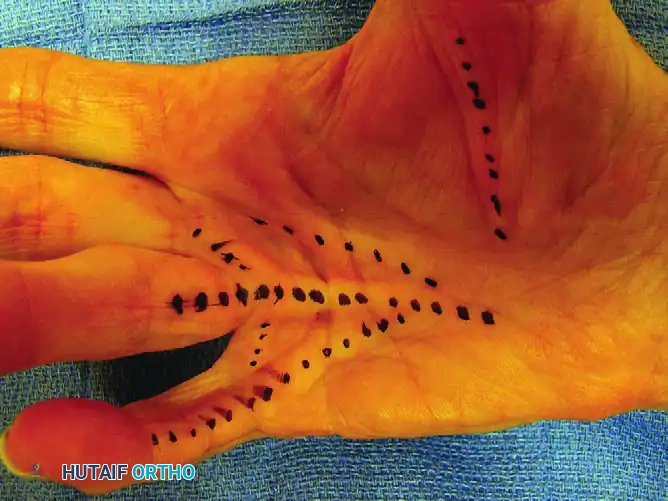
Figure 75-10 B: The cords to be released are dotted with a skin-marking pen.
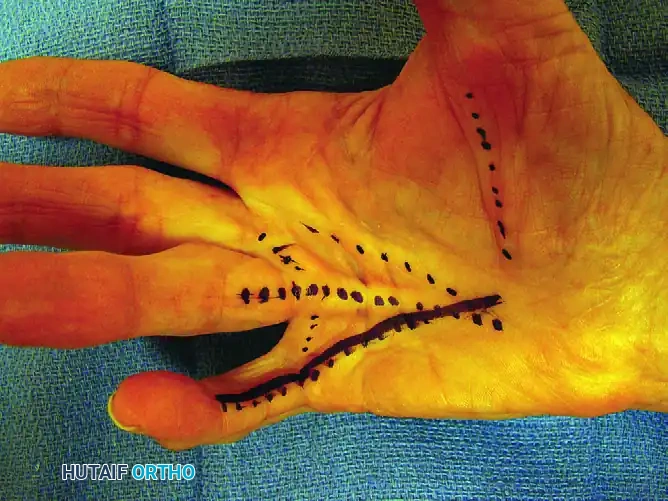
Figure 75-10 C: Planned incisions are solidly outlined before tourniquet inflation.
3. Dissection and Cord Excision
Skin flaps are elevated with meticulous care to preserve the subdermal vascular plexus. The diseased fascia is often intimately adherent to the dermis; sharp dissection with a scalpel (No. 15 blade) is required to separate the skin from the underlying cord.
Dissection should proceed from proximal to distal. The proximal end of the pretendinous cord is identified in the palm, transected, and elevated.
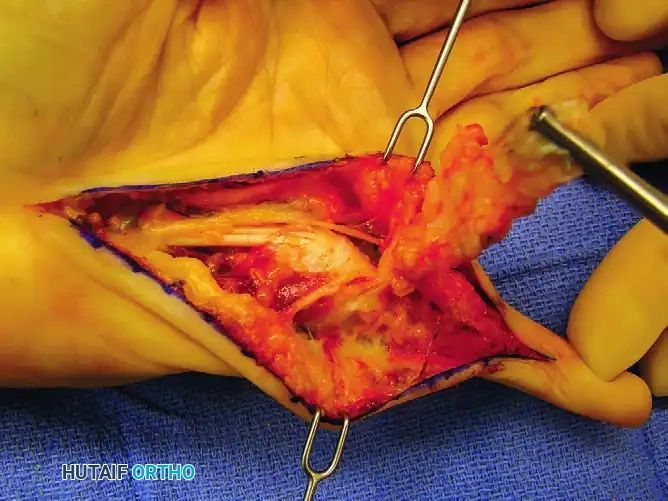
Figure 75-9 D: Disease dissection from proximal to distal, clearly exposing the digital nerves. Note the Kocher clamp on the proximal end of the pretendinous cord providing traction.
Traction on the cord helps delineate its distal extensions. The neurovascular bundles must be identified proximally in the palm, where the anatomy is relatively undisturbed, and traced distally into the digit.
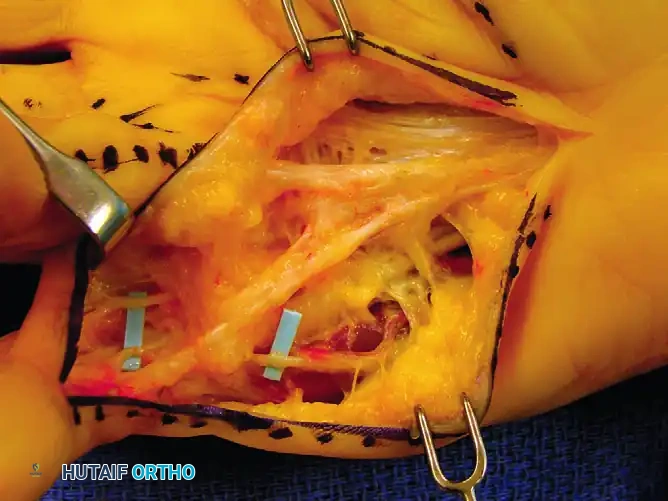
Figure 75-10 D: The palmar and digital cords are exposed through the longitudinal incision.
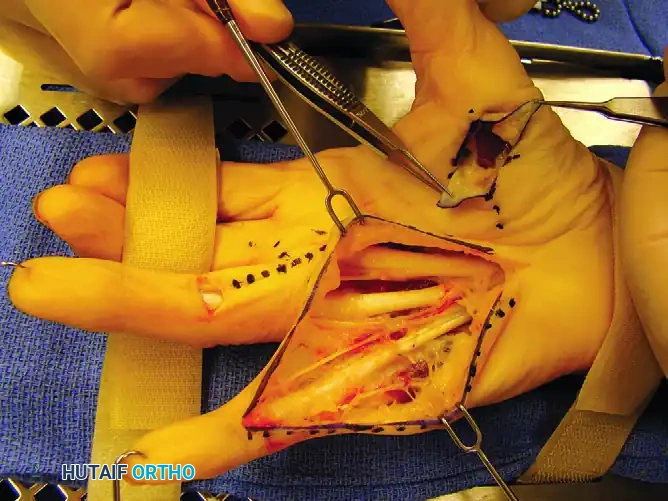
Figure 75-10 E: Exposure after cord excisions. Note the additional transverse incision required to excise the ring finger central cord and the additional thumb cord incision.
Pitfall: Never blindly divide fascial bands in the web space or the proximal phalanx without direct visualization of the neurovascular bundle. The spiral cord can displace the nerve superficially, making it highly susceptible to transection.
4. Hemostasis and Closure
Once the fasciectomy is complete and maximal extension is achieved, the tourniquet is deflated. Meticulous hemostasis is achieved using bipolar electrocautery. Hematoma formation is the most significant preventable complication and can lead to flap necrosis, infection, and severe flare reactions.
If a longitudinal incision was used, it is now converted into a series of Z-plasties to lengthen the volar skin and prevent linear scar contracture.
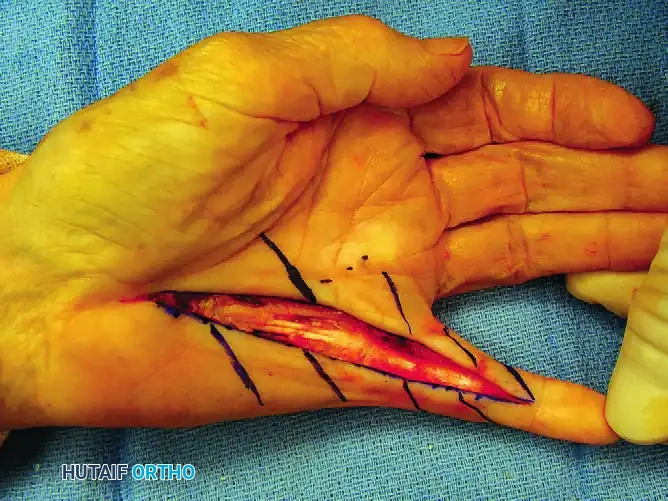
Figure 75-9 E: Planned Z-plasty skin flaps drawn over the longitudinal incision.
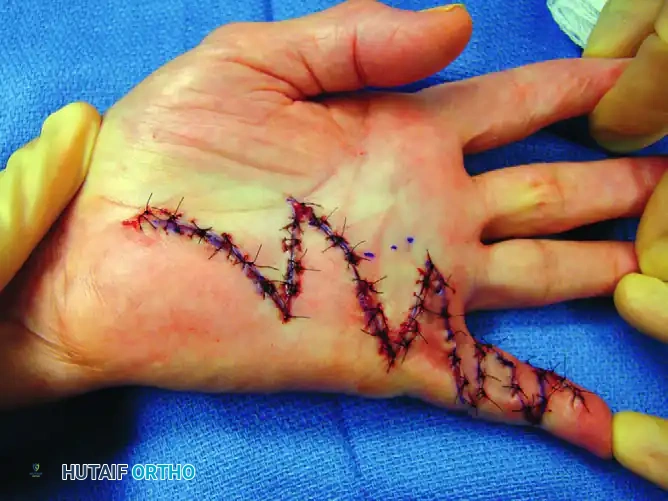
Figure 75-9 F: Subsequent closure of the Z-plasties.
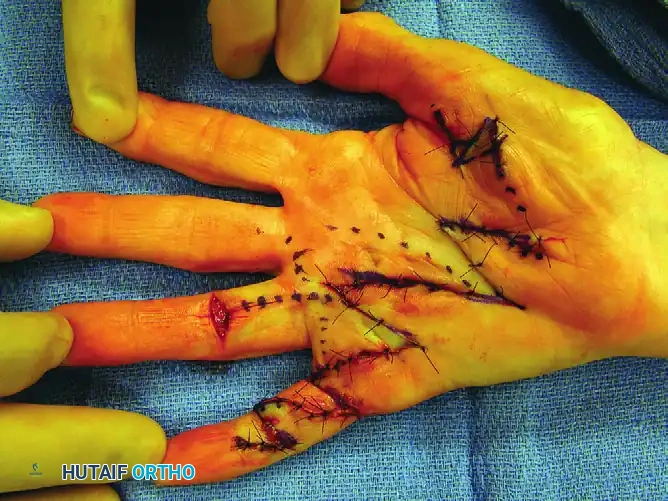
Figure 75-10 F: After meticulous hemostasis, the complex Z-plasty wounds are closed.
5. Drainage Systems and Steroid Instillation
To prevent hematoma, adequate drainage is mandatory. The surgeon may place a few loose sutures in the palm to allow necessary drainage around a simple rubber drain.
Alternatively, a highly effective closed suction drainage system can be constructed using 21-gauge sterile butterfly catheters and Vacutainer tubes. One catheter tube for each operated finger provides adequate and efficient drainage.
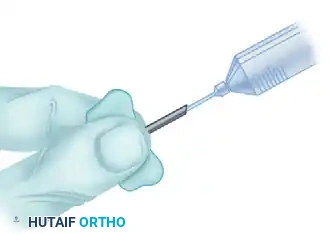
Figure 75-11 A: Multiple tubing holes are made with scissors, and the 21-gauge catheter is placed under the skin flaps.
The Steroid Pearl: The likelihood of a "flare reaction" (a hyperactive inflammatory response characterized by diffuse swelling, redness, and stiffness) occurring 4 to 6 weeks postoperatively may be significantly decreased by infusing a corticosteroid directly into the wound bed.
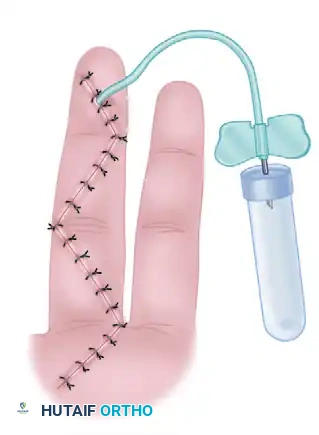
Figure 75-11 B: After wound closure, a 25-gauge needle can be used to instill 15 to 20 mg of betamethasone (Celestone) through the 21-gauge catheter needle.
This technique not only mitigates the flare reaction but also seems to decrease the amount of postoperative discomfort, substantially decreasing the need for narcotic analgesics in many patients, even after complex fasciectomies.
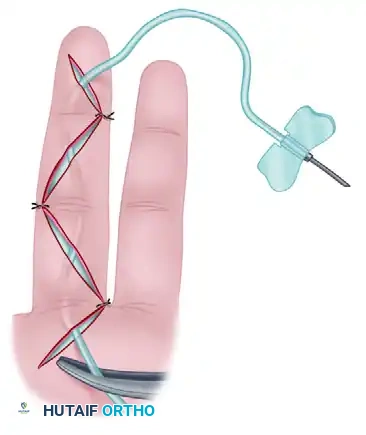
Figure 75-11 C: The Vacutainer tube is attached after the dressing is applied and the tourniquet is deflated, creating active closed suction.
Postoperative Care and Rehabilitation
The success of a partial fasciectomy relies as much on postoperative rehabilitation as it does on surgical execution.
Immediate Postoperative Phase (0-48 Hours)
- Dressing: Apply a layer of nonadherent gauze and a moist dressing compressed gently against the wound to conform to the contours of the palm and fingers. Apply a compression dressing over this.
- Splinting: Use a volar plaster splint to maintain the fingers in the exact degree of extension achieved at surgery. Do not force extension beyond what was surgically obtained, as this causes skin ischemia.
- Elevation: The hand is kept strictly elevated for a minimum of 48 hours.
- Drain Management: Drains are usually removed within 24 to 48 hours after surgery.
- Early Motion: Early proximal interphalangeal motion is encouraged within the confines of the dressing. The shoulder must be moved actively at intervals during this period to avoid cramping and secondary adhesive capsulitis.
Acute Complication Management
If there is undue pain in the hand or fever after 48 hours, the wound must be inspected immediately.
* Hematoma: If a hematoma is found elevating the skin, it must be evacuated in sterile conditions, and the involved area of the wound should be left open to heal by secondary intention. Failure to evacuate a hematoma will lead to flap necrosis and severe scarring.
Subacute Phase (3 Days to 2 Weeks)
- Dressing Change: The first dressing change is done 3 to 5 days after surgery.
- Therapy: Formal range-of-motion exercises are begun under the guidance of a certified hand therapist.
- Night Splinting: A custom-molded resting pan splint is fitted with the fingers in maximal extension. This is to be worn strictly at night.
- Suture Removal: At 2 weeks, the sutures are removed, and the hand is left free of all dressings during the day.
- Precautions: The patient is warned not to place the hand in a dependent position for rest and not to soak the hand in hot water (which promotes edema). Active exercise in warm water is permissible, but no passive stretching is allowed at this stage, as it can induce microtrauma and exacerbate inflammation.
Long-Term Rehabilitation (3 Weeks to 3 Months)
- Activity: Moderate use of the hand for activities of daily living is permitted at 3 weeks. However, patients must be counseled that several months of dedicated rehabilitation may be necessary to achieve maximal medical improvement.
- Splinting Duration: The resting pan splint is worn at night for a total of 3 months after surgery to prevent recurrent contracture during the critical phase of scar remodeling.
- Strengthening: Silicone putty may be a valuable adjunct to an exercise program to rebuild grip strength once the wounds are fully healed and stable.
Management of Central Slip Attenuation
As noted during the preoperative assessment, chronic PIP joint contractures of more than 60 degrees may result in central slip attenuation.
* If the intraoperative tenodesis test was positive, specific PIP joint splinting in extension for 3 weeks postoperatively may be indicated to allow the central slip to tighten.
* During these 3 weeks, isolated distal interphalangeal (DIP) joint exercises are performed to mobilize the lateral bands dorsally, preventing them from subluxating volarly and creating a boutonnière deformity.
Conclusion
Partial selective fasciectomy is a demanding but highly rewarding procedure for the treatment of Dupuytren's contracture. By adhering to strict anatomical principles, utilizing meticulous surgical technique to protect neurovascular structures, employing effective drainage and steroid instillation to prevent flare reactions, and enforcing a rigorous postoperative hand therapy protocol, the orthopedic surgeon can consistently achieve excellent functional outcomes and restore quality of life to the patient.
You Might Also Like

