Radial Head & Neck Fracture ORIF: An Intraoperative Masterclass
Key Takeaway
Step into the operating room for a comprehensive masterclass on open reduction and internal fixation of radial head and neck fractures. This guide covers essential anatomy, detailed preoperative planning, granular intraoperative execution, and crucial pearls for successful outcomes. Master the nuances of elbow trauma, from precise dissection techniques and implant application to managing complex associated injuries and optimizing postoperative rehabilitation.
Comprehensive Introduction and Patho-Epidemiology
The management of radial head and neck fractures represents a profound challenge in orthopedic trauma, requiring an exquisite understanding of elbow kinematics and articular congruity. Historically, the radial head was viewed as an expendable structure, often subjected to primary excision following traumatic injury. However, contemporary orthopedic surgery recognizes the radial head as a vital secondary stabilizer of the elbow joint, particularly in the presence of ligamentous incompetence. Our objective in treating these injuries has definitively shifted from mere osseous union to the anatomic restoration of the radiocapitellar and proximal radioulnar joints (PRUJ). This intraoperative masterclass delineates the highly technical nuances of Open Reduction and Internal Fixation (ORIF) for radial head and neck fractures, providing the reconstructive surgeon with a comprehensive blueprint for optimizing functional outcomes.
Epidemiologically, radial head and neck fractures are the most frequently encountered elbow fractures in the adult population, comprising approximately 33% of all elbow fractures and up to 20% of all acute elbow trauma. The typical demographic involves young to middle-aged adults, though a bimodal distribution is increasingly recognized with the aging, osteoporotic population. The pathomechanics classically involve a fall onto an outstretched hand (FOOSH) with the elbow positioned in varying degrees of extension and the forearm in pronation. This mechanism generates a forceful axial load coupled with a valgus moment, driving the radial head violently against the capitellum. The resulting shear and impaction forces dictate the fracture morphology, ranging from simple non-displaced marginal fractures to highly comminuted, unsalvageable articular destruction.
The pathogenesis of these fractures must be viewed through the lens of the "Horii circle" of soft tissue disruption. Energy dissipation during the traumatic event rarely isolates the radial head; rather, it propagates through the lateral soft tissue envelope to the medial structures. Consequently, the orthopedic surgeon must maintain a high index of suspicion for associated injuries. Valgus loading frequently compromises the medial collateral ligament (MCL), while posterolateral rotatory forces disrupt the lateral ulnar collateral ligament (LUCL). The notorious "terrible triad" of the elbow—encompassing a radial head fracture, a coronoid process fracture, and a posterior elbow dislocation—represents the zenith of this destabilizing cascade. Furthermore, axial forces can propagate distally, rupturing the interosseous membrane (IOM) and destabilizing the distal radioulnar joint (DRUJ), culminating in the catastrophic Essex-Lopresti lesion.

The evolution of treatment paradigms heavily emphasizes joint preservation whenever feasible. While arthroplasty remains an invaluable tool for highly comminuted, irreparable fractures (typically defined as those with greater than three articular fragments), ORIF is the gold standard for reconstructable displaced fractures. The decision to proceed with ORIF is predicated on the surgeon's ability to achieve absolute stability of the articular surface to allow for early, aggressive mobilization. Failure to recognize the intricate interplay between the osseous anatomy and the surrounding ligamentous constraints will inevitably result in stiffness, chronic instability, or post-traumatic arthrosis. Therefore, mastering the nuances of radial head ORIF is an indispensable skill for the modern orthopedic trauma surgeon.
Detailed Surgical Anatomy and Biomechanics
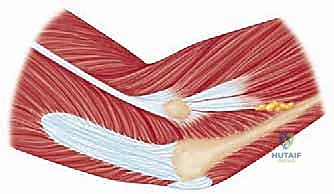
The radial head is a uniquely complex, entirely intra-articular structure that articulates synergistically with both the capitellum of the humerus and the lesser sigmoid notch of the ulna. The superior articular surface is concavely shaped (the articular "dish" or fovea) to accommodate the spherical capitellum, forming the radiocapitellar joint. This articulation is responsible for flexion and extension, as well as providing a pivot point for forearm rotation. The peripheral margin of the radial head articulates with the lesser sigmoid notch of the ulna, forming the PRUJ. This margin is not perfectly cylindrical but rather elliptical, with variable offset from the radial neck. The PRUJ is tightly constrained by the annular ligament, which originates and inserts on the anterior and posterior margins of the lesser sigmoid notch, respectively, facilitating smooth pronation and supination.
The vascular anatomy of the proximal radius is notoriously tenuous, rendering the radial head highly susceptible to avascular necrosis (AVN) following trauma or aggressive surgical dissection. The primary arterial supply is derived from a single branch of the radial recurrent artery. This vessel typically penetrates the radial head and neck at a specific non-articular quadrant known as the "safe zone." Minor collateral contributions arise from the interosseous recurrent arteries, which penetrate the capsule near its distal insertion. Because the articular cartilage lacks perichondrial vessels, the subchondral bone relies entirely on this retrograde intraosseous blood flow. Extensive circumferential soft tissue stripping or indiscriminate placement of hardware outside the safe zone can obliterate this fragile vascular network, leading to catastrophic ischemic collapse of the radial head.
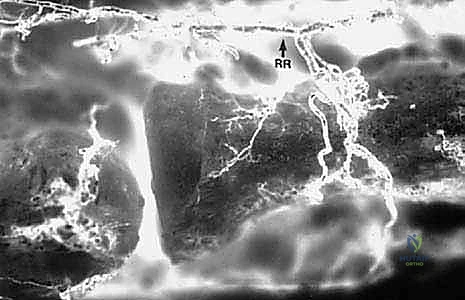
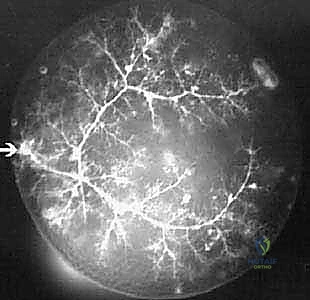
Neurovascular proximity demands meticulous surgical technique. The posterior interosseous nerve (PIN), a major motor branch of the radial nerve, is the structure at greatest risk during lateral and posterolateral approaches to the elbow. After bifurcating from the radial nerve anterior to the radiocapitellar joint, the PIN dives beneath the arcade of Frohse and winds around the radial neck within the substance of the supinator muscle. The distance from the radiocapitellar joint to the PIN varies significantly depending on forearm rotation; pronation moves the nerve anteriorly and medially, safely distancing it from the surgical field during a lateral approach, whereas supination brings it perilously close to the radial neck. Aggressive distal retraction or blind placement of retractors (such as Hohmann retractors) around the radial neck can result in devastating traction neuropraxia or direct transection.
Biomechanically, the radial head is a critical load-bearing structure and a vital stabilizer. It transmits approximately 60% of the axial load from the wrist across the elbow joint. In terms of stability, the anterior band of the MCL is the primary restraint to valgus stress. However, the radial head acts as the primary secondary stabilizer; in the setting of an incompetent MCL, the radial head provides up to 30% of the resistance to valgus forces. Consequently, excision of the radial head in the presence of an MCL tear will inevitably lead to gross valgus instability. Furthermore, the radial head is integral to longitudinal stability. The interosseous membrane (IOM) transfers axial loads from the radius to the ulna. If the IOM is disrupted (Essex-Lopresti injury), the radial head becomes the sole restraint to proximal radial migration. In such scenarios, radial head excision is absolutely contraindicated, and anatomic reconstruction or rigid arthroplasty is mandatory to prevent proximal radioulnar impingement and ulnocarpal impaction.
Exhaustive Indications and Contraindications
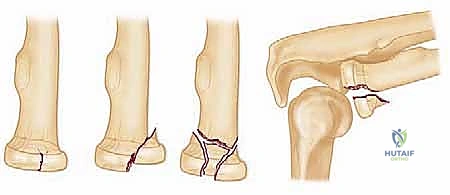
Patient selection and precise operative indications are the cornerstones of successful outcomes in radial head trauma. The modified Mason classification, refined by Johnston and Morrey, remains the standard framework for guiding treatment decisions, despite known limitations in interobserver reliability. The fundamental philosophy dictates that the native radial head should be preserved whenever technically feasible, provided that a stable, anatomic reduction can be achieved that permits immediate postoperative mobilization. The decision matrix is heavily influenced not only by the fracture pattern but also by the presence of associated ligamentous injuries, the patient's physiological age, and their functional demands.
Indications for ORIF
The primary indication for ORIF is a displaced Mason Type II fracture (marginal segment displacement >2 mm) that creates a mechanical block to forearm pronation or supination. Even in the absence of a frank mechanical block, ORIF is strongly considered for displaced fragments involving more than 30% of the articular surface to prevent post-traumatic arthrosis and joint incongruity. Mason Type III fractures (comminuted) present a more complex decision tree. ORIF is indicated for Type III fractures if the comminution is limited to three or fewer primary articular fragments that can be anatomically reduced and rigidly fixed. Furthermore, in any fracture pattern associated with elbow instability (Mason Type IV)—such as the terrible triad, Monteggia variants, or Essex-Lopresti lesions—the radial head must be managed operatively. If the fracture is reconstructable, ORIF is preferred; if it is unsalvageable, radial head arthroplasty is indicated. Acute excision is historically obsolete and strictly contraindicated in the setting of instability.

Contraindications to ORIF
Absolute contraindications to ORIF include non-displaced or minimally displaced Mason Type I fractures lacking any mechanical block to motion, which are universally managed non-operatively with a brief period of immobilization followed by early active range of motion. Severe comminution (greater than three articular fragments) is a strong relative contraindication; literature consistently demonstrates that attempting to piece together highly fragmented radial heads ("humpty dumpty" surgery) leads to high rates of hardware failure, nonunion, AVN, and profound stiffness. In such cases, primary radial head arthroplasty yields vastly superior and more predictable functional outcomes. Additionally, severe osteopenia or pre-existing advanced radiocapitellar osteoarthritis may preclude stable internal fixation, tilting the scale toward arthroplasty or, in low-demand elderly patients with isolated injuries, fragment excision.
| Clinical Scenario / Parameter | Indication for ORIF | Contraindication / Alternative |
|---|---|---|
| Mason Type I | None | Non-operative management |
| Mason Type II | Displacement >2mm, mechanical block | Excision (only if isolated and low demand) |
| Mason Type III | ≤ 3 fragments, reconstructable | >3 fragments (Arthroplasty indicated) |
| Mason Type IV (Instability) | Reconstructable fracture | Unsalvageable (Arthroplasty indicated). Excision is ABSOLUTELY CONTRAINDICATED. |
| Essex-Lopresti Lesion | Reconstructable fracture | Arthroplasty. Excision is ABSOLUTELY CONTRAINDICATED. |
| Bone Quality | Good cortical density | Severe osteopenia (Arthroplasty preferred) |
The "grey zone" in decision-making typically involves borderline Mason Type III fractures. The surgeon must critically evaluate preoperative computed tomography (CT) scans to assess the size, location, and bone stock of the articular fragments. The threshold for converting from a planned ORIF to an arthroplasty should be low if intraoperative assessment reveals that rigid fixation cannot be achieved or if the articular surface remains incongruent. It is imperative that the surgeon consents the patient for both ORIF and possible radial head replacement, and that the appropriate arthroplasty systems are readily available in the operating theater prior to making the incision.
Pre-Operative Planning, Templating, and Patient Positioning
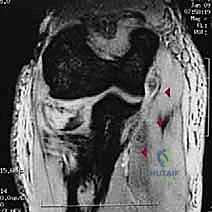
Meticulous preoperative planning is the vanguard against intraoperative complications and suboptimal outcomes. The clinical evaluation must begin with a rigorous assessment of the neurovascular status, specifically documenting the function of the posterior interosseous, median, and ulnar nerves prior to any intervention. Inspection of the soft tissue envelope is critical; medial ecchymosis is a pathognomonic sign of a concomitant MCL tear, alerting the surgeon to underlying valgus instability. Palpation of the entire forearm, including the distal radioulnar joint (DRUJ), is mandatory to rule out an Essex-Lopresti lesion. The "squeeze test" of the forearm can help elicit pain indicative of interosseous membrane disruption.
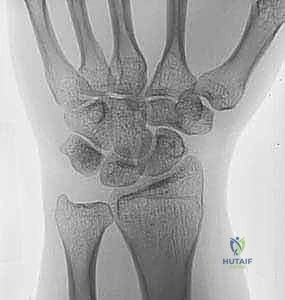
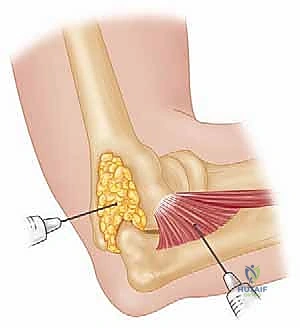
Assessing the range of motion (ROM) for mechanical blocks is a pivotal step in the clinical pathway. Because acute hemarthrosis and pain induce severe muscle guarding, a reliable examination often requires intra-articular local anesthesia. The joint can be aspirated via the lateral "soft spot" (the anconeus triangle, bordered by the radial head, lateral epicondyle, and olecranon tip) to decompress the hematoma, followed by the injection of 5 to 10 mL of local anesthetic (e.g., lidocaine or bupivacaine). Once the joint is anesthetized, the surgeon can accurately assess for a true osseous block to pronation and supination, which strongly dictates the need for surgical intervention. Furthermore, varus and valgus stress testing can be performed to evaluate collateral ligament integrity.
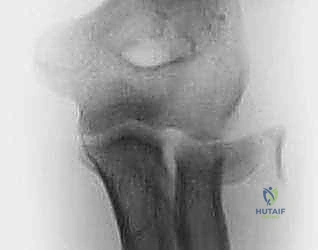
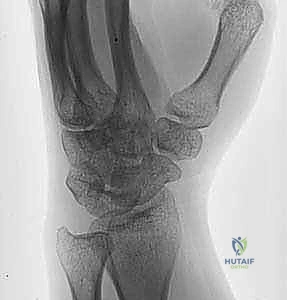
Imaging requires orthogonal anteroposterior (AP) and lateral radiographs of the elbow. However, standard views frequently underestimate the degree of comminution and articular impaction. A radiocapitellar view—obtained with the elbow flexed to 90 degrees, the forearm in neutral rotation, and the x-ray beam angled 45 degrees cephalad—provides an unobstructed, tangential projection of the radiocapitellar articulation. For any complex fracture, a non-contrast CT scan with 3D reconstructions is considered the standard of care. CT imaging allows the surgeon to map the fracture lines, quantify the number of articular fragments, identify central impaction, and template the size and trajectory of planned implants. Templating should include a selection of low-profile minifragment plates (1.5 mm to 2.4 mm), headless compression screws, and threaded K-wires.
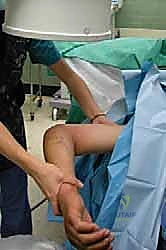
Patient positioning is optimized for both surgical access and fluoroscopic evaluation. The patient is typically placed in the supine position with the operative arm extended onto a radiolucent hand table. A sterile tourniquet is applied high on the brachium. The C-arm fluoroscopy unit is positioned parallel to the table, coming in from the head or the axilla, allowing for unhindered AP and lateral imaging by simply rotating the shoulder, rather than manipulating the injured elbow. Alternatively, for complex cases requiring simultaneous medial and lateral approaches (e.g., terrible triad injuries), the lateral decubitus position with the arm draped over a post provides excellent access to both sides of the joint, though it requires a more cumbersome fluoroscopy setup.
Step-by-Step Surgical Approach and Fixation Technique
The surgical approach to the radial head must balance adequate exposure of the fracture with the preservation of critical soft tissue stabilizers and the protection of the posterior interosseous nerve (PIN). The two primary lateral approaches are the Kocher (posterolateral) and the Kaplan (direct lateral). The Kocher approach utilizes the internervous plane between the anconeus (radial nerve) and the extensor carpi ulnaris (ECU, posterior interosseous nerve). This approach provides excellent exposure of the posterior aspect of the radial head and capitellum but poses a slightly higher risk of iatrogenic injury to the LUCL if dissection strays too far anteriorly or distally. The Kaplan approach exploits the interval between the extensor digitorum communis (EDC) and the extensor carpi radialis brevis (ECRB). While it provides more direct anterior access, it places the PIN at greater risk during distal dissection.
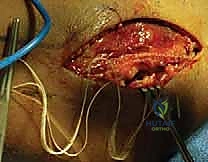
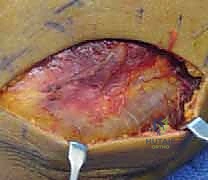
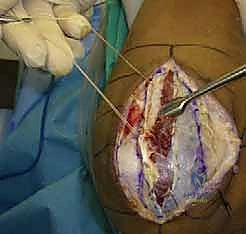
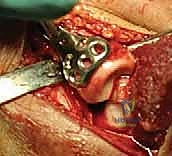
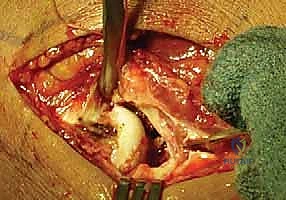
Once the superficial interval is developed, the common extensor origin is retracted. The capsule and the annular ligament are identified. An arthrotomy is performed, typically longitudinally, taking care to incise anterior to the LUCL to preserve lateral column stability. The joint is thoroughly irrigated to evacuate the fracture hematoma, and any loose osteochondral debris is meticulously removed. The fracture pattern is then directly visualized and correlated with the preoperative CT scan. The forearm is pronated to draw the PIN anteriorly and away from the surgical field. Careful subperiosteal dissection is performed only as necessary to visualize the fracture lines; circumferential stripping of the radial neck must be strictly avoided to preserve the tenuous intraosseous blood supply and prevent AVN.
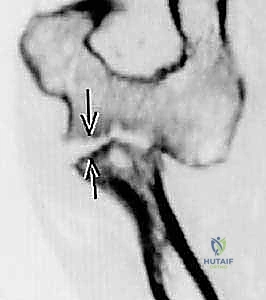
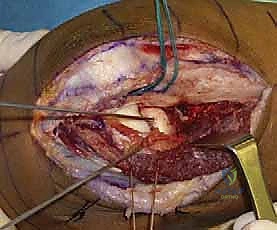
Reduction is achieved using fine dental picks, Freer elevators, and small pointed reduction forceps. Kirschner wires (0.045-inch or 0.062-inch) are frequently utilized as "joysticks" to manipulate fragments and provisionally hold the reduction. The primary goal is the anatomic restoration of the articular "dish" to ensure congruent tracking against the capitellum. Central impacted fragments must be elevated, and the resulting metaphyseal void may require grafting with cancellous allograft or autograft to support the articular surface. Once provisional reduction is achieved, it is verified under direct visualization and orthogonal fluoroscopy. The surgeon must rotate the forearm through a full arc of pronation and supination to ensure that no hardware impinges on the lesser sigmoid notch of the ulna.
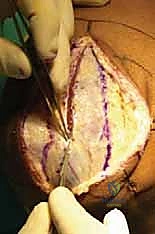
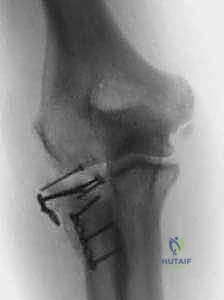
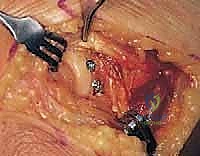
Definitive fixation strategies are dictated by the fracture morphology. For isolated, large marginal fragments (Mason II), headless compression screws or countersunk minifragment screws (1.5 mm or 2.0 mm) directed from the articular surface into the intact neck are highly effective. For fractures exiting the radial neck (Mason III), plate fixation is usually required. A low-profile, pre-contoured radial head plate is applied to the "safe zone." The safe zone is defined as the 90- to 110-degree arc of the radial head that does not articulate with the ulna during forearm rotation. It can be identified intraoperatively by marking the middle of the radial head with the forearm in neutral, then maximally supinating and pronating the forearm; the area that remains visible laterally and does not contact the ulna is the safe zone. Screws placed in the articular surface must be meticulously countersunk beneath the cartilage to prevent catastrophic gouging of the capitellum.
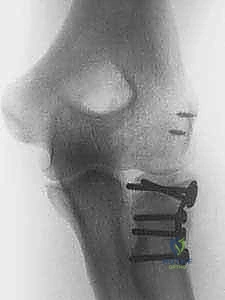
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, ORIF of the radial head is fraught with potential complications. The most ubiquitous complication is postoperative stiffness, affecting up to 30-50% of patients to some degree. The elbow joint is exquisitely sensitive to trauma and immobilization, rapidly developing capsular contractures. Loss of terminal extension (typically 10 to 15 degrees) is incredibly common and often permanent, though usually well-tolerated functionally. Heterotopic ossification (HO) can also severely restrict motion, particularly in cases involving delayed surgery, severe soft tissue trauma, or concurrent central nervous system injury. Prophylaxis with non-steroidal anti-inflammatory drugs (NSAIDs), such as indomethacin, or localized radiation therapy is considered in high-risk patients.
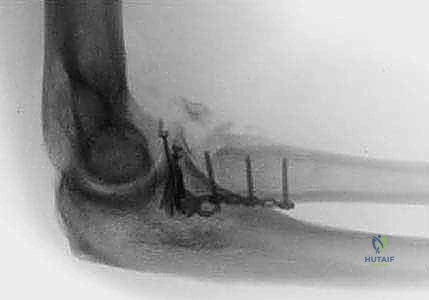
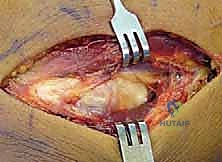
Neurologic injury, specifically to the posterior interosseous nerve (PIN), is a devastating complication with an incidence ranging from 1% to 5%. It typically presents as an inability to extend the digits at the metacarpophalangeal joints and a radial deviation of the wrist during extension (due to preservation of the ECRL, which is innervated proximal to the PIN). The vast majority of these injuries are traction neuropraxias resulting from overly aggressive retraction during the surgical approach. These typically resolve spontaneously within 3 to 6 months. However, if a direct transection is suspected (e.g., aberrant retractor placement or errant drill bit), early surgical exploration and microsurgical repair or nerve grafting are mandated.
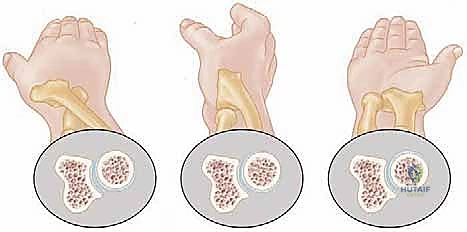
Hardware impingement and failure are significant risks, particularly if implants are placed outside the safe zone or if articular screws are not adequately countersunk. Prominent hardware will abrade the capitellum or the lesser sigmoid notch, leading to severe pain, mechanical blocks to rotation, and rapid onset of early osteoarthritis. Avascular necrosis (AVN) of the radial head occurs in up to 10% of ORIF cases, secondary to the disruption of the tenuous retrograde blood supply. While some cases of AVN remain asymptomatic, structural collapse requires intervention. Nonunion of the radial neck is less common but can occur with inadequate fixation or severe comminution.
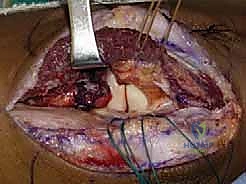
Salvage management for failed ORIF—whether due to painful hardware, AVN with collapse, nonunion, or post-traumatic arthrosis—involves conversion to a radial head arthroplasty. Excision of the radial head as a salvage procedure should only be entertained if the surgeon has definitively proven that the MCL, LUCL, and interosseous membrane are completely intact and competent. In the setting of any chronic instability, radial head replacement with a modular, metallic implant is required to restore the lateral column strut and secondary valgus stability. If severe stiffness is the primary complaint without hardware failure, an open or arthroscopic capsular release and hardware removal may be indicated once fracture union is confirmed.
| Complication | Estimated Incidence | Primary Etiology / Risk Factor | Salvage / Management Strategy |
|---|---|---|---|
| Post-op Stiffness | 30 - 50% | Prolonged immobilization, capsular contracture | Aggressive PT, dynamic splinting, surgical capsular release |
| Hardware Impingement | 5 - 15% | Implants outside "safe zone", inadequate countersinking | Hardware removal (after union), Arthroplasty if cartilage destroyed |
| Avascular Necrosis (AVN) | 5 - 10% | Extensive soft tissue stripping, high-energy trauma | Observation if asymptomatic; Radial Head Arthroplasty if collapsed/painful |
| PIN Neuropraxia | 1 - 5% | Retractor traction, supinated forearm during approach | Observation (EMG at 6 weeks if no improvement), exploration if transected |
| Heterotopic Ossification | 5 - 10% | Severe trauma, delayed surgery, head injury | Indomethacin prophylaxis, delayed surgical excision once mature |
Phased Post-Operative Rehabilitation Protocols
The overarching goal of postoperative rehabilitation following radial head ORIF is to navigate the delicate balance between protecting the fragile osseous fixation and preventing the rapid onset of debilitating elbow stiffness. The protocol must be highly individualized, dictated by the security of the internal fixation achieved intraoperatively and the presence of any concomitant ligamentous repairs. Communication between the orthopedic surgeon and the physical therapist is paramount. If rigid fixation was achieved in an isolated fracture, the rehabilitation can be surprisingly aggressive.
Phase I: Immediate Post-Operative Phase (Days 1 to 14)
The primary objectives in the initial phase are edema control, pain management, and the initiation of early, protected range of motion. The patient is typically placed in a bulky Jones dressing and a posterior splint at 90 degrees of flexion with the forearm in neutral rotation immediately post-op. Within 3 to 5
Clinical & Radiographic Imaging Archive