Intravenous Regional & Brachial Plexus Anesthesia Guide
Key Takeaway
Intravenous regional anesthesia (Bier block) and brachial plexus blocks are essential techniques for upper extremity orthopedic surgery. This comprehensive guide details the precise indications, step-by-step methodologies, and safety protocols required for optimal patient outcomes. From double-tourniquet management in Bier blocks to ultrasound-guided interscalene, supraclavicular, and axillary approaches, mastering these regional anesthetic modalities minimizes systemic complications while maximizing intraoperative efficacy and postoperative pain control.
INTRODUCTION TO REGIONAL ANESTHESIA IN ORTHOPAEDICS
Regional anesthesia has revolutionized the practice of operative orthopaedics, offering profound intraoperative analgesia, reducing the need for systemic opioids, and facilitating Enhanced Recovery After Surgery (ERAS) protocols. For operations on the hand and upper extremity, four primary methods of regional anesthesia are in widespread clinical use:
1. Brachial plexus blocks (utilizing interscalene, supraclavicular, infraclavicular, or axillary approaches).
2. Intravenous regional anesthesia (IVRA, commonly known as the Bier block).
3. Peripheral nerve blocks distal to the axilla (including targeted blocks of the median, radial, ulnar, and digital nerves).
4. Local infiltration of anesthetic agents (such as Wide Awake Local Anesthesia No Tourniquet - WALANT).
The successful application of regional anesthesia requires meticulous preoperative planning. Sufficient time must be allocated in the immediate preoperative period for patient preparation, administration of the anesthetic agent, and allowance for the block to achieve full surgical density before the skin incision is made. Furthermore, the patient should be satisfactorily sedated (e.g., with midazolam and fentanyl) prior to the block to minimize anxiety and improve cooperation, though simple distal nerve blocks at the wrist or fingers often require minimal to no premedication.
Surgical Warning: Regional anesthesia is generally less satisfactory in pediatric populations or in extremely nervous, anxious, or uncooperative adults. It is strictly contraindicated in patients with documented, true anaphylactic allergies to local anesthetic agents and must be approached with extreme caution in patients on active anticoagulant therapy.
INTRAVENOUS REGIONAL ANESTHESIA (BIER BLOCK)
Intravenous regional anesthesia (IVRA), pioneered by August Bier, is a highly reliable and efficient technique utilizing a double tourniquet system. It is particularly useful for superficial and deep procedures of the hand, wrist, and forearm that are of relatively short duration (typically 60 to 90 minutes). Beyond 90 minutes, tourniquet pain becomes intolerable for the conscious patient, and the risk of tissue ischemia increases significantly.
Indications and Patient Selection
IVRA is ideal for procedures such as closed reduction of distal radius fractures, carpal tunnel release, ganglion cyst excision, and foreign body removal. It is highly advantageous in patients with unstable cardiac, severe pulmonary, or advanced renal comorbidities, where the physiologic stress and hemodynamic instability associated with general anesthesia pose an unacceptable risk.
However, IVRA may be technically difficult or contraindicated in patients with severe joint contractures that prevent proper exsanguination, or in patients with poor venous access.
Pharmacologic Principles
The standard anesthetic agent of choice is lidocaine. Bupivacaine is strictly contraindicated for IVRA due to its high potential for irreversible cardiotoxicity upon tourniquet deflation.
* Dosage: In most adult situations, 30 to 60 mL of 0.5% lidocaine provides sufficient and safe anesthesia.
* Calculation: The exact dosage must be meticulously calculated based on the patient’s age, lean body weight, and cardiovascular status (typically 3 mg/kg of lidocaine without epinephrine).
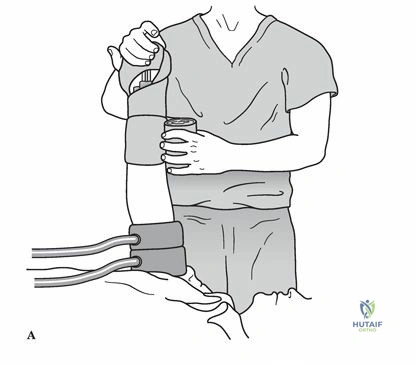
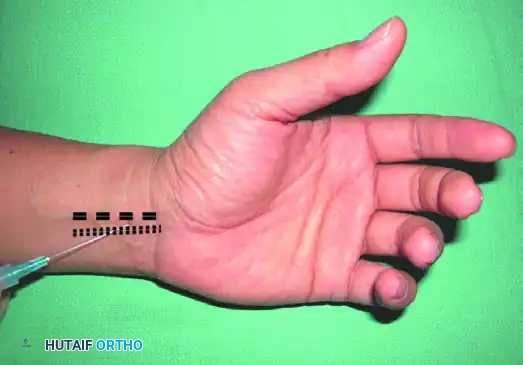
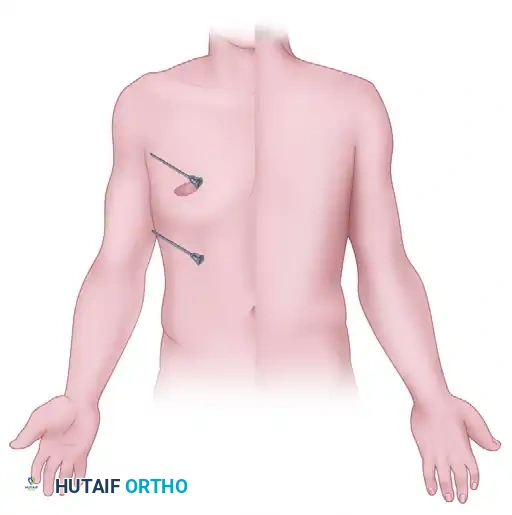
Step-by-Step Surgical Technique

Clinical setup demonstrating the intravenous regional anesthesia (Bier block) technique, highlighting the double tourniquet system and distal venous access.
- Preparation: The patient must be satisfactorily premedicated. Crucially, a secure intravenous infusion line must be established in the contralateral arm for the administration of resuscitative medications (e.g., 20% lipid emulsion) in the event of Local Anesthetic Systemic Toxicity (LAST).
- Venous Access: A small-gauge intravenous cannula (usually 20G or 22G) is inserted into a distal vein of the operative extremity, preferably on the dorsum of the hand.
- Exsanguination: The limb is elevated and tightly wrapped with an Esmarch bandage from the fingertips to the distal edge of the double tourniquet to exsanguinate the venous system.
- Tourniquet Inflation: The proximal tourniquet is inflated to a pressure of at least 100 mm Hg greater than the patient's systolic blood pressure (usually between 250 to 300 mm Hg). The Esmarch bandage is then removed.
- Injection: Using strict sterile technique, the anesthesiologist or surgeon slowly introduces the previously determined volume of 0.5% lidocaine into the indwelling distal cannula. Satisfactory surgical anesthesia is typically obtained within 5 to 10 minutes.
- Tourniquet Management (The "Switch"): As the procedure progresses (usually around 30-45 minutes), the unanesthetized tissue under the proximal tourniquet will begin to cause ischemic discomfort. At this point, the distal tourniquet (which is situated over tissue that is now anesthetized) is inflated. Only after the distal tourniquet is fully inflated and secure should the proximal tourniquet be deflated.
Clinical Pearl: The tourniquet must remain inflated for a minimum of 30 minutes after the injection of the anesthetic agent, regardless of how quickly the surgical procedure is completed. Premature deflation releases a massive bolus of local anesthetic into the systemic circulation, precipitating LAST.
Forearm Tourniquet Variation
Recent literature suggests the use of a forearm tourniquet for IVRA in highly selected distal hand procedures. The reported advantages include enhanced safety due to a lower required anesthetic dose, preservation of proximal hand and arm motor function, and a reduced risk of systemic complications.
Complications of IVRA
Complications are primarily related to equipment failure (premature tourniquet deflation) or overdosage, leading to LAST. Reported systemic reactions include:
* Neurologic: Perioral numbness, metallic taste, vertigo, nystagmus, unconsciousness, and generalized tonic-clonic seizures.
* Cardiovascular: Bradycardia, conduction blocks, ventricular arrhythmias, and catastrophic cardiac arrest.
BRACHIAL PLEXUS BLOCKS: ANATOMY AND MODALITIES
When surgical duration exceeds 90 minutes, or when profound postoperative analgesia is required, brachial plexus blocks are the gold standard. The traditional approaches for administering anesthesia to the major components of the brachial plexus include the interscalene, supraclavicular, infraclavicular, and axillary routes.
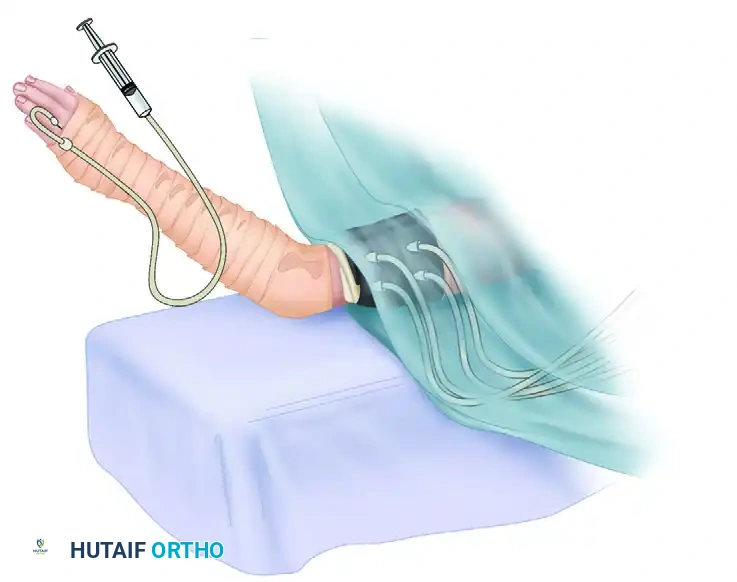
FIGURE 64-5 Distribution of brachial plexus blocks. A, Interscalene. B, Supraclavicular. C, Infraclavicular axillary, single injection. D, Axillary, multiple injections and high humeral block.
Ultrasound vs. Neurostimulation
Historically, needle placement for brachial plexus blocks relied on anatomic landmarks, fascial "pops," and nerve localization via a peripheral nerve stimulator. However, modern orthopaedic anesthesia relies heavily on ultrasound guidance. A comprehensive meta-analysis of 13 studies comparing neurostimulation with ultrasound-guided blocks conclusively demonstrated that ultrasound guidance:
* Significantly increases the success rate of the block.
* Reduces the time required to perform the procedure.
* Results in a faster onset of surgical anesthesia.
* Drastically decreases the risk of inadvertent vascular puncture and intraneural injection.
SPECIFIC BRACHIAL PLEXUS APPROACHES
1. Interscalene Block
The interscalene block targets the roots and trunks of the brachial plexus (C5-C7) as they pass between the anterior and middle scalene muscles.
Indications: It is the ideal block for shoulder surgery (e.g., rotator cuff repair, total shoulder arthroplasty) and proximal humerus fractures. It can also be utilized for elbow surgery, though it frequently spares the ulnar nerve (C8-T1), making it less reliable for medial elbow or hand procedures.
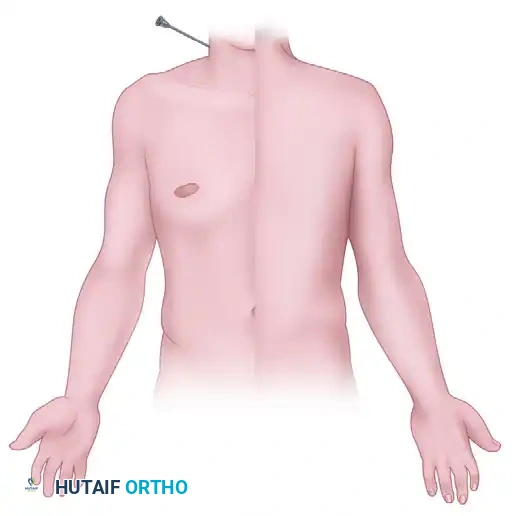
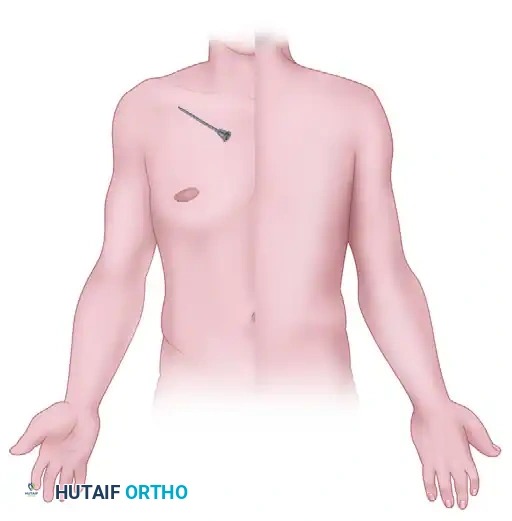
Anatomical trajectory demonstrating the needle approach for an interscalene block in the lateral neck.
Cutaneous distribution of anesthesia achieved via the interscalene approach, highlighting profound coverage of the shoulder and lateral arm.
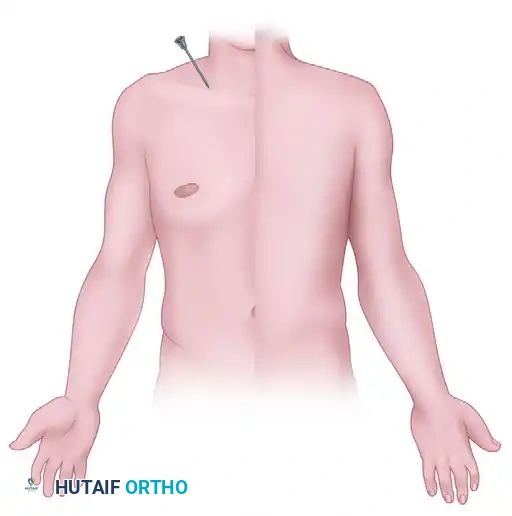
Posterior view of the anesthetic distribution following a successful interscalene block.
Pitfall: The interscalene block almost universally results in ipsilateral phrenic nerve blockade (hemidiaphragmatic paresis). It should be avoided in patients with severe chronic obstructive pulmonary disease (COPD) or contralateral diaphragmatic dysfunction.
2. Supraclavicular Block
Often referred to as the "spinal of the arm," the supraclavicular block targets the brachial plexus at the level of the trunks, where the plexus is most compactly arranged over the first rib, posterior to the subclavian artery.
Indications: It provides rapid, dense, and complete anesthesia for surgery of the upper arm distal to the shoulder, elbow, forearm, and hand.

Needle positioning for the supraclavicular block, targeting the plexus as it crosses the first rib.
Anterior cutaneous distribution of the supraclavicular block, providing comprehensive coverage of the entire upper extremity distal to the shoulder.
Posterior cutaneous distribution mapping for the supraclavicular approach.
Safety Profile: Historically, the supraclavicular route carried a notable risk of pneumothorax (1% to 5%) due to the proximity of the pleural dome. While ultrasound guidance has dramatically reduced this complication, the axillary and interscalene approaches are still generally considered safer regarding pulmonary complications.
3. Infraclavicular Block
The infraclavicular block targets the cords of the brachial plexus (lateral, posterior, and medial) as they surround the axillary artery beneath the clavicle and pectoralis minor muscle.
Indications: This approach provides excellent regional anesthesia for surgery of the elbow, forearm, wrist, and hand. It is particularly advantageous for continuous catheter placement because the chest wall provides a stable surface for securing the catheter.

Parasagittal needle trajectory for the infraclavicular block, avoiding the pleura while targeting the neurovascular bundle.

Anterior distribution of the infraclavicular block, demonstrating excellent coverage for distal extremity procedures.

Posterior distribution of the infraclavicular block.
4. Axillary Block
The axillary block targets the terminal branches of the brachial plexus (median, ulnar, and radial nerves) within the axillary sheath.
Indications: It provides anesthesia similar to that of an infraclavicular block (forearm, wrist, and hand surgery). Because it is performed distal to the pleura, there is zero risk of pneumothorax, making it exceptionally safe for patients with pulmonary pathology.

Anterior cutaneous distribution of the axillary block. Note that the musculocutaneous nerve must often be blocked separately.

Posterior cutaneous distribution of the axillary block.
Surgical Pitfall: Access to the axillary space requires the patient to abduct the arm to 90 degrees. This positioning may be excruciating or physically impossible for patients with severe acute trauma (e.g., complex elbow fractures) or chronic glenohumeral contractures. Furthermore, the musculocutaneous nerve exits the axillary sheath proximally to pierce the coracobrachialis muscle; therefore, it must be identified and blocked separately to ensure complete anesthesia of the lateral forearm.
COMPLICATION MANAGEMENT IN REGIONAL ANESTHESIA
While regional anesthesia is remarkably safe when performed with ultrasound guidance, the orthopaedic surgeon must be vigilant regarding potential complications:
- Intravascular Injection & LAST: Always aspirate prior to injection. If LAST occurs (seizures, arrhythmias), immediately halt the injection, call for help, secure the airway, and administer 20% Intravenous Lipid Emulsion (Intralipid) as the primary antidote.
- Pneumothorax: Most commonly associated with the supraclavicular approach. Patients presenting with postoperative dyspnea or pleuritic chest pain require an immediate upright chest radiograph or bedside lung ultrasound.
- Nerve Injury: Can result from direct mechanical trauma (needle laceration), intraneural injection (indicated by high injection pressures or severe sharp pain during administration), or neurotoxicity from the local anesthetic. Always inject slowly and monitor the patient's feedback if awake.
- Dissemination of Infection: Never advance a needle through infected tissue (e.g., cellulitis or abscess) to perform a block, as this can seed bacteria directly into the neurovascular sheath.
By mastering these regional anesthetic techniques, the orthopaedic surgeon and anesthesia team can collaboratively ensure optimal surgical conditions, minimize systemic morbidity, and provide superior postoperative pain control for upper extremity procedures.
📚 Medical References
- Regional anesthesia in the anticoagulated patient: defi ning the risks (the second ASRA Consensus Conference on Neuraxial Anesthesia and Anticoagulation), Reg Anesth Pain Med 28:172, 2003.
- Johnson R, Carmichael JHE, Almond HGA, et al: Deep venous thrombosis following Charnley arthroplasty, Clin Orthop Relat Res 132:24, 1978.
- Johnson R, Green JR, Charnley J: Pulmonary embolism and its prophylaxis following the Charnley total hip replacement, Clin Orthop Relat Res 127:123, 1977.
- Lassen MR, Bauer KA, Eriksson BI, et al: Postoperative fondaparinux versus preoperative enoxaparin for prevention of venous thromboembolism in elective hip-replacement surgery: a randomised double-blind comparison, Lancet 359:1715, 2002.
- Lieberman JR, Hsu WK: Prevention of venous thromboembolic disease after total hip and knee arthroplasty, J Bone Joint Surg 87A:2097, 2005.
- Pellegrini VD Jr, Clement D, Lush-Ehmann C, et al: The John Charnley Award. National history of thromboembolic disease after total hip arthroplasty, Clin Orthop Relat Res 333:27, 1996.
- Sikorski JM, Hampson WG, Staddon GE: The natural history and aetiology of deep vein thrombosis after total hip replacement, J Bone Joint Surg 63B:171, 1981.
- Skinner HB, Schulz MM: Clinical implications of thromboprophylaxis in the management of total hip and knee arthroplasty, Am J Orthop 31(9S):20, 2002.
- Turpie AG, Bauer KA, Eriksson BI, et al: Postoperative fondaparinux versus postoperative enoxaparin for prevention of venous thromboembolism after elective hip-replacement surgery: a randomised double-blind trial, Lancet 359:1721, 2002.
- White RH, Romano PS, Zhou H, et al: Incidence and time course of thromboembolic outcomes following total hip or knee arthroplasty, Arch Intern Med 158:1525, 1998.
- Hematoma Acus RW, Clark JM, Gradisar IA, et al: The use of postoperative suction drainage in total hip arthroplasty, Orthopedics 15:1325, 1992.
- Hallstrom BR, Steele JF: Postoperative course after total hip arthroplasty: wound drainage versus no drainage, Orthop Rev 21:847, 1992.
- Niskanen RO, Korkala OL, Haapala J, et al: Drainage is of no use in primary uncomplicated cemented hip and knee arthroplasty for osteoarthritis: a prospective randomized study, J Arthroplasty 15:567, 2000.
- Infection Callaghan JJ, Katz RP, Johnston RC: One-stage revision surgery of the infected hip: a minimum of 10-year follow-up study, Clin Orthop Relat Res 369:139, 1999.
- Charnley J: Postoperative infection after total hip replacement with special reference to air contamination in the operating room, Clin Orthop Relat Res 87:167, 1972.
- Di Cesare PE, Change E, Preston CF, et al: Serum interleukin-6 as a marker of periprosthetic infection following total hip and knee arthroplasty, J Bone Joint Surg 87A:1921, 2005.
- Duncan CP, Beauchamp C: A temporary antibiotic-loaded joint replacement system for management of complex infections involving the hip, Orthop Clin North Am 24:751, 1993.
- Durbhakula SM, Czajka J, Fuchs MD, et al: Spacer endoprosthesis of the treatment of infected total hip arthroplasty, J Arthroplasty 19:760, 2004.
- Etienne G, Waldman B, Rajadhyaksha AD, et al: Use of a functional temporary prosthesis in a two-stage approach to infection at the site of a total hip arthroplasty, J Bone Joint Surg 85A(suppl 4):94, 2003.
- Fitzgerald RH, Nolan DR, Ilstrup DM, et al: Deep wound sepsis following total hip arthroplasty, J Bone Joint Surg 59A:847, 1977.
- Gristina AG, Costerton JW: Bacterial adherence to biomaterials and tissue: the signifi cance of its role in clinical sepsis, J Bone Joint Surg 67A:264, 1985.
- Hecht RH, Meyers MH, Thornhill-Joynes M, et al: Reactivation of tuberculosis infection following total joint replacement: a case report, J Bone Joint Surg 65A:1015, 1983.
- Hsieh P-H, Shih C-H, Change Y-H, et al: Two-stage revision hip arthroplasty for infection: comparison between the interim use of antibiotic-loaded cement beads and a spacer prosthesis, J Bone Joint Surg 86A:1989, 2004.
- Jackson WO, Schmalzried TP: Limited role of direct exchange arthroplasty in the treatment of infected total hip replacements, Clin Orthop Relat Res 381:101, 2000.
- Joseph TN, Muitaba M, Chen AL, et al: Effi cacy of combined technetium-99m sulfur colloid/indium-111 leukocyte scans to detect infected total hip and knee arthroplasties, J Arthroplasty 16:753, 2001.
- Koo KH, Yang JW, Cho SH, et al: Impregnation of vancomycin, gentamicin, and cefotaxime in a cement spacer for two-stage cementless reconstruction in infected total hip arthroplasty, J Arthroplasty 16:882, 2001.
- Lentino JR: Prosthetic joint infections: bane of orthopedists, challenge for infectious disease specialists, Clin Infect Dis 36:1157, 2003.
- Miley GB, Scheller AD Jr, Turner RH: Medical and surgical treatment of the septic hip with one-stage revision arthroplasty, Clin Orthop Relat Res 170:76, 1982.
- Morshed S, Huffman GR, Ries MD: Extended trochanteric osteotomy for 2-stage revision of infected total hip arthroplasty, J Arthroplasty 20:294, 2005.
- Salvati EA, Gonzalez Della Valle A, Masri BA, et al: The infected total hip arthroplasty, Instr Course Lect 52:223, 2003.
- Scher DM, Pak K, Lonner JH, et al: The predictive value of indium-111 leukocyte scans in the diagnosis of infected total hip, knee, or resection arthroplasties, J Arthroplasty 15:295, 2000.
- Shih LY, Wu JJ, Yang DJ: Erythrocyte sedimentation rate and C-reactive protein values in patients with total hip arthroplasty, Clin Orthop Relat Res 225:238, 1987.
- Spangehl MJ, Masri BA, O’Connell JX, et al: Prospective analysis of preoperative and intraoperative investigations for the diagnosis of infection at the sites of two hundred and two revision total hip arthroplasties, J Bone Joint Surg 81A:672. Tsukayama DT, Estrada R, Gustilo RB: Infection after total hip arthroplasty: a study of the treatment of one hundred and six infections, J Bone Joint Surg 78A:512, 1996.
- Wentworth SJ, Masri BA, Duncan CP, et al: Hip prosthesis of antibiotic-loaded acrylic cement for the treatment of infections following total hip arthroplasty, J Bone Joint Surg 84A(suppl 2):123, 2002.
- Wilson PD, Amstutz HC, Czernieki A, et al: Total hip replacement with fi xation by acrylic cement: a preliminary study of 100 consecutive McKee-Farrar prosthetic replacements, J Bone Joint Surg 54A:207, 1972.
- Neurovascular Amstutz HC, Ma SM, Jinnah RH, et al: Revision of aseptic loose total hip arthroplasties, Clin Orthop Relat Res 170:21, 1982.
- Brown DM, McGinnis WC, Mesghali H: Neurophysiologic intraoperative monitoring during revision total hip arthroplasty, J Bone Joint Surg 84A(suppl 2):56, 2002.
- Edwards BN, Tullos HS, Noble PC: Contributory factors and etiology of
You Might Also Like