Brachial Plexus Birth Palsy: Comprehensive Surgical Guide
Key Takeaway
Brachial plexus birth palsy results from traction injuries during delivery, affecting 0.1% to 0.4% of live births. Management requires a multidisciplinary approach, beginning with conservative therapy to prevent contractures. If spontaneous recovery is absent by three months, microsurgical nerve reconstruction is indicated. Secondary orthopedic procedures, including tendon transfers and osteotomies, are essential for addressing progressive glenohumeral dysplasia and fixed contractures, ultimately restoring upper extremity biomechanics and function.
Introduction and Epidemiology
Brachial plexus palsy in the pediatric population is most frequently encountered as an obstetric traction injury sustained during birth. The reported incidence ranges from 0.1% to 0.4% of live births. While many infants experience spontaneous recovery, a significant subset develops permanent neurological deficits, muscle imbalances, and progressive osteoarticular deformities.
Numerous risk factors have been identified, primarily revolving around fetopelvic disproportion and complicated deliveries. Key risk factors include macrosomia (large birth weight), maternal diabetes, prolonged labor, difficult delivery, forceps or vacuum-assisted delivery, and a history of previous births with brachial plexopathy.
Biomechanics of Injury
The biomechanical vector of injury dictates the anatomical pattern of the plexopathy:
* Shoulder Dystocia (Cephalic Presentation): This is the most common mechanical factor resulting in an upper trunk lesion. As the shoulder is impacted behind the maternal symphysis pubis, lateral traction applied to the fetal head and neck severely stretches the C5 and C6 nerve roots.
* Breech Delivery: Traction applied to the fetal trunk while the arm is abducted often results in a stretch injury to the lower plexus (C8-T1). Conversely, a difficult arm extraction during a breech delivery can cause severe traction, leading to an avulsion injury of the upper trunk.
Anatomical Classification and Prognosis
The severity and clinical presentation of brachial plexus palsy depend entirely on which nerve roots are injured and the histopathological extent of that injury.
Narakas Classification
Narakas described four distinct groups based on anatomical involvement, which Bora later modified to correlate with the prognosis of recovery:
* Group I (Upper Plexus / Erb-Duchenne Palsy): Involves C5 and C6. This is the most common presentation (approximately 90% of patients). The supraspinatus, infraspinatus, deltoid, and biceps are most frequently paralyzed. Prognosis is generally the most favorable.
* Group II (Extended Upper Plexus): Involves C5, C6, and C7. Adds weakness to elbow, wrist, and finger extension.
* Group III (Panplexus / Mixed Palsy): Complete sensory and motor paralysis of the entire upper extremity due to severe injury across all roots (C5-T1). The arm is completely flail.
* Group IV (Lower Plexus / Klumpke Palsy): Involves C8 and T1. Characterized by paralysis of the forearm and hand intrinsic muscles. Often accompanied by an ipsilateral Horner syndrome. This type carries the worst prognosis but is exceedingly rare in isolation.
Nerve Injury Grading
Injury to the brachial plexus exists on a spectrum from transient ischemia to complete anatomical disruption.
* Seddon’s Classification:
* Neurapraxia: Conduction block without peripheral Wallerian degeneration. Complete recovery is expected.
* Axonotmesis: Disruption of the axon with Wallerian degeneration, but intact endoneurial tubes. Good recovery is anticipated as axons regenerate at 1 mm/day.
* Neurotmesis: Complete disruption of the nerve and supporting structures (neuroma-in-continuity, rupture, or root avulsion). Requires surgical intervention.
* Sunderland’s Classification: Expands Seddon’s model into five degrees, with 1st-degree being neurapraxia, 2nd to 4th-degree representing progressive connective tissue disruption (axonotmesis to neuroma-in-continuity), and 5th-degree representing complete transection or avulsion.
Clinical Evaluation and Natural History
The diagnosis of brachial plexus birth palsy is usually evident immediately postpartum. The newborn exhibits decreased spontaneous movement and asymmetry of infantile reflexes, such as the Moro reflex or the asymmetrical tonic neck reflex.
Clinical Presentation
In classic upper root involvement (Erb's Palsy), the infant assumes the "waiter's tip" posture: the shoulder is held in internal rotation and adduction, the elbow is extended (or slightly flexed), the forearm is pronated, and the wrist and fingers are flexed.

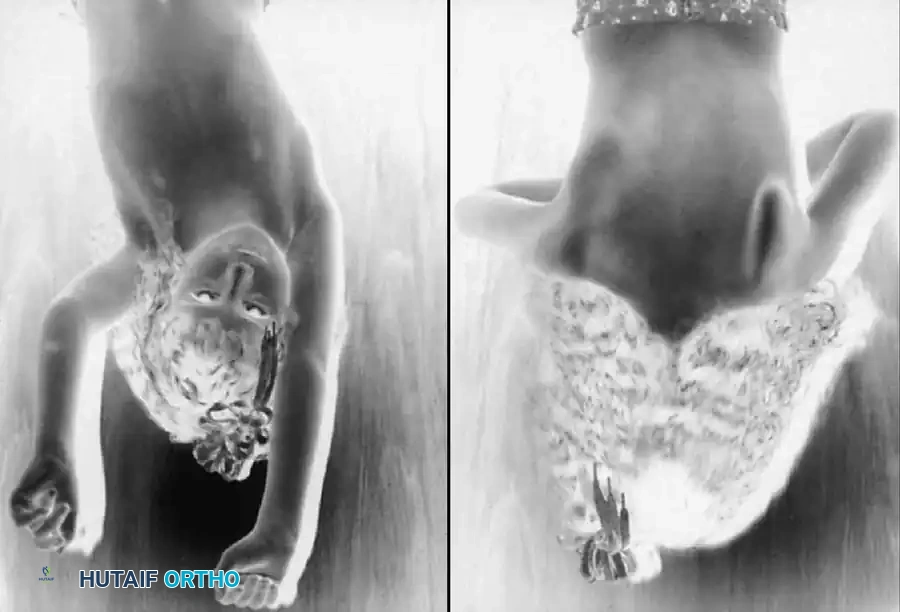
Fig. 31-56 Anterior (A) and posterior (B) views of child with brachial plexus palsy. Note winging of the scapula due to muscle imbalance and serratus anterior weakness.
In complete panplexus paralysis, the entire arm and hand are flail, and pinching produces no withdrawal reaction. Vasomotor impairment may manifest as relative paleness of the involved extremity.
Clinical Pearl: Always assess for an ipsilateral Horner syndrome (ptosis, miosis, anhidrosis). Its presence indicates a severe traction injury or avulsion of the T1 cervical sympathetic nerve roots and is a major indicator of a poor spontaneous recovery.
Differential Diagnosis: Pseudoparalysis
It is critical to differentiate true neurological palsy from pseudoparalysis caused by birth trauma or infection:
* Fractures: Clavicular fractures occur in 10% to 15% of patients with plexus palsy. Proximal humeral epiphyseal separations can also occur. Pseudoparalysis from a fracture should resolve within 10 to 21 days as the bone stabilizes. If limited motion persists beyond 1 month of age, a concomitant brachial plexus palsy is highly likely.
* Septic Arthritis: A septic shoulder in an infant causes painful pseudoparalysis. This is differentiated by systemic signs of illness (fever, elevated inflammatory markers) and resolution following appropriate antibiotic therapy and joint washout.
Progressive Glenohumeral Deformity
If recovery is incomplete, characteristic deformities develop promptly due to persistent muscle imbalance. The strong, innervated internal rotators (subscapularis, pectoralis major, latissimus dorsi) overpower the weak or paralyzed external rotators (infraspinatus, teres minor).
The shoulder becomes fixed in flexion, internal rotation, and slight abduction. Active abduction decreases, and external rotation disappears. Over time, this dynamic imbalance leads to structural osseous changes. The humeral head flattens against the glenoid, leading to progressive posterior subluxation, increased glenoid retroversion, and eventual posterior dislocation.
Waters classified this progressive glenohumeral deformity into seven types based on CT and MRI findings, guiding secondary surgical interventions:
* Type I: Normal glenohumeral joint.
* Type II: Minimal glenoid hypoplasia (>5 degrees increased retroversion).
* Type III: Posterior subluxation of the humeral head.
* Type IV: Development of a false glenoid (biconcave glenoid).
* Type V: Posterior flattening of the humeral head.
* Type VI: Infantile dislocation.
* Type VII: Proximal humeral growth arrest.
Diagnostic Imaging and Electrophysiology
Evaluation of the brachial plexus injury requires a multimodal approach:
1. Radiographs: AP and lateral views of the shoulder and clavicle to rule out fractures.
2. Advanced Imaging (CT/MRI): MRI is the gold standard for evaluating the non-ossified pediatric shoulder joint and assessing glenohumeral dysplasia (Waters classification). Combined CT myelography and MRI are highly reliable for identifying nerve root avulsions. The presence of large pseudomeningoceles or diverticula strongly indicates preganglionic root avulsions, which are not amenable to direct repair and require nerve transfers.
3. Electromyography (EMG) and Nerve Conduction Studies (NCS): Useful for establishing a baseline and detecting early signs of reinnervation (fibrillation potentials vs. nascent motor unit potentials). However, clinical examination remains the primary driver for surgical decision-making.
Primary Management: Conservative and Microsurgical
Conservative Treatment
Minimal injuries (neurapraxia) respond excellently to conservative treatment. While full recovery may require up to 18 months, significant recovery is usually evident within the first 3 months.
The primary aim of initial treatment is the prevention of joint contractures. Gentle, consistent passive range-of-motion (ROM) exercises are initiated immediately to maintain:
* Full extension of the fingers, hand, and wrist.
* Full pronation and supination of the forearm.
* Full extension of the elbow.
* Full abduction, extension, and external rotation of the shoulder.
Surgical Warning: Most authors strongly discourage rigid splinting in the neonatal period, as it can exacerbate joint stiffness. However, functional bracing may be utilized later to encourage early hand use and prevent fixed contractures.
Indications for Primary Microsurgery
The timing of microsurgical intervention remains a topic of debate, generally ranging from 3 to 6 months of age. The consensus indications for early microsurgical exploration and repair (neurolysis, nerve grafting, or nerve transfers) include:
1. Absence of Biceps Function: Lack of anti-gravity biceps recovery by 3 months of age.
2. Poor Toronto Score: A score of less than 3.5 at 3 months. The Toronto scale grades elbow flexion, elbow extension, wrist extension, finger extension, and thumb extension (0 = no motion, 1 = limited, 2 = normal; max score 12).
3. Total Plexopathy with Horner Syndrome: Indicates severe avulsion injuries requiring early nerve transfers.
Secondary Reconstructive Procedures
Surgery in unresolved brachial plexus palsy is directed toward improving shoulder function, correcting joint contractures, and addressing glenohumeral dysplasia. The Modified Mallet System is widely used to assess upper trunk function and guide reconstructive decision-making.
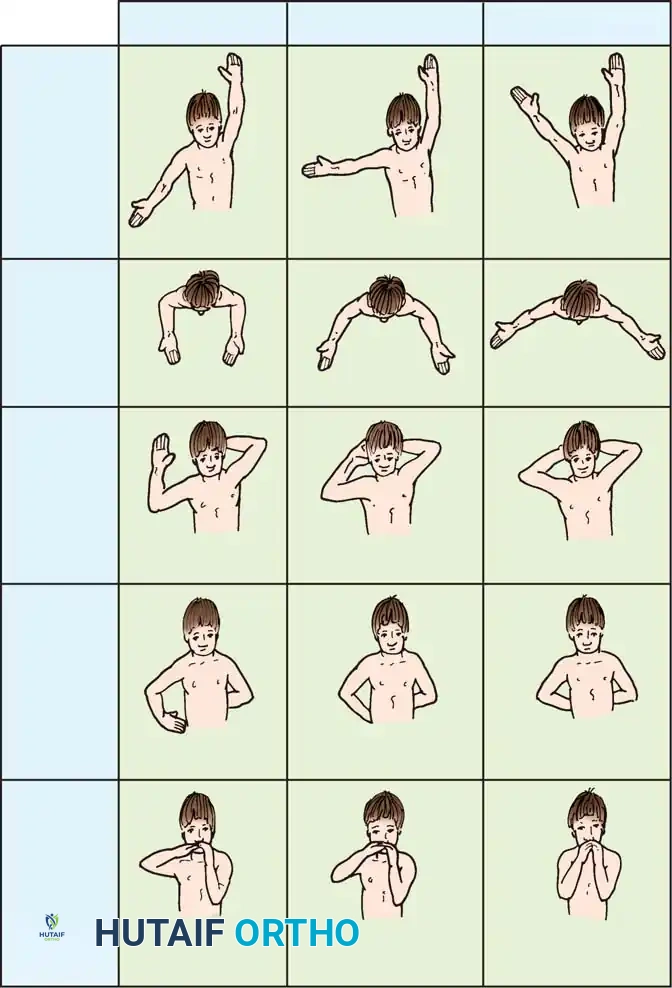
Fig. 31-57 Modified Mallet classification for assessing upper trunk function in patients with brachial plexus birth palsy. Grade I represents no function, and grade V represents normal function. This system evaluates global abduction, external rotation, hand-to-neck, hand-on-spine, and hand-to-mouth movements.
Management of Internal Rotation Contractures of the Shoulder
Children with residual upper trunk palsy develop severe internal rotation contractures due to the unopposed pull of the subscapularis and pectoralis major.
Anterior Shoulder Releases
For mild-to-moderate internal rotation contractures without severe bony deformity (Waters Type I or II), soft tissue releases are indicated.
* Sever Procedure: Historically recommended, involving an anterior release of the subscapularis and pectoralis major.
* Carlioz Procedure: Carlioz and Brahimi described a highly effective release of the subscapularis muscle at its origin on the anterior scapula, accessed through a transverse incision in the posterior axillary fold. This extraperiosteal release avoids the anterior capsule, reducing the risk of anterior instability.
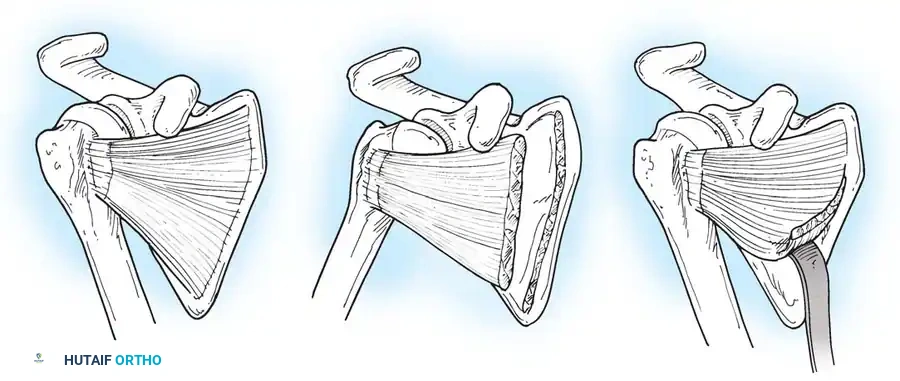
Fig. 31-58 Carlioz technique. A, Disinsertion of the subscapularis muscle at its medial border. B, At the end of disinsertion, external rotation is freed. C, Extraperiosteal release is complete, allowing the muscle to slide laterally.
Alternatively, an anterior approach can be utilized to lengthen the contracted anterior structures directly.
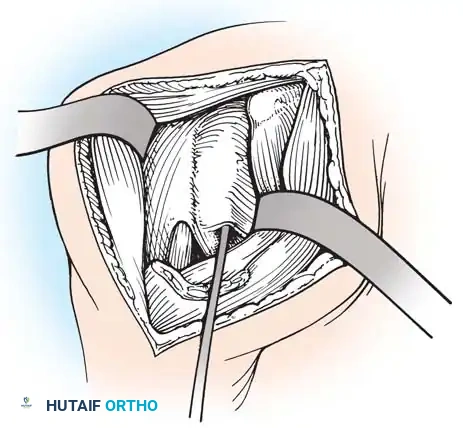
Fig. 31-60 Anterior shoulder release for internal rotation contracture. The subscapularis and anterior capsule are carefully released or lengthened to restore external rotation.
Tendon Transfers for Active External Rotation
If the child lacks active external rotation and abduction but has a congruent joint (Waters Type I, II, or mild III), tendon transfers are combined with anterior releases. The most common transfer is the Hoffer Procedure, which transfers the latissimus dorsi and teres major to the rotator cuff (infraspinatus/teres minor insertion).
Surgical Steps for Latissimus Dorsi/Teres Major Transfer:
1. Positioning: Lateral decubitus with the affected arm draped free.
2. Incision & Exposure: A posterior axillary incision is made. The latissimus dorsi and teres major are identified and mobilized.
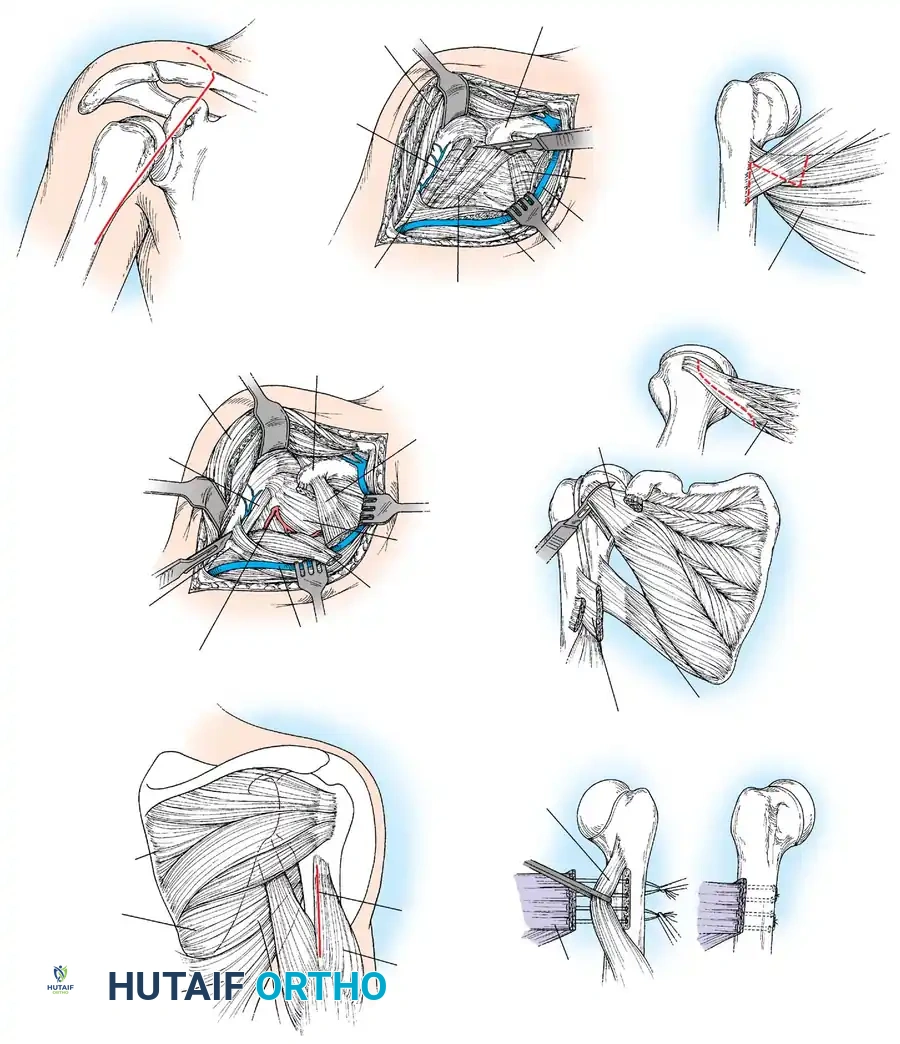
Surgical approach and exposure of the latissimus dorsi and teres major tendons, isolating them from the neurovascular bundle.
- Tendon Harvest: The tendons are detached from their humeral insertions near the bicipital groove.
- Transfer: The tendons are passed posterior to the humerus and attached to the posterosuperior rotator cuff.
Routing of the latissimus dorsi and teres major tendons posteriorly to act as external rotators and abductors.
- Fixation: The arm is held in maximum external rotation and abduction while the tendons are sutured into the infraspinatus tendon or directly to the greater tuberosity.
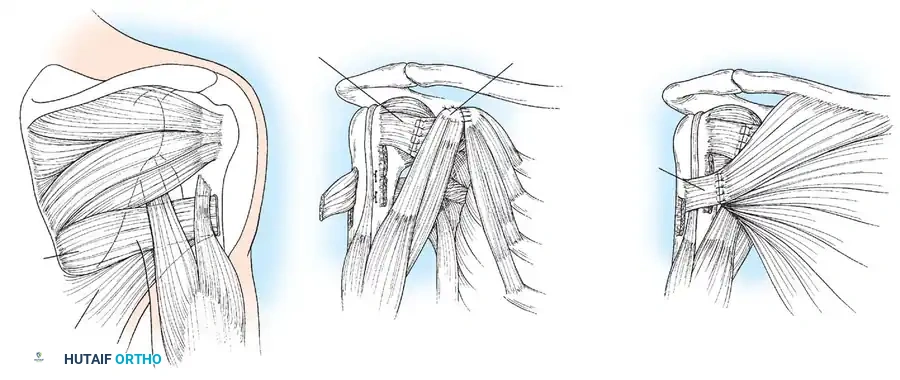
Final fixation of the transferred tendons to the rotator cuff footprint, securing the arm in the corrected position.
Humeral Osteotomy
For patients with advanced glenohumeral deformities (Waters Type IV or V) or severe fixed rotation contractures in older children, soft tissue procedures alone will fail. A proximal humeral derotation osteotomy is indicated. The humerus is osteotomized externally rotated 20 to 30 degrees, and fixed with a plate or crossed K-wires, placing the hand in a functional position for activities of daily living.
Management of Elbow and Forearm Deformities
Elbow flexion and forearm supination deformities frequently occur in lower trunk (Klumpke) or mixed brachial plexus lesions. The pathophysiology involves weak or absent triceps, pronator teres, and pronator quadratus muscles, combined with an intact, unopposed biceps muscle.
This dynamic imbalance creates a progressive elbow flexion and supination contracture. Over time, the interosseous membrane contracts, and the radial head may dislocate anteriorly or laterally.
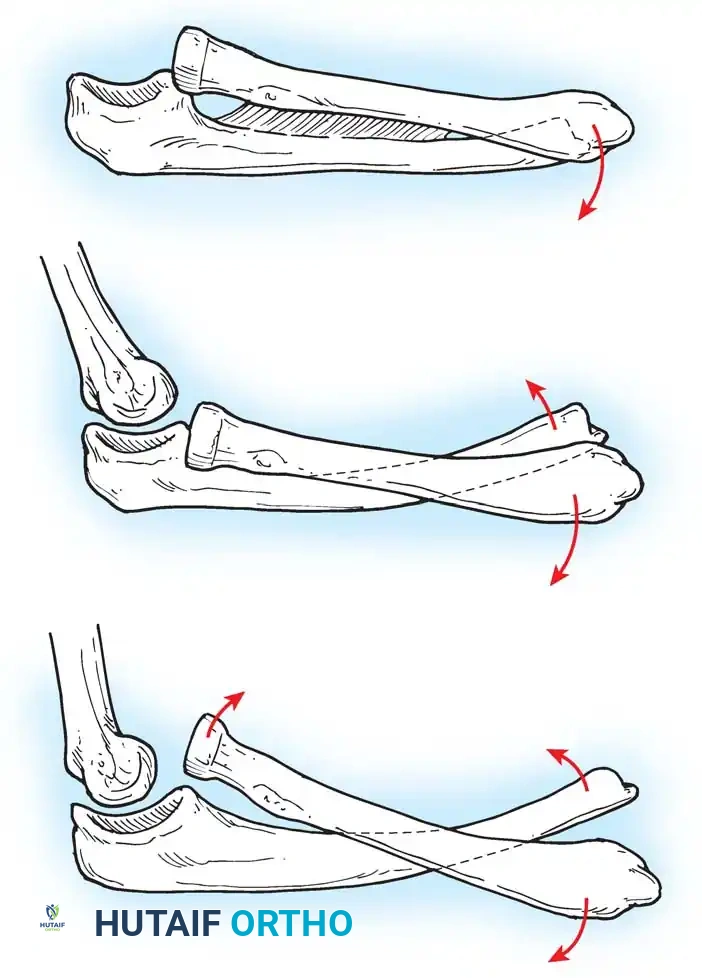
Fig. 31-59 Physiopathology of supination deformity and progressive deformity with growth. A, Simple contracture with supination of the radius (1) and contracture of the interosseous membrane (2). B, Volar dislocation of the distal epiphysis of the ulna. C, Volar dislocation of the distal ulnar epiphysis and head of the radius.
Surgical Correction of Supination Deformity
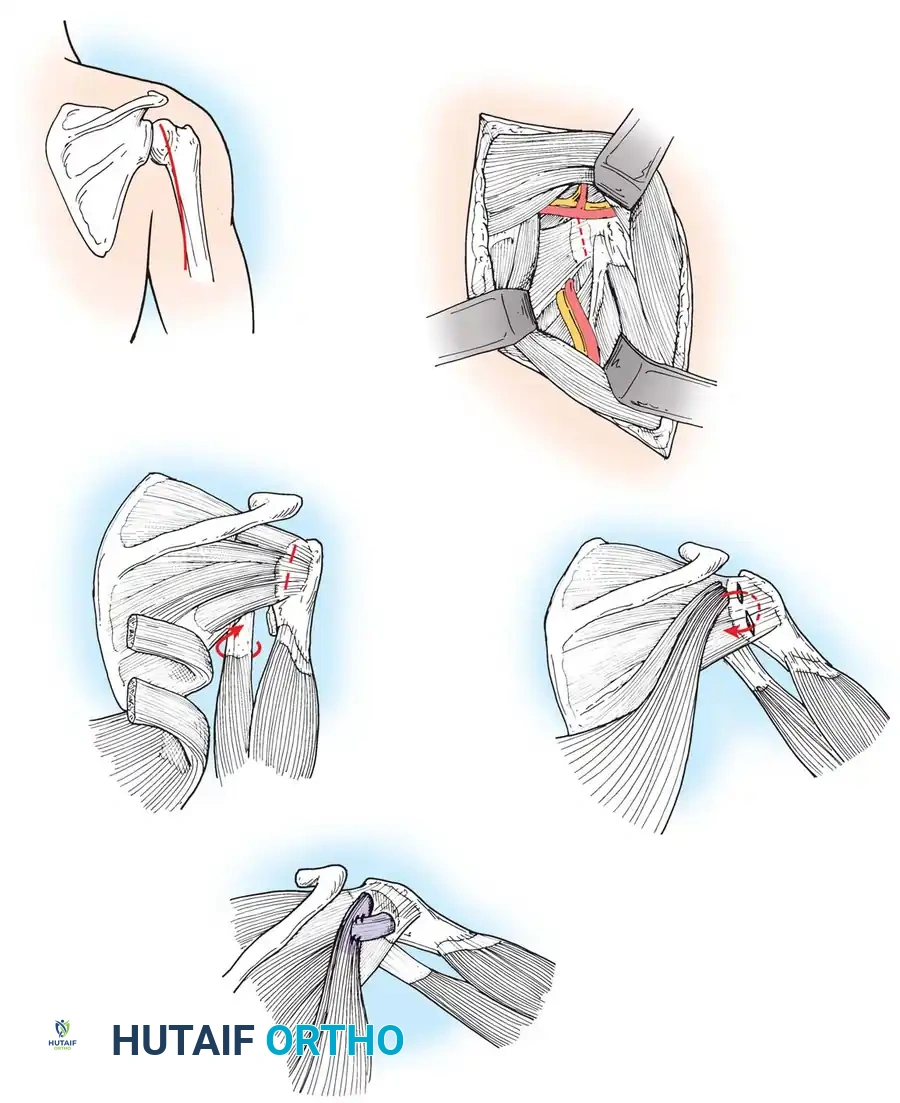
To correct this, the deforming force of the biceps must be neutralized or reversed:
* Biceps Rerouting (Zancolli Technique): The biceps tendon is Z-lengthened. The distal stump is rerouted around the radial neck from medial to lateral, converting its mechanical action from a supinator to a pronator. This simultaneously improves elbow extension and active forearm pronation.
* Interosseous Membrane Release: In the presence of a rigid, fixed supination contracture, Zancolli recommended performing a simultaneous release of the interosseous membrane to allow the radius to rotate over the ulna into pronation.
Postoperative Protocol and Rehabilitation
Meticulous postoperative care is critical to the success of secondary reconstructive procedures.
- Immobilization: Following shoulder tendon transfers or releases, an abduction splint (or shoulder spica cast) is applied in the operating room. The shoulder is immobilized in 90 degrees of abduction and mild external rotation.
- Duration: The splint is worn constantly for the first 4 to 6 weeks to allow for tendon-to-bone or tendon-to-tendon healing. Following this, it is worn intermittently (usually at night) for an additional 4 to 6 weeks.
- Rehabilitation: Active-assisted and active ROM exercises are initiated once continuous immobilization is discontinued. Physical therapy focuses on neuromuscular re-education of the transferred muscles (e.g., training the latissimus dorsi to fire during external rotation). Therapy is continued for 6 to 12 months until maximal functional improvement is achieved.
Clinical Pearl: The success of tendon transfers relies heavily on the child's cognitive ability to participate in postoperative rehabilitation. Therefore, these secondary procedures are typically delayed until the child is at least 2 to 4 years of age, ensuring compliance with complex motor retraining protocols.
📚 Medical References
- brachial plexus palsy: a comparison of functional results, J Bone Joint Surg 86B:692, 2004.
- Chandler RW: Glenohumeral arthrodesis, Tech Orthop 3:65, 1989.
- Charnley J: Compression arthrodesis of the ankle and shoulder, J Bone Joint Surg 33B:180, 1951.
- Charnley J, Houston JK: Compression arthrodesis of the shoulder, J Bone Joint Surg 46B:614, 1964.
- Cheng EY, Gebhardt MC: Allograft reconstructions of the shoulder after bone tumor resections, Orthop Clin North Am 22:37, 1991.
- Clare DJ, Wirth MA, Groh GI, et al: Current concepts review:
You Might Also Like