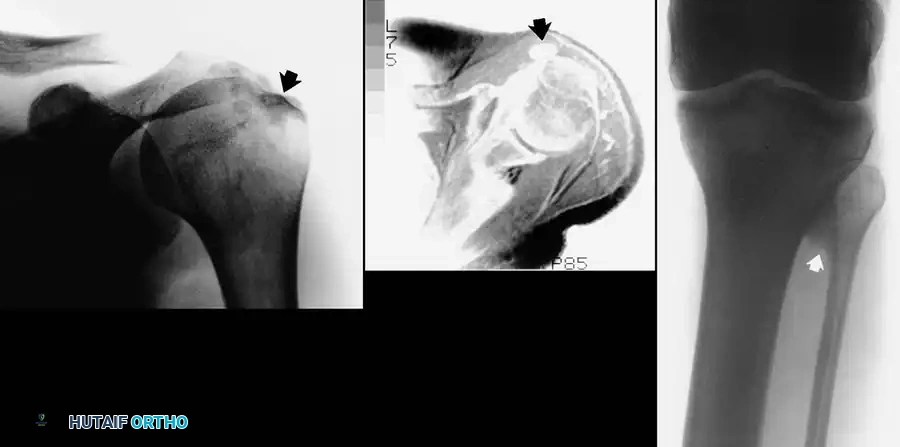
Paralytic Hand: Principles of Tendon Transfer and Surgical Reconstruction
Key Takeaway
The surgical rehabilitation of the paralytic hand requires a profound understanding of hand biomechanics, precise timing, and meticulous tendon transfer techniques. This guide details the evaluation of muscle imbalance, correction of claw deformities, and evidence-based protocols for restoring pinch, opposition, and intrinsic function following peripheral nerve injuries, tetraplegia, or systemic neuromuscular diseases.
Pathophysiology of the Paralytic Hand
The paralytic hand represents a complex reconstructive challenge characterized by dynamic muscle imbalance, loss of motor function, and often, profound sensory deficits. Whether the loss of function is caused by systemic disease, spinal cord injury, or peripheral nerve trauma, the resulting dynamic muscle imbalance predictably leads to debilitating deformities. When normal antagonistic muscle tension is lost, the unopposed functioning muscles gradually drive the hand into fixed, non-functional postures.
Sensation in the paralytic hand varies significantly according to the underlying etiology, dictating both the surgical approach and the ultimate functional prognosis:
* Poliomyelitis: Motor paralysis is present, but sensation remains entirely normal.
* Peripheral Nerve Lesions: Sensory deficits follow specific dermatomal or autonomous nerve distributions depending on the level and severity of the nerve lesion.
* Hansen Disease (Leprosy): Sensation is typically absent in a progressive, glove-like distribution.
* Syringomyelia: Patients exhibit a dissociated sensory loss (loss of pain and temperature sensation with preserved fine touch and proprioception), which can complicate postoperative motor re-education.
Furthermore, upper motor neuron lesions may introduce muscle spasticity. Spasticity disrupts hand balance because the tension of spastic muscles cannot be effectively controlled or balanced by opposing normal muscles. Tendon transfers in the spastic hand require entirely different biomechanical considerations compared to the flaccid paralytic hand.
Biomechanics of the Paralytic Hand and Claw Deformity
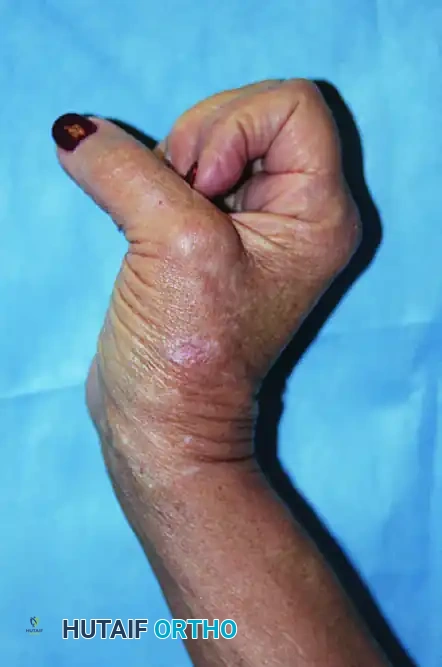
The most iconic presentation of intrinsic muscle paralysis is the "claw hand" (intrinsic-minus posture). This deformity is a direct result of the disruption of the delicate balance between the extrinsic and intrinsic musculature of the hand.

In a normal hand, the intrinsic muscles (lumbricals and interossei) flex the metacarpophalangeal (MCP) joints and extend the proximal and distal interphalangeal (PIP and DIP) joints. When these muscles are paralyzed, the extrinsic extensor digitorum communis (EDC) acts unopposed at the MCP joint, driving it into hyperextension.
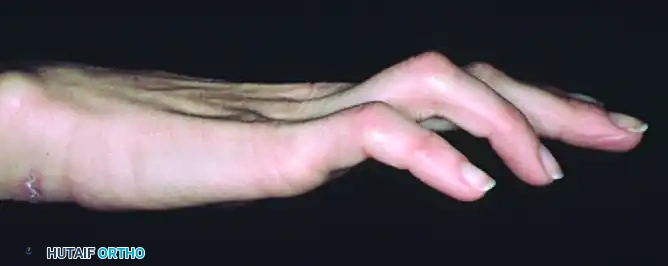
Concurrently, the extrinsic flexors (flexor digitorum superficialis [FDS] and flexor digitorum profundus [FDP]) act unopposed at the IP joints, pulling them into acute flexion.
Clinical Pearl: The Bouvier Principle
The long finger extensors are mechanically capable of extending the interphalangeal joints only if the hyperextension of the metacarpophalangeal joints is prevented. If the MCP joint is allowed to hyperextend, the EDC tendon bowstrings dorsally, losing its mechanical advantage to extend the PIP and DIP joints.
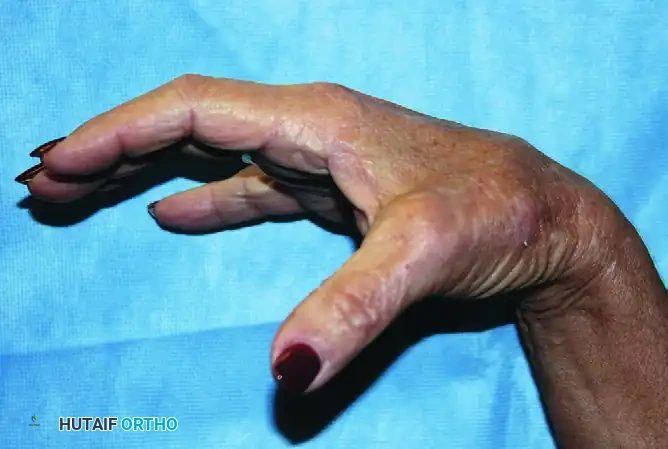
This biomechanical phenomenon is the foundation of the Bouvier test. When the examiner manually blocks the MCP joints from hyperextending (simulating intrinsic muscle function), the patient's extrinsic extensors can successfully extend the IP joints. If IP extension is achieved, a simple MCP joint stabilization procedure (such as a Zancolli lasso or bone-block operation) will correct the clawing. If IP extension is not achieved (a negative Bouvier test), the extensor apparatus is attenuated, and a dynamic tendon transfer to the lateral bands is required.
Fundamental Principles of Tendon Transfer
Tendon transfer surgery is the final, definitive step in the rehabilitation of the paralytic hand. To achieve predictable, functional outcomes, the surgeon must adhere strictly to the established biomechanical principles of tendon transfer.
1. Restoration of Tissue Equilibrium
A transferred tendon must glide freely to function. It cannot be routed through dense scar tissue or placed directly over raw bone, as it will inevitably adhere, rendering the transfer useless. If the soft tissue bed is compromised by trauma or previous surgery, scar tissue must be excised and replaced with healthy, vascularized tissue (such as a pedicled or free fasciocutaneous flap) prior to, or concurrent with, the tendon transfer. The tendon must be surrounded by healthy subcutaneous fat to ensure unhindered excursion.
2. Supple Joints and Passive Range of Motion
A tendon transfer provides dynamic motor power; it cannot overcome fixed joint contractures. A satisfactory range of passive joint motion is an absolute prerequisite. The passive range of motion of the target joint must equal or exceed the desired active range of motion post-transfer. If joint stiffness or contracture is present, it must be corrected via serial splinting, therapy, or surgical ligamentous release before the tendon transfer is performed.
Surgical Warning:
Attempting a tendon transfer across a stiff joint is a recipe for failure. The transferred tendon will be unable to move the joint during the critical early postoperative phase, leading to permanent adherence of the tendon to surrounding tissues.
3. Skeletal Stability
Malalignment of bone must be corrected by osteotomy, and any necessary bone grafting or arthrodesis must be fully healed before a tendon transfer is undertaken. A stable skeletal foundation is required for the transferred muscle to exert its force effectively across the desired joints.
4. Donor Muscle Selection
- Strength: A muscle loses approximately one grade of strength (on the MRC 0-5 scale) following transfer. Therefore, only muscles with a preoperative strength of Grade 4 or 5 should be selected as donors.
- Excursion (Amplitude): The excursion of the donor tendon must closely match the physiological requirement of the paralyzed muscle. Wrist flexors/extensors have an excursion of ~33 mm, finger extensors ~50 mm, and finger flexors ~70 mm. A wrist flexor cannot fully substitute for a finger flexor without utilizing the tenodesis effect of the wrist.
- Synergy: Muscles that normally work together (e.g., wrist extensors and finger flexors) are easier for the patient to consciously control postoperatively. Synergistic transfers require less cortical re-education.
Timing of Tendon Transfer
The timing of surgical intervention is critical and varies fundamentally based on the etiology of the paralysis. Tendon transfers should only be performed when the neurological deficit is deemed permanent, or when early transfer is utilized as an "internal splint."
Poliomyelitis
In poliomyelitis, spontaneous recovery of muscle power can occur for up to 18 months following the acute stage of the disease. Consequently, this entire period must elapse before an accurate evaluation of residual muscle strength is possible. Any further recovery beyond 18 months cannot be expected to improve muscle strength by more than one grade. During this waiting period, the hand must be meticulously splinted to prevent fixed contractures and to maximize the function of available musculature.
Congenital Anomalies
In congenital paralytic conditions, the relative muscle strength does not change over time. Tendon transfers can be planned once the child is old enough to cooperate with postoperative rehabilitation, typically between 3 and 5 years of age.
Syringomyelia and Progressive Neuropathies
In conditions like syringomyelia or Charcot-Marie-Tooth disease, muscle weakness is progressive. Tendon transfers must be approached with caution, as the donor muscle itself may eventually succumb to the disease process, leading to late failure of the transfer.
Peripheral Nerve Injuries
Peripheral nerve injuries require individualized timelines based on the level of the injury, the distance to the motor endplates, and the status of nerve repair (neurorrhaphy).
* Radial Nerve Palsy: In a division of the radial nerve at the midhumerus, definitive transfers for finger and thumb extension should generally be delayed for 6 months or longer after neurorrhaphy to allow for nerve regeneration. However, early transfer of the pronator teres (PT) to the extensor carpi radialis brevis (ECRB) is highly recommended. This early transfer acts as an internal splint, immediately restoring wrist extension, improving grip strength, and eliminating the need for cumbersome external orthoses while awaiting radial nerve regeneration.
* Median Nerve Palsy: In high median nerve lesions, clinical signs of reinnervation in the most proximal muscles (e.g., pronator teres) should be evident within 4 months. In low median nerve lesions (at the wrist), reinnervation of the thenar muscles should begin within 3 months. If no clinical or electromyographic (EMG) evidence of recovery is present within these timeframes, the nerve should be surgically explored, or tendon transfers for thumb opposition should be executed.
Technical Considerations for Tendon Transfer
Surgical Approach and Tendon Routing
Incisions must be planned to avoid crossing the path of the transferred tendon at a right angle, which can cause bowstringing or restrictive adhesions. The optimal route for a transferred tendon is through the subcutaneous fat layer. Routing tendons through fascial windows or interosseous membranes increases the risk of adhesions and should be done with a wide fascial excision to prevent tethering.
The Pulvertaft Weave
The gold standard for tendon coaptation in transfers is the Pulvertaft weave. This technique involves weaving the donor tendon through the recipient tendon at 90-degree angles for a minimum of three passes.
1. The recipient tendon is split longitudinally.
2. The donor tendon is passed through the split.
3. The process is repeated 2 to 3 times, alternating the axis of the split by 90 degrees.
4. Each pass is secured with a mattress suture (typically 3-0 or 4-0 non-absorbable braided suture).
This weave provides immense immediate biomechanical strength, allowing for early active mobilization protocols without the risk of rupture.
Tensioning the Transfer
Setting the correct tension is arguably the most difficult and critical step of the operation. The muscle must be tensioned at its optimal resting length to maximize the number of actin-myosin cross-bridges (the peak of the Blix curve).
* If tensioned too loosely, the muscle will expend its entire excursion simply taking up the slack, resulting in no joint movement.
* If tensioned too tightly, the muscle will undergo ischemic necrosis or permanent stretch-induced weakness, and the antagonistic joint motion will be restricted.
As a general rule, the transfer is tensioned such that the resting posture of the hand mimics the normal physiological cascade when the wrist is in neutral.
Postoperative Rehabilitation Protocol
The success of a tendon transfer relies as much on the postoperative rehabilitation as it does on the surgical execution.
Phase I: Immobilization (0 to 3-4 Weeks)
Immediately postoperatively, the limb is immobilized in a rigid cast or custom orthosis. The joints are positioned to remove all tension from the tendon anastomosis. For example, following a radial nerve tendon transfer, the wrist is immobilized in 40 degrees of extension, the MCP joints in neutral, and the thumb in radial abduction and extension.
Phase II: Early Active Mobilization (3-4 to 6 Weeks)
At 3 to 4 weeks, the cast is removed, and a removable protective splint is fabricated. The patient begins supervised, gravity-eliminated active range of motion.
* Motor Re-education: The patient is instructed to perform the original action of the donor muscle to achieve the new action. For instance, if the Pronator Teres was transferred to the ECRB, the patient is asked to forcefully pronate the forearm, which will now result in wrist extension. Biofeedback and mirror therapy are highly effective during this phase.
* Passive stretching of the transfer is strictly prohibited during this phase to prevent elongation of the healing weave.
Phase III: Strengthening and Integration (6 to 12 Weeks)
By 6 weeks, the tendon healing is robust enough to withstand gentle passive stretching to overcome any mild contractures. Progressive resistance exercises are initiated. The protective splint is gradually weaned during the day but may be worn at night until 8 to 10 weeks postoperatively. Full integration of the transfer into subconscious, spontaneous hand use may take 6 to 12 months.
📚 Medical References
- paralytic hand, J Bone Joint Surg 57A:730, 1975.
- Brown PW: Zancolli capsulorrhaphy for ulnar claw hand: appraisal of forty-four cases, J Bone Joint Surg 52A:868, 1970.
- Brown PW: Reconstruction for pinch in ulnar intrinsic palsy, Orthop Clin North Am 2:323, 1974.
- Bruner JM: Tendon transfer to restore abduction of the
You Might Also Like