Patient Presentation & History
Our case involves a 32-year-old male patient who presented with an 18-month history of insidious onset, progressively worsening pain in his left distal leg. Initially, the pain was dull, intermittent, and exacerbated by prolonged standing or ambulation. Over the last three months, the pain localized specifically to the anteromedial aspect of the distal tibia, becoming constant and significantly impacting his quality of life and work as a construction supervisor. He reported a recent, acute increase in pain following a minor misstep from a curb, without direct trauma, suggesting a possible pathological event.
He denied any fevers, chills, night sweats, or unintentional weight loss. There was no history of prior trauma to the affected limb, nor any family history of bone tumors or genetic syndromes. His past medical history was unremarkable, with no significant comorbidities. He was a non-smoker and consumed alcohol occasionally. His social history was stable, living with his family.
At the time of presentation to our trauma service, his primary complaint was severe, localized pain in the left distal tibia, significantly exacerbated by any weight-bearing. He reported an inability to ambulate without crutches and the development of a noticeable swelling in the area over the past two months. This presentation, particularly the protracted nature of symptoms followed by an acute worsening suggestive of a pathological event, raised immediate concerns for an underlying osseous lesion.
Clinical Examination
Upon initial assessment, the patient appeared to be in mild distress due to pain, favoring his left lower extremity.
Inspection
- Left Distal Tibia: Gross inspection revealed a subtle but palpable fusiform swelling involving the anteromedial aspect of the distal tibia, approximately 10 cm proximal to the ankle joint. The overlying skin appeared normal, without erythema, warmth, or trophic changes. There were no visible open wounds, sinuses, or prominent venous patterns. A slight anterior bowing of the tibia was noted, although this was difficult to ascertain definitively due to the swelling.
- Alignment: Overall alignment of the limb was maintained, with no gross rotational or angular deformities apparent beyond the localized swelling.
- Gait: The patient was non-weight bearing on the left lower extremity, ambulating with axillary crutches.
Palpation
- Tenderness: Exquisite point tenderness was elicited over the anteromedial distal tibia, precisely correlating with the area of swelling.
- Mass: A firm, non-mobile, bony-hard mass was palpable within the distal tibial shaft, confirming a primary osseous rather than soft tissue lesion. The margins of the palpable mass were ill-defined proximally and distally, blending into the normal bone contours.
- Temperature: No localized increase in skin temperature was noted, which often suggests an acute inflammatory or infectious process.
- Regional Lymph Nodes: No palpable inguinal or popliteal lymphadenopathy was detected.
- Muscles: Surrounding muscle compartments were soft and non-tender.
Range of Motion (ROM)
- Ankle Joint: Active and passive dorsiflexion and plantarflexion of the ankle were mildly restricted due to pain at the extremes of motion but otherwise preserved. Subtalar and midfoot motions were similarly affected by pain.
- Knee Joint: Full, pain-free range of motion was demonstrated at the ipsilateral knee joint, confirming the localized nature of the lesion to the distal tibia.
- Hip Joint: Full, pain-free range of motion.
Neurological and Vascular Assessment
- Neurological: Distal neurological function was intact. Sensation to light touch was preserved in the superficial peroneal, deep peroneal, sural, saphenous, and tibial nerve distributions. Motor strength was 5/5 for ankle dorsiflexion (deep peroneal), plantarflexion (tibial), eversion (superficial peroneal), and toe movements, though pain made maximal effort difficult. No pathological reflexes were elicited.
- Vascular: Distal pulses (dorsalis pedis and posterior tibial) were strong, palpable, and symmetrical bilaterally. Capillary refill was brisk (<2 seconds) in all digits. No signs of venous insufficiency or arterial compromise were present.
In summary, the clinical examination strongly indicated a primary osseous lesion of the distal tibia, characterized by chronic pain, progressive swelling, and acute exacerbation consistent with a pathological event, demanding urgent comprehensive imaging and diagnostic workup.
Imaging & Diagnostics
Following the clinical assessment, a structured imaging and diagnostic approach was initiated to characterize the lesion and guide further management.
Radiographs
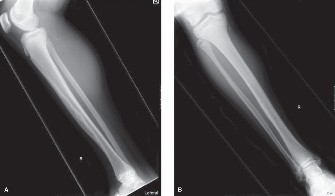
Initial plain radiographs of the left tibia and fibula (anteroposterior and lateral views) were obtained.
*
Findings:
The radiographs revealed a large, eccentric, expansile lytic lesion involving the distal third of the tibial diaphysis and metaphysis. The lesion measured approximately 8 cm in craniocaudal dimension and 3 cm in anteroposterior dimension. Characteristic features included:
*
Cortical expansion and thinning:
Significant ballooning and thinning of the anterior and medial cortices were evident.
*
Sclerotic rim:
A thin, irregular sclerotic rim was noted around portions of the lesion, suggesting a slow growth rate.
*
Multicystic or "soap bubble" appearance:
Internal trabeculation gave the lesion a loculated, multicystic, or "soap bubble" appearance, particularly on the lateral view.
*
Pathological Fracture:
A subtle transverse cortical fracture was identified through the anterior cortex within the lesion, accounting for the acute exacerbation of pain. There was no significant displacement.
*
Periosteal reaction:
Minimal to no periosteal reaction was observed, which is atypical for aggressive malignancies or infections but consistent with the indolent nature of certain primary bone tumors.
*
Matrix:
No overt chondroid or osteoid matrix was evident within the lesion.
*
Fibula:
The adjacent fibula appeared unremarkable.
Computed Tomography (CT) Scan
A non-contrast CT scan of the left lower leg was performed to further delineate the cortical destruction, medullary involvement, and precise extent of the lesion, particularly concerning the pathological fracture.
*
Indications:
High-resolution bone detail, assessment of cortical integrity, evaluation of the pathological fracture geometry, and precise tumor margins for surgical planning.
*
Findings:
The CT scan confirmed the extensive lytic and expansile nature of the lesion. It provided superior detail regarding the thin, compromised cortices, confirming the subtle pathological fracture through the anterior cortex. The "soap bubble" appearance was more clearly visualized, consistent with intralesional septations. While primarily intraosseous, subtle evidence of cortical breach and possible minimal soft tissue extension was suspected anteriorly, necessitating further evaluation with MRI. The CT also helped in evaluating the extent of medullary cavity involvement and the precise transition zones to healthy bone, which are crucial for pre-operative osteotomy planning.
Magnetic Resonance Imaging (MRI)
An MRI of the left tibia, with and without intravenous gadolinium contrast, was performed for comprehensive soft tissue assessment, intraosseous tumor extent, and detection of skip lesions or neurovascular involvement.
*
Indications:
Superior soft tissue contrast, evaluation of marrow involvement, detection of potential skip lesions, assessment of soft tissue extension, and relationship to neurovascular structures.
*
Findings:
*
T1-weighted images:
Showed a heterogeneous lesion with areas of low signal intensity, consistent with solid tumor components and possible fibrous stroma.
*
T2-weighted images (including fat-suppressed sequences):
Demonstrated high signal intensity areas within the lesion, reflecting its cellular and sometimes cystic or edematous components, as well as areas of fluid-fluid levels indicative of cystic degeneration or hemorrhage. The heterogeneous signal further supported the presence of solid and cystic areas.
*
Gadolinium-enhanced T1-weighted images:
Revealed heterogeneous enhancement, primarily at the periphery of solid components, further outlining the lesion's extent and any areas of cortical disruption with associated soft tissue enhancement.
*
Extent:
Confirmed significant intraosseous involvement of the distal tibial diaphysis and metaphysis, extending approximately 8.5 cm. Importantly, it clarified that while the cortex was markedly thinned and breached anteriorly at the fracture site, gross extraosseous soft tissue mass was minimal, confined to a small region immediately adjacent to the anterior cortical defect. No skip lesions were identified in the proximal tibia. The relationship to the neurovascular bundle (tibial nerve and posterior tibial artery/vein) was carefully assessed and appeared to be free of direct involvement, though in close proximity.
Biopsy
Given the imaging findings highly suspicious for a primary bone tumor, an image-guided core needle biopsy was performed.
*
Procedure:
Under CT guidance, multiple core biopsies were obtained from different areas of the lesion, including both solid and cystic components, ensuring adequate tissue sampling while avoiding neurovascular structures.
*
Histopathology:
The biopsy specimen revealed a biphasic pattern characterized by nests and cords of epithelial cells within a spindle cell stroma. The epithelial cells exhibited basaloid or squamoid differentiation, with some areas showing clear cell features. Immunohistochemical staining was crucial for definitive diagnosis: positive for cytokeratins (CK AE1/AE3, CK5/6) and epithelial membrane antigen (EMA) in the epithelial component, and positive for vimentin in the spindle cell component. These findings were pathognomonic for
adamantinoma
.
Staging
Following the definitive diagnosis of adamantinoma:
*
Local Staging (Enneking/MSTS):
The lesion was classified as a Grade 1 (low-grade malignant) tumor with intraosseous but minimal extraosseous extension (following cortical breach at the fracture site). This corresponds to a Stage IB tumor in the MSTS classification due to its low-grade malignant potential and extracompartmental extension (even if minor).
*
Systemic Staging:
*
CT Chest:
Performed to rule out pulmonary metastases, which, though rare, are the most common site of distant spread for adamantinoma. The scan was negative for metastatic disease.
*
Bone Scan (Technetium-99m Methylene Diphosphonate):
Performed to identify any other synchronous bone lesions. The scan showed increased uptake only at the site of the primary lesion in the distal tibia, confirming no other osseous metastases.
The comprehensive diagnostic workup confirmed a low-grade malignant adamantinoma of the distal left tibia with a pathological fracture, without evidence of distant metastasis.
Differential Diagnosis
The imaging characteristics of a lytic, expansile lesion in the tibia, particularly in a young adult, necessitate a broad differential diagnosis. The table below outlines key differentials considered for this case:
| Feature | Adamantinoma (Our Case) | Fibrous Dysplasia | Osteofibrous Dysplasia | Aneurysmal Bone Cyst (ABC) | Low-Grade Chondrosarcoma |
|---|---|---|---|---|---|
| Typical Age | 20-40s (patient is 32) | <30 years (often 10-30s) | <20 years (often <10) | <30 years | >40 years |
| Location | Tibia (90% - distal diaphysis/metaphysis) | Any bone, often proximal femur, tibia, ribs, craniofacial | Tibia/Fibula (cortical, anterior aspect) | Any bone, often long bones, spine, pelvis | Pelvis, proximal femur, humerus, ribs |
| X-ray Appearance | Lytic, eccentric, expansile, sclerotic rim, "soap bubble", cortical thinning/breach | "Ground glass" matrix, sclerotic margins, bowing, cortical thinning, endosteal scalloping | Cortical lucency, often anterior tibia, non-expansile, minimal medullary involvement | Expansile, lytic, septations, thin sclerotic rim, fluid-fluid levels | Lytic with endosteal scalloping, ill-defined margins, punctate or ring-and-arc calcifications (chondroid matrix) |
| CT Findings | Confirms cortical destruction, multicystic nature, pathological fracture | Confirms "ground glass" density, cortical expansion | Cortical based, usually purely intracortical, no medullary extension | Fluid-fluid levels, thin septations, cortical destruction | Calcified chondroid matrix, endosteal erosion, cortical breach |
| MRI Findings | Heterogeneous signal (solid/cystic), enhancement, minimal soft tissue extension, marrow involvement | Variable signal, can enhance, absence of aggressive features | Hypointense T1, hyperintense T2, minimal enhancement, strictly cortical | Fluid-fluid levels (pathognomonic), heterogeneous enhancement, soft tissue component | Lobulated masses, high signal on T2 (hyaline cartilage), heterogeneous enhancement |
| Histology | Biphasic pattern: epithelial cells (basaloid/squamoid) within spindle cell stroma. Cytokeratin+, EMA+, Vimentin+ | Fibrous stroma with immature woven bone trabeculae, "Chinese characters" pattern. No osteoblastic rimming. | Identical to fibrous dysplasia, but exclusively in cortical bone. | Blood-filled cystic spaces, fibrous septa, osteoclast-like giant cells. No true endothelial lining. | Lobules of hyaline cartilage, increased cellularity, nuclear atypia, myxoid change. |
| Biological Behavior | Low-grade malignant, locally aggressive, high recurrence if inadequate excision. Potential for late metastasis (lungs). | Benign, can cause deformity, pain, pathological fracture. Usually self-limiting. | Benign, self-limiting (usually resolves after skeletal maturity). Rarely transforms to adamantinoma. | Benign, locally aggressive, high recurrence if incomplete treatment. | Low-grade malignant, locally aggressive, slow growth, potential for dedifferentiation. |
| Management | Wide en bloc resection (limb salvage). Adjuvant therapy generally ineffective. | Observation, curettage/grafting for symptoms or fracture. Bisphosphonates for extensive polyostotic disease. | Observation, curettage for symptoms/fracture (often no treatment needed). | Intralesional curettage, adjuvant therapy (phenol, cryotherapy, argon beam), embolization, resection. | Wide en bloc resection. |
Surgical Decision Making & Classification
The definitive diagnosis of adamantinoma, a rare low-grade malignant primary bone tumor, with an associated pathological fracture, profoundly influenced the surgical decision-making process.
Why Operative Intervention?
- Malignant Nature: Adamantinoma, despite its low-grade designation, is a true malignancy. It is locally aggressive and carries a significant risk of local recurrence if not completely excised with adequate margins. While distant metastases are rare (occurring in 10-30% of cases, primarily to the lungs, often years after initial presentation), the primary goal is complete local control.
- Pathological Fracture: The presence of a pathological fracture within the tumor necessitates operative stabilization. Conservative management of such fractures is fraught with risks, including non-union, progressive deformity, and potential for tumor progression or soft tissue contamination if left untreated. More importantly, it provides an opportunity for definitive tumor resection.
- Resistance to Adjuvant Therapies: Adamantinoma is notoriously resistant to both conventional chemotherapy and radiation therapy. Therefore, surgical resection with wide margins remains the cornerstone and often the sole curative treatment modality.
- Preservation of Function: The patient's young age and active lifestyle underscored the need for a reconstructive strategy that would achieve tumor control while maximizing long-term limb function and durability.
Surgical Goals:
- Achieve Wide Oncologic Margins: The primary objective is a complete en bloc resection of the tumor with a surrounding cuff of healthy tissue (wide margin) to minimize the risk of local recurrence. This includes excising the entire biopsy tract.
- Reconstruct the Tibial Defect: The resection of a significant segment of the distal tibia creates a large intercalary bone defect that requires robust reconstruction to restore structural integrity, maintain limb length, and allow for functional weight-bearing.
- Preserve Limb Function: Minimize morbidity, avoid neurovascular compromise, and allow for a return to pre-morbid activity levels as much as possible.
Classification (MSTS Staging System):
Based on the diagnostic workup, the tumor was classified according to the Musculoskeletal Tumor Society (MSTS) staging system:
*
Grade (G):
G1 (Low-grade malignancy) – Adamantinoma is inherently a low-grade tumor.
*
Site (T):
T1 (Intracompartmental) – While there was cortical breach at the pathological fracture site, the extraosseous extension was minimal and confined to the immediate vicinity, not extending extensively into an adjacent compartment. However, a strict interpretation of cortical breach indicates extracompartmental extension (T2). Given the indolent nature, it is often treated as a T1, but carefully considered for T2 if significant. For adamantinoma, even minimal cortical breach implies a T2 lesion.
*
Metastasis (M):
M0 (No regional or distant metastasis) – Confirmed by CT chest and bone scan.
Therefore, the tumor was staged as G1T2M0 – Stage IIB (low-grade, extracompartmental, no metastases). This stage mandates wide en bloc resection for limb salvage.
Limb Salvage vs. Amputation:
Given the MSTS Stage IIB classification, limb salvage was the preferred treatment strategy.
*
Favorable Factors for Limb Salvage:
* Low-grade malignancy.
* No evidence of distant metastasis.
* Minimal extraosseous soft tissue extension.
* Intact neurovascular bundle, not directly involved by the tumor.
* Patient's desire for limb preservation.
* Location (distal tibia is amenable to various reconstructive options).
*
Amputation Considerations:
Amputation would only be considered in cases of extensive neurovascular involvement, gross infection, massive extraosseous extension making wide margins impossible, or failed limb salvage procedures. None of these were present in this case.
Reconstruction Planning:
The length of the planned resection (approximately 10 cm, including 2 cm margins proximally and distally to the visible tumor extent) dictated the need for a structural reconstruction. Several options were considered:
*
Structural Allograft:
A bulk intercalary allograft is a common choice for long bone defects. It provides immediate structural support and allows for gradual incorporation.
*
Vascularized Autogenous Fibula Graft (VFG):
Can be used for smaller defects or in conjunction with allografts (allograft-VFG composite) to enhance healing and biological integration, particularly useful in challenging situations like infection or non-union.
*
Endoprosthetic Reconstruction:
Less common for the mid-diaphyseal tibia unless massive defects or joint involvement.
*
Masquelet Technique (Induced Membrane):
A two-stage procedure typically used for septic non-unions or large defects where vascularization is critical, but perhaps less ideal for immediate oncologic reconstruction in this context.
After careful consideration, a structural intercalary tibia allograft combined with intramedullary nailing and plate augmentation was selected as the optimal reconstructive strategy. This approach offers immediate mechanical stability, the potential for biological integration, and restoration of function.
Surgical Technique / Intervention
The surgical procedure was meticulously planned to achieve wide oncologic margins and stable reconstruction of the tibial defect.
Patient Positioning and Preparation
- The patient was positioned supine on the operating table.
- A high thigh tourniquet was applied to ensure a bloodless field during the resection and initial reconstruction phases.
- The entire left lower extremity, from the midthigh to the toes, was prepped and draped free to allow for full manipulation of the limb and access to the entire tibia.
- Pre-operative planning involved precise marking of skin incisions based on MRI and CT images, outlining the planned resection margins and ensuring inclusion of the biopsy tract.
Surgical Approach and Tumor Resection
- Incision: A long anteromedial curvilinear incision was made over the distal tibia, extending well beyond the proximal and distal planned osteotomy sites. The incision was designed to allow a wide margin of healthy skin and subcutaneous tissue around the biopsy tract and tumor. The biopsy tract, which traversed the anterior skin and soft tissues to the tibia, was carefully circumscribed and marked for en bloc removal.
- Soft Tissue Dissection: Subcutaneous tissue and fascia were incised. Care was taken to identify and protect the saphenous nerve and vein. The anterior compartment muscles (tibialis anterior, extensor digitorum longus, extensor hallucis longus, peroneus tertius) were retracted laterally, and the posterior compartment muscles were retracted posteriorly, exposing the anteromedial aspect of the tibia.
- Biopsy Tract Excision: The previously performed core needle biopsy tract was excised en bloc with the tumor specimen. This is a critical step to prevent local recurrence from tumor seeding. All tissues traversed by the biopsy needle were included in the resection.
- Periosteal Stripping: The periosteum was carefully stripped from the bone surface both proximally and distally to the planned osteotomy sites, maintaining a wide cuff of periosteal and soft tissue margin around the tumor. The anterior tibial neurovascular bundle (anterior tibial artery, veins, and deep peroneal nerve) was identified and protected.
- Osteotomies: Based on pre-operative templating and intra-operative fluoroscopic confirmation, proximal and distal osteotomies were performed with an oscillating saw. These osteotomies were made perpendicular to the long axis of the tibia, ensuring clear 2 cm macroscopic margins from the radiographic and MRI extent of the tumor. For this case, the proximal osteotomy was approximately 12 cm from the ankle joint, and the distal osteotomy was 2 cm proximal to the ankle joint articular surface. A total segment of approximately 10 cm of tibia was resected.
- Specimen Removal: The resected segment of the distal tibia, containing the adamantinoma and the excised biopsy tract, was carefully removed en bloc. The specimen was oriented and sent for pathological analysis, including frozen sections of the proximal and distal bone margins to confirm clear margins.
Reconstruction of Tibial Defect
- Allograft Preparation: A size-matched, pre-selected fresh-frozen intercalary tibia allograft (approximately 10 cm in length) was prepared. The ends of the allograft were shaped to perfectly match the host bone osteotomy sites.
-
Intramedullary Nailing:
A long, reamed intramedullary nail (e.g., an unreamed tibial nail) was chosen to span the host bone-allograft junctions and provide robust axial stability.
- Pilot Hole: A guide wire was inserted into the proximal host tibia fragment and advanced proximally, crossing the knee joint, for nail entry.
- Nail Insertion: The intramedullary nail was then carefully inserted antegrade from the proximal tibia, through the proximal host bone, across the proximal allograft-host junction, through the allograft, across the distal allograft-host junction, and into the distal host bone fragment. Distal and proximal locking screws were inserted to secure the construct. This provided immediate axial and rotational stability.
- Plate Augmentation: To enhance rotational and bending stability, particularly at the host-allograft junctions, a pre-contoured locking plate (e.g., LCP Distal Tibia Plate) was applied to the anterolateral aspect of the reconstructed tibia. The plate spanned both osteotomy sites, with screws placed into both the host bone and the allograft, providing additional compression and stability. This is crucial for optimizing allograft incorporation and preventing non-union.
- Autograft at Junctions: Cancellous autogenous bone graft (harvested from the ipsilateral iliac crest or distal fibula if available) was packed around the proximal and distal host-allograft junctions. This promotes biological incorporation and accelerates union at the osteotomy sites.
- Soft Tissue Coverage: The surrounding soft tissues were carefully repaired to provide good coverage of the reconstruction.
Wound Closure
- Hemostasis was achieved.
- A suction drain was placed to prevent hematoma formation.
- Fascia, subcutaneous tissue, and skin were closed in layers.
- A sterile dressing and a well-padded splint were applied to provide immediate immobilization and protection.
Post-Operative Protocol & Rehabilitation
The post-operative management focused on pain control, wound healing, protection of the reconstructive construct, and gradual rehabilitation to restore function.
Immediate Post-Operative Period (Day 0-7)
- Pain Management: Multimodal analgesia including epidural or regional nerve blocks, oral opioids, and NSAIDs.
- Wound Care: Daily dressing changes, monitoring for signs of infection (erythema, warmth, purulent discharge). Drain output was monitored and removed when output was minimal (<30 ml/24h).
- Neurovascular Monitoring: Hourly neurovascular checks of the foot (color, temperature, capillary refill, sensation, motor function) for the first 24-48 hours, then regular daily checks.
- Immobilization: The limb was kept in a rigid, well-padded posterior splint or a removable CAM walker boot to provide protection and support.
- Weight-bearing: Strict non-weight bearing (NWB) on the operative limb was enforced.
- Mobilization: Ambulation with crutches or a walker, NWB on the affected side. Encouragement of upper extremity strengthening and core stability exercises.
- DVT Prophylaxis: Standard DVT prophylaxis initiated (e.g., low molecular weight heparin).
Early Rehabilitation Phase (Weeks 1-12)
- Wound Healing: Suture/staple removal at 2-3 weeks, assuming good wound healing.
-
ROM Exercises:
- Ankle: Gentle active and passive range of motion exercises for the ankle and foot were initiated, within the limits of pain and stability provided by the boot. Avoidance of aggressive mobilization at the allograft junction.
- Knee/Hip: Full active and passive ROM exercises for the knee and hip were encouraged to prevent stiffness.
-
Weight-bearing Progression:
- Weeks 0-6: Strict NWB.
- Weeks 6-12: Gradual progression to toe-touch weight-bearing (TTWB) or 10-20% partial weight-bearing (PWB), depending on radiographic evidence of early healing and stability of the construct. This was guided by physical therapy and closely monitored.
- Strengthening: Isometric exercises for calf, quadriceps, and hamstring muscles were initiated.
- Edema Control: Compression stockings and elevation to manage swelling.
Intermediate Rehabilitation Phase (Months 3-6)
- Radiographic Assessment: Regular radiographs (monthly initially, then every 6-8 weeks) to assess allograft incorporation, host-allograft junction healing, and integrity of the fixation.
- Weight-bearing Progression: Progressive increase in weight-bearing, advancing from PWB to full weight-bearing (FWB) as tolerated and as radiographic union progresses. This is a critical phase and requires patience, as allograft incorporation is slower than autograft.
-
Physical Therapy:
- Focus on full ankle and knee ROM.
- Progressive strengthening exercises for all lower extremity muscle groups, including eccentric and concentric contractions.
- Balance and proprioception training.
- Gait training to normalize walking pattern.
- Stationary cycling or swimming to improve cardiovascular fitness and limb function without excessive impact.
- Orthosis: The CAM walker boot was typically discontinued once FWB was achieved and confidence regained, usually replaced by supportive footwear.
Long-Term Rehabilitation and Follow-up (Months 6 onwards)
- Functional Progression: Continuation of strengthening and conditioning. Gradual return to activities of daily living and work-related tasks, avoiding high-impact activities initially.
- Sport-Specific Training: For patients desiring to return to sports, specific training was initiated once strength, balance, and allograft healing were confirmed. High-impact sports were generally discouraged for at least 12-18 months.
-
Oncologic Surveillance:
Long-term follow-up is crucial for adamantinoma.
- Clinical Examination: Annually for the first 5 years, then biannually for up to 10-15 years, assessing for local recurrence, pain, or functional deficits.
- Radiographs: Annual X-rays of the operative limb to monitor allograft integrity, incorporation, and signs of recurrence.
- MRI: Local MRI every 6-12 months for the first 2-3 years, then annually for several years to detect subtle local recurrence not visible on X-ray.
- CT Chest: Annual CT scan of the chest for the first 5-10 years to screen for pulmonary metastases, which can occur late.
- Monitoring for Complications: Close monitoring for allograft-related complications such as non-union, infection, allograft fracture, or stress shielding.
- Hardware Removal: Consideration for hardware removal after complete union and allograft incorporation, if symptomatic. However, often the intramedullary nail and plates are retained indefinitely if well-tolerated and not interfering with function.
The entire rehabilitation process for such a complex reconstruction can span 12-18 months or more until maximal functional recovery is achieved.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- Think Adamantinoma for Tibial Lesions: Adamantinoma is a rare primary bone tumor, but 90% occur in the tibia (most commonly mid-distal diaphysis). Any uncharacteristic lytic lesion in the tibia, especially in a young to middle-aged adult with chronic pain, should raise suspicion.
- Biphasic Histology is Key: The definitive diagnosis relies on histopathology showing the characteristic biphasic pattern of epithelial cells (often basaloid or squamoid, CK+, EMA+) nested within a spindle cell stromal component (vimentin+). This requires an experienced musculoskeletal pathologist.
- Wide En Bloc Resection is Curative: Adamantinoma is resistant to chemotherapy and radiotherapy. Surgical resection with wide, clear oncologic margins is the only curative treatment. Inadequate margins lead to high local recurrence rates.
- Always Excise the Biopsy Tract: The entire biopsy tract must be resected en bloc with the primary tumor to prevent local recurrence from tumor seeding. This is a non-negotiable principle in oncologic surgery.
- Comprehensive Staging: Despite its low-grade nature, complete local and systemic staging (including MRI for local extent and CT chest for lung metastases) is essential. Adamantinoma can metastasize, albeit usually late.
- Reconstruction Planning: For large tibial defects, structural intercalary allografts, often augmented with intramedullary nails and plates, provide stable reconstruction. Vascularized fibula grafts can be considered for smaller defects or in composite constructs.
- Long-Term Follow-up: Due to the potential for late local recurrence and distant metastasis (sometimes decades later), lifelong follow-up with clinical exams, local imaging, and surveillance CT chest is critical.
Pitfalls
-
Misdiagnosis:
- Clinical: Can be mistaken for chronic osteomyelitis, stress fracture, or fibrous dysplasia due to similar symptoms and indolent presentation.
- Radiological: Early adamantinoma can mimic fibrous dysplasia, osteofibrous dysplasia, or aneurysmal bone cyst. The multicystic/soap-bubble appearance can be misleading.
- Pathological: Superficial or inadequate biopsy can lead to misdiagnosis, especially if only the fibrous stromal component is sampled, or if epithelial cells are sparse or misinterpreted. Close collaboration with pathology is vital.
- Inadequate Surgical Margins: This is the most common reason for local recurrence. R0 resection (no residual tumor) is paramount. Don't be timid with resection length or soft tissue margins.
- Neglecting Biopsy Tract Excision: Failing to excise the entire biopsy tract is a guaranteed way to leave behind microscopic tumor cells, leading to recurrence.
- Underestimating Tumor Extent: Relying solely on X-rays can lead to underestimation of the true intraosseous and subtle extraosseous extent. MRI is indispensable for surgical planning.
-
Allograft Complications:
Structural allografts carry inherent risks, including:
- Non-union: At host-allograft junctions, requiring revision surgery. Autograft augmentation helps mitigate this.
- Infection: A devastating complication, especially in devascularized allografts, often requiring removal and reconstructive challenges.
- Fracture: Allograft fatigue fracture can occur prior to complete incorporation.
- Delayed Incorporation: Healing and remodeling are slow, prolonging rehabilitation and risk of complications.
- Delayed Diagnosis: The indolent nature often leads to a long symptom duration. Delayed diagnosis can result in a larger tumor, pathological fracture, and more extensive resection required.
- Lack of Systemic Surveillance: While rare, pulmonary metastases can occur. Failing to perform and maintain long-term CT chest surveillance means missing these potentially treatable distant recurrences.
