Clinical Case Study: Distal Femoral Osteosarcoma Diagnosis & Management
Key Takeaway
Diagnosing adolescent distal femoral osteosarcoma involves evaluating progressive bone pain, followed by comprehensive imaging. Radiographs show aggressive lytic/sclerotic lesions; MRI details local tumor extent and neurovascular involvement. CT chest screens for metastases. A CT-guided core needle biopsy provides the definitive histopathological diagnosis, essential for tailoring effective orthopedic oncology treatment.
Patient Presentation and History
A 16-year-old male presented to the orthopedic oncology clinic with a chief complaint of progressive right distal thigh and knee pain for approximately 4 months. Initially, the pain was intermittent and activity-related, attributed by the patient to sports participation (basketball). However, over the past 6 weeks, the pain had become constant, insidious in onset, significantly worse at night, and was poorly controlled with over-the-counter analgesics, including NSAIDs. He reported waking frequently due to pain and experiencing an increasing limp, particularly with weight-bearing activities.
He denied any specific traumatic event leading to the onset of symptoms, recent fevers, chills, or night sweats. There was no reported history of weight loss. His past medical history was unremarkable, and he was otherwise healthy and active. Family history was non-contributory for malignancy, Li-Fraumeni syndrome, hereditary retinoblastoma, or other genetic syndromes associated with sarcomagenesis. He was a non-smoker and denied recreational drug use.
The insidious nature of this presentation is a hallmark of primary malignant bone tumors in the adolescent population. The initial misattribution to a sports-related injury or "growing pains" frequently delays diagnosis. The transition from mechanical, activity-related pain to non-mechanical, nocturnal rest pain indicates progressive biological activity, intraosseous hypertension, and cortical micro-fracturing secondary to tumor expansion.
Clinical Examination Findings
On general inspection, the patient appeared thin but in no acute distress at rest. An antalgic gait was noted upon ambulation, characterized by a shortened stance phase on the right lower extremity to minimize joint reactive forces across the distal femur.
Local examination of the right lower extremity revealed subtle but noticeable swelling in the distal thigh region, specifically around the distal femur. There was no overt erythema or ecchymosis. Prominent superficial venous engorgement was observed over the anteromedial aspect of the distal thigh, indicative of tumor-induced angiogenesis and increased regional blood flow driven by vascular endothelial growth factor (VEGF) expression.
Palpation revealed a firm, fixed, and exquisitely tender mass palpable over the anterior and medial aspects of the distal right femur, approximately 5 cm proximal to the joint line. The mass measured roughly 8 x 6 cm. Localized warmth was present over the lesion. There was no regional lymphadenopathy in the popliteal or inguinal basins, which is consistent with the typical hematogenous, rather than lymphatic, dissemination pattern of osteosarcoma.
Range of motion of the right knee was limited by pain, particularly at the extremes of flexion (0-90 degrees, compared to 0-140 degrees on the contralateral side) and extension (lacked 5 degrees extension). Patellar tracking was grossly normal, though the suprapatellar pouch felt full, suggesting a sympathetic effusion. Ligamentous stability testing (Lachman, anterior/posterior drawer, varus/valgus stress) was deferred due to significant pain and concern for structural compromise of the distal femoral metaphysis, which could precipitate a pathologic fracture under torsional or bending loads.
Neurological examination of the right lower extremity revealed intact motor strength (5/5) in all major muscle groups distal to the knee, including ankle dorsiflexion, plantarflexion, toe extension, and flexion. Sensation was intact to light touch in all dermatomes (L2-S1). Deep tendon reflexes (patellar and Achilles) were 2+ bilaterally and symmetric.
Vascular assessment demonstrated palpable pedal pulses (dorsalis pedis and posterior tibial) bilaterally, with good capillary refill in the toes of the affected limb. No bruits were audible over the mass or adjacent major vessels. The popliteal artery was palpable and symmetric to the contralateral side. A comprehensive examination of the ipsilateral hip and contralateral lower extremity was unremarkable.
Imaging and Diagnostics
Initial diagnostic workup commenced with plain radiographs of the right knee and femur. Radiographic findings of the distal right femur demonstrated an aggressive, mixed lytic and sclerotic lesion centered in the metaphysis. Features included significant cortical destruction, spiculated periosteal reaction ("sunburst" pattern), and an ill-defined Codman's triangle along the anteromedial cortex. A large associated soft tissue mass was also evident, extending beyond the bone confines. There was no evidence of a pathologic fracture at this stage.
Following the plain radiographs, further advanced imaging was immediately pursued. A Magnetic Resonance Imaging scan of the entire right femur with contrast was performed for detailed local staging. The MRI confirmed a large, heterogeneous mass arising from the distal femoral metaphysis, measuring approximately 8.5 x 6.2 x 9.0 cm (AP x ML x CC). The lesion demonstrated aggressive marrow replacement, cortical breakthrough, and a substantial extraosseous soft tissue component.

Local Staging and MRI Protocol
The MRI protocol included T1-weighted, T2-weighted, Short Tau Inversion Recovery (STIR), and gadolinium-enhanced sequences. The T1-weighted coronal and sagittal images were critical for defining the proximal extent of marrow involvement, which dictates the level of the surgical osteotomy. The tumor exhibited low signal intensity on T1 and heterogeneous high signal intensity on T2/STIR sequences, reflecting areas of cellularity, necrosis, and chondroblastic or osteoblastic matrix production.

Crucially, the MRI evaluated the relationship of the extraosseous mass to the neurovascular bundle. The soft tissue component displaced the vastus medialis anteriorly and abutted the adductor hiatus, but a distinct fat plane was maintained between the tumor pseudocapsule and the superficial femoral artery/vein, suggesting that a limb-salvage resection with negative margins was anatomically feasible. Whole-bone imaging of the femur ruled out the presence of "skip" metastases within the same medullary cavity.
Systemic Staging
Systemic staging was initiated to evaluate for distant metastasis, which is present in 15-20% of patients at initial diagnosis. High-resolution computed tomography (HRCT) of the chest was performed to assess for pulmonary metastases, the most common site of dissemination. A Positron Emission Tomography combined with Computed Tomography (PET-CT) using 18F-FDG was utilized to detect osseous metastases and evaluate the metabolic activity of the primary lesion.
Laboratory investigations revealed an elevated serum alkaline phosphatase (ALP) and lactate dehydrogenase (LDH). Elevated levels of these enzymes correlate with tumor burden and osteoblastic activity, serving as independent prognostic indicators for decreased progression-free survival and overall survival.
Diagnostic Biopsy Principles
Following completion of all staging imaging, a core needle biopsy was performed under fluoroscopic and ultrasound guidance. The biopsy tract was meticulously planned by the operating orthopedic oncologist to ensure it passed through the involved compartment (anterior compartment via the vastus intermedius) and could be excised en bloc with the surgical specimen during definitive resection. Trans-compartmental contamination or biopsy tracts placed in the path of future skin flaps represent catastrophic errors that can mandate amputation in an otherwise salvageable limb.
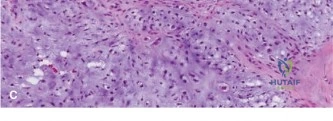
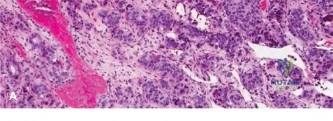
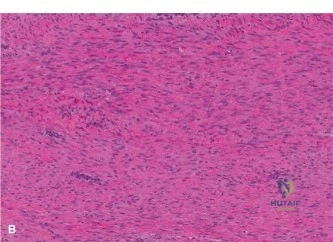
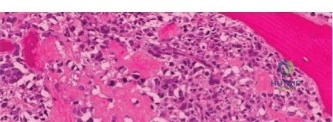
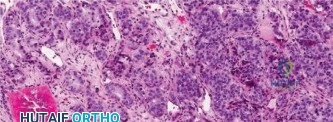
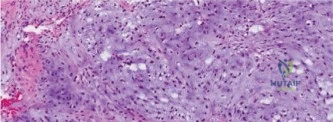
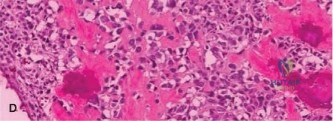
Histopathological analysis of the core samples revealed highly pleomorphic spindle cells producing disorganized, lace-like osteoid matrix. Frequent atypical mitotic figures, areas of coagulative necrosis, and hyperchromatic nuclei were observed. Immunohistochemistry was positive for SATB2 and negative for cytokeratins and S100, confirming the diagnosis of high-grade conventional osteosarcoma (osteoblastic subtype).
Differential Diagnosis
The clinical and radiographic presentation of an aggressive, bone-destructive lesion in the metaphysis of an adolescent necessitates a focused differential diagnosis. While the classic "sunburst" periosteal reaction and osteoid matrix production strongly suggest osteosarcoma, other malignant, benign, and infectious etiologies must be considered until histological confirmation is obtained.
| Pathology | Clinical Presentation | Radiographic Features | Histological Hallmarks |
|---|---|---|---|
| Conventional Osteosarcoma | Adolescent/young adult. Insidious pain, worse at night. Palpable mass. | Metaphyseal. Mixed lytic/sclerotic. Cortical destruction. Codman's triangle, sunburst periosteal reaction. | Malignant spindle cells producing disorganized, lace-like osteoid matrix. High mitotic rate. |
| Ewing Sarcoma | Child/adolescent. Pain, swelling. May present with systemic symptoms (fever, elevated ESR/CRP). | Diaphyseal or metadiaphyseal. Permeative lytic lesion. "Onion-skin" (lamellated) periosteal reaction. | Small round blue cells. CD99 positive. t(11;22) translocation (EWSR1-FLI1 fusion). |
| Osteomyelitis | Acute or subacute onset. Fevers, chills, localized erythema, severe tenderness. | Metaphyseal. Lytic destruction, surrounding sclerosis. Periosteal reaction (usually solid or lamellated, not spiculated). | Acute/chronic inflammatory cells (neutrophils, lymphocytes). Reactive woven bone. Positive cultures. |
| Aneurysmal Bone Cyst | Adolescent. Pain, swelling. Often presents with a pathologic fracture. | Metaphyseal. Expansile, eccentric, purely lytic lesion. "Soap bubble" appearance. Thin cortical shell. | Blood-filled cystic spaces lacking endothelial lining. Fibrous septa with multinucleated giant cells. |
| Chondrosarcoma | Older adults (rare in adolescents unless secondary to osteochondroma). Dull, aching pain. | Metaphyseal/diaphyseal. Lytic lesion with intralesional "popcorn" or "ring and arc" calcifications. | Atypical chondrocytes in hyaline cartilage matrix. Myxoid changes. Permeation of trabecular bone. |
Surgical Decision Making and Classification
The patient's tumor was classified according to the Musculoskeletal Tumor Society (MSTS / Enneking) staging system as Stage IIB: a high-grade, extracompartmental malignancy without regional or distant metastasis. According to the American Joint Committee on Cancer (AJCC) 8th Edition, the tumor was Stage IIA (T1, N0, M0, High Grade).
Multidisciplinary Tumor Board Review
The case was presented at the multidisciplinary sarcoma tumor board, comprising orthopedic oncology, medical oncology, musculoskeletal radiology, and bone pathology. The consensus recommendation was to initiate neoadjuvant chemotherapy, followed by definitive surgical resection (limb salvage) and adjuvant chemotherapy.
Neoadjuvant Chemotherapy Rationale
The standard of care for high-grade conventional osteosarcoma involves the MAP protocol: Methotrexate (high-dose with leucovorin rescue), Doxorubicin (Adriamycin), and Cisplatin. The rationale for neoadjuvant chemotherapy is multifaceted:
1. Eradication of Micrometastases: Osteosarcoma is considered a systemic disease at presentation. Early systemic therapy targets subclinical pulmonary micrometastases.
2. Tumor Cytoreduction: Shrinking the primary tumor and its extraosseous soft tissue component facilitates a safer limb-salvage procedure with wider margins and reduces the risk of intraoperative tumor spillage.
3. Assessment of Histologic Response: The degree of tumor necrosis observed in the resected specimen (Huvos grading) is the single most powerful prognostic indicator. Greater than 90% necrosis (Huvos Grade III/IV) correlates with significantly improved long-term survival.
Surgical Planning and Limb Salvage Criteria
Following 10 weeks of neoadjuvant MAP chemotherapy, restaging MRI demonstrated a reduction in the soft tissue mass and increased peripheral sclerosis, indicating a positive biological response. The neurovascular bundle remained free of tumor involvement.
The criteria for limb salvage were met:
* Ability to achieve wide surgical margins.
* Preservation of a functional neurovascular bundle.
* Adequate soft tissue coverage.
* The reconstructed limb would provide better function than an amputation.
The selected reconstruction method was a modular distal femoral replacement (megaprosthesis). Alternative options, such as an osteoarticular allograft or an allograft-prosthetic composite, were rejected due to the high risk of non-union, fracture, and infection in a patient undergoing aggressive adjuvant chemotherapy. Rotationplasty was discussed with the patient and family as a durable alternative, particularly given his young age and athletic desires, but they strongly preferred endoprosthetic limb salvage.
Pre-operative templating utilized orthogonal radiographs and MRI to determine the exact level of the femoral osteotomy. Oncologic principles dictate a minimum marrow margin of 3 cm proximal to the MRI-defined abnormality. The implant size, stem diameter, and hinge mechanism were selected to restore the joint line, mechanical axis, and leg length.
Surgical Technique and Intervention
The patient was positioned supine on a radiolucent operating table. A sterile tourniquet was applied to the proximal right thigh but not inflated, reserving it for catastrophic hemorrhage control. The entire right lower extremity and ipsilateral iliac crest were prepped and draped in a standard sterile fashion.
Surgical Approach and Biopsy Excision
An extensile anteromedial approach to the distal femur was utilized. The skin incision incorporated the prior biopsy tract, utilizing an elliptical incision to excise the tract en bloc with a 1 cm margin of healthy skin and subcutaneous tissue. The incision extended from the mid-thigh, curving medially around the patella, to the tibial tubercle.
Thick fasciocutaneous flaps were developed. The quadriceps muscle was inspected. The vastus medialis, which was displaced by the tumor pseudocapsule, was sacrificed en bloc with the specimen to ensure a wide soft tissue margin. The rectus femoris and vastus lateralis were mobilized and preserved.
Neurovascular Dissection
The superficial femoral artery and vein were identified at the apex of the femoral triangle and traced distally into the adductor canal. The adductor magnus tendon was released from the adductor tubercle, allowing access to the popliteal space. The popliteal vessels were meticulously dissected away from the posterior aspect of the tumor pseudocapsule. Due to the neoadjuvant chemotherapy effect, a distinct fibrotic plane was present, allowing safe retraction of the vessels laterally. The tibial and common peroneal nerves were identified and protected.
Tumor Resection and Osteotomy
The joint capsule was incised medially and laterally. The anterior cruciate ligament, posterior cruciate ligament, medial collateral ligament, and lateral collateral ligament were transected. The menisci were excised. The patella was everted, and the articular surface was inspected; there was no evidence of intra-articular tumor extension.
Based on pre-operative MRI measurements, the femoral osteotomy site was marked 4 cm proximal to the proximal-most extent of marrow edema. The femur was circumferentially cleared of soft tissue at this level, taking care to remain strictly in the subperiosteal plane only at the exact site of the cut. A transverse osteotomy was performed using an oscillating saw.

The distal femur, containing the tumor, the excised vastus medialis, and the biopsy tract, was delivered en bloc from the surgical field. The specimen was immediately sent to pathology for margin assessment and subsequent necrosis mapping. Frozen sections of the femoral marrow margin returned negative for malignancy.
Endoprosthetic Reconstruction
Attention was turned to the reconstruction using a modular rotating-hinge distal femoral replacement. The proximal tibia was prepared using standard total knee arthroplasty instrumentation. The tibial plateau was resected perpendicular to the mechanical axis, and the medullary canal was broached. A cemented tibial baseplate and stem were implanted using antibiotic-loaded polymethylmethacrylate (PMMA) bone cement.
The femoral medullary canal was sequentially reamed. Given the patient's young age and excellent bone stock at the diaphysis, a press-fit, hydroxyapatite-coated, fluted, tapered titanium stem was selected to encourage osteointegration and provide long-term biologic fixation, avoiding the stress shielding associated with massive cemented stems.
The modular distal femoral segment was assembled to match the exact length of the resected bone, ensuring restoration of the native joint line and leg length equality. The femoral component was impacted into the canal. The rotating hinge mechanism, which limits stress transfer to the bone-implant interface by allowing rotational freedom, was assembled, connecting the femoral and tibial components.
Soft Tissue Coverage and Closure
Meticulous hemostasis was achieved. The patellar tracking was assessed; due to the sacrifice of the vastus medialis, a lateral retinacular release was performed to centralize the patella and prevent lateral subluxation.
To prevent prosthetic joint infection and provide a robust soft tissue envelope over the metallic endoprosthesis, a medial gastrocnemius rotational flap was harvested. The medial head of the gastrocnemius was detached from its femoral origin, mobilized on its sural vascular pedicle, and rotated anteriorly to cover the medial aspect of the prosthesis and the extensor mechanism repair.
The extensor mechanism was reconstructed by suturing the remaining quadriceps tendon to the proximal aspect of the endoprosthesis using heavy non-absorbable sutures passed through designated holes in the implant. The wound was closed in multiple layers over two deep subfascial closed-suction drains. The skin was approximated using surgical staples. A sterile dressing and a hinged knee brace locked in full extension were applied.
Post Operative Protocol and Rehabilitation
The patient was transferred to the surgical intensive care unit for overnight monitoring due to the extensive nature of the resection and potential for fluid shifts.
Inpatient Management
Intravenous antibiotics (Cefazolin) were administered for 24 hours post-operatively. Deep vein thrombosis (DVT) prophylaxis was initiated on post-operative day 1 using low-molecular-weight heparin (Enoxaparin) and mechanical sequential compression devices. Pain control was managed via a multimodal approach, including a continuous peripheral nerve block (adductor canal catheter), scheduled acetaminophen, gabapentin, and PRN intravenous opioids.
The closed-suction drains were removed on post-operative day 3 when output decreased to less than 30 cc per 8-hour shift.
Rehabilitation Phases
Phase I (Weeks 0-6): Protection and Early Mobilization
* Weight Bearing: Touch-down weight-bearing (TTWB) on the right lower extremity with crutches to protect the press-fit femoral stem during initial osteointegration.
* Range of Motion: The hinged knee brace remained locked in extension during ambulation. Passive and active-assisted range of motion exercises were initiated under the supervision of physical therapy, restricted to 0-60 degrees of flexion to protect the extensor mechanism repair and the gastrocnemius flap.
* Strengthening: Isometric quadriceps sets, gluteal sets, and ankle pumps were encouraged.
Phase II (Weeks 6-12): Progressive Loading
* Weight Bearing: Progression to partial weight-bearing (PWB) and eventually weight-bearing as tolerated (WBAT) based on radiographic evidence of stem stability.
* Range of Motion: Gradual progression of flexion past 90 degrees.
* Strengthening: Closed kinetic chain exercises, gentle resistance training.
Adjuvant Chemotherapy and Surveillance
Wound healing is a critical prerequisite for the resumption of systemic therapy. The surgical staples were removed at 3 weeks post-operatively once the incision was completely healed. Adjuvant chemotherapy (completion of the MAP protocol) was reinitiated at week 4 post-operatively.
Final histopathology of the resected specimen confirmed clear surgical margins (R0 resection) and demonstrated 95% tumor necrosis (Huvos Grade III), indicating an excellent response to neoadjuvant chemotherapy.
Long-term oncologic surveillance includes physical examination, plain radiographs of the reconstructed femur, and non-contrast CT of the chest every 3 months for the first 2 years, every 6 months for years 3-5, and annually thereafter to monitor for local recurrence or pulmonary metastasis.
Clinical Pearls and Pitfalls
- Biopsy Tract Placement is Critical: The biopsy tract must be considered contaminated with tumor cells. It must be placed in line with the planned surgical incision so it can be excised en bloc. Transverse incisions or trans-compartmental biopsies are oncologic disasters.
- Beware of "Growing Pains": Unrelenting, non-mechanical night pain in an adolescent is a red flag for malignancy. Plain radiographs of the entire symptomatic bone should be the first-line investigation.
- Margin Assessment: The goal of limb salvage is an R0 resection (negative microscopic margins). A positive margin drastically increases the risk of local recurrence and negatively impacts overall survival. If a wide margin cannot be achieved without sacrificing critical neurovascular structures, amputation is the oncologically sound choice.
- Extensor Mechanism Management: Sacrificing the vastus medialis often leads to patellar maltracking. Utilizing a medial gastrocnemius flap not only provides excellent soft tissue coverage over the prosthesis, reducing infection risk, but also aids in centralizing the extensor mechanism.
- Infection Risk in Megaprostheses: The risk of periprosthetic joint infection in oncologic reconstructions is significantly higher than in primary arthroplasty (up to 10-15%) due to extensive soft tissue stripping, long operative times, and the immunosuppressive effects of chemotherapy. Meticulous soft tissue handling, dead space management, and robust flap coverage are paramount. Silver-coated implants or iodine-supported titanium implants are increasingly utilized to mitigate this risk.
