Peripheral Nerve Decompression: Lateral Femoral Cutaneous and Common Peroneal Nerve Masterclass
Key Takeaway
This masterclass guides fellows through the intricate surgical decompression of the Lateral Femoral Cutaneous Nerve (LFCN) and Common Peroneal Nerve (CPN). We meticulously detail preoperative planning, precise patient positioning, comprehensive surgical anatomy, and granular intraoperative execution with real-time directives. Essential pearls, critical pitfalls, and robust postoperative management strategies are emphasized to optimize outcomes and prevent complications.
Comprehensive Introduction and Patho-Epidemiology
The surgical management of peripheral nerve pathology represents one of the most technically demanding and intellectually rigorous sub-disciplines within orthopedic surgery. Today, we are diving deep into the nuanced, microscopic world of peripheral nerve decompression, specifically focusing on two frequently entrapped and highly morbid nerves: the Lateral Femoral Cutaneous Nerve (LFCN) and the Common Peroneal Nerve (CPN). Unlike primary orthopedic procedures such as arthroplasty or fracture fixation, a significant proportion of peripheral nerve surgery is inherently reoperative. We are frequently tasked with addressing severe neuropathy in patients who have already undergone multiple previous interventions, experienced catastrophic trauma, or developed dense, unyielding scar tissue. This reality demands an unparalleled level of precision, an encyclopedic understanding of anatomical variations, and intraoperative adaptability from the operating surgeon.
To master the care of peripheral nerve problems, one must cultivate an intimate, microscopic knowledge of nerve pathology. This goes far beyond simple gross anatomy; it requires a profound appreciation for the myriad anatomic variations that dictate nerve routing, a keen diagnostic eye for the subtle patterns of damage and entrapment that follow trauma or common operative procedures, and specialized microsurgical techniques for the delicate manipulation of damaged neural tissue. It is a fundamental tenet of peripheral nerve surgery that these structures are not merely inert electrical conduits. They are highly complex, living tissues endowed with a robust intrinsic blood supply, capable of remarkable regeneration when provided with an optimized anatomical environment, yet equally capable of generating debilitating, refractory neuropathic pain when mechanically or vascularly compromised.
Peripheral nerve injury generally becomes clinically symptomatic under two primary conditions: when a critical physiological function is lost (manifesting as profound motor weakness or dense sensory numbness) or, more frequently in the setting of chronic entrapment, when agonizing paresthesia, dysesthesia, and neuropathic pain replace normal afferent sensory signaling. The pathophysiology of this dysfunction can typically be categorized into three distinct mechanisms. First, accidental nerve transection or severe direct crush injuries lead to immediate, catastrophic dysfunction, triggering Wallerian degeneration and frequently resulting in painful terminal neuroma formation if axonal sprouting is blocked by scar tissue. Second, dense perineurial scar formation—often arising from prior surgery, hematoma, or localized trauma—can circumferentially engulf a peripheral nerve, leading to dynamic compression and tethering during normal limb movement. Symptomatic relief in these complex cases is only achieved through meticulous surgical neurolysis, freeing the nerve from its fibrotic prison while preserving its delicate vascular network.
Finally, we must consider the critical role of localized ischemia. Chronic mechanical compression on a peripheral nerve directly compromises the vasa nervorum, inducing a localized ischemic environment that disrupts the blood-nerve barrier, promotes intraneural edema, and ultimately contributes to focal demyelination and neuroma-in-continuity formation. A neuroma, at its fundamental histological level, is a bundled, chaotic, and disorganized mass of regenerating nerve endings trapped within a dense collagenous scar. It serves as a potent anatomical generator of ectopic electrical discharges, translating clinically into localized pain, severe allodynia, and a strongly positive Tinel’s sign following nerve damage. Understanding these distinct but overlapping pathophysiological cascades is the absolute prerequisite for selecting the appropriate surgical intervention and achieving a successful clinical outcome.
Detailed Surgical Anatomy and Biomechanics
Before a scalpel ever touches the skin, a comprehensive, three-dimensional understanding of the relevant surgical anatomy and its biomechanical behavior is paramount. While peripheral nerves generally follow a predictable anatomical course, they are notoriously susceptible to subtle, yet clinically significant, anatomical variations. Recognizing these variations is the key to avoiding catastrophic iatrogenic injury and ensuring a complete, effective decompression.
Lateral Femoral Cutaneous Nerve Anatomy
The Lateral Femoral Cutaneous Nerve (LFCN) is a purely sensory nerve responsible for the cutaneous innervation of the anterolateral thigh. It originates from the lumbar plexus, specifically drawing its fibers from the dorsal divisions of the L2 and L3 spinal nerve roots. As it descends through the retroperitoneum, it typically emerges from the lateral border of the psoas major muscle, crossing the iliacus muscle deep to the iliac fascia. The critical zone for the LFCN—and the primary site of its pathology—is its exit from the pelvis into the anterior thigh.
In the majority of patients, the nerve courses just medial to the Anterior Superior Iliac Spine (ASIS), passing under or through the inguinal ligament before piercing the fascia lata. However, this region is characterized by extreme anatomical variability. Cadaveric studies have demonstrated that the LFCN can run directly through the substance of the inguinal ligament, chafe against the sharp bony prominence of the ASIS, or even course over the most medial portion of the ASIS itself, completely deviating from its typical, deeper, and more medial trajectory. This inherent anatomical aberrancy is precisely why the LFCN is so exquisitely prone to spontaneous entrapment (meralgia paresthetica) or iatrogenic injury during anterior approaches to the hip, pelvic osteotomies, or bone graft harvesting from the iliac crest.
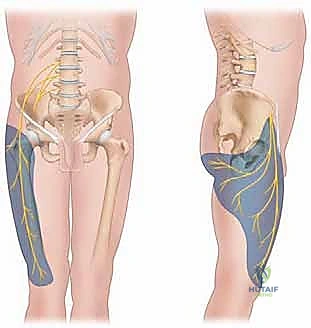
The biomechanics of LFCN entrapment are closely tied to the patient's posture and body habitus. The most common site of impingement is at the level of the inguinal ligament, where the nerve can be dynamically compressed by the ligament itself during hip extension, or strangulated by the deep fascia of the internal oblique and transversalis muscles as it sharply angulates to exit the retroperitoneum. Furthermore, during surgical exploration and decompression of the LFCN, the operating surgeon must exercise extraordinary caution due to the immediate proximity of the deep circumflex iliac artery. This critical vascular structure frequently crosses the nerve in the retroperitoneal space just proximal to the inguinal ligament. Failure to identify and meticulously protect this artery can lead to severe hemorrhage, obscuring the surgical field and dramatically increasing the risk of iatrogenic nerve transection.
Common Peroneal Nerve Anatomy
The Common Peroneal Nerve (CPN) is a major, mixed motor and sensory branch of the sciatic nerve, formed from the dorsal branches of the ventral rami of the L4, L5, S1, and S2 sacral plexus roots. It typically bifurcates from the tibial nerve in the distal third of the posterior thigh, descending obliquely along the lateral border of the popliteal fossa, immediately medial to the biceps femoris tendon. The CPN carries critical motor fibers that innervate the anterior and lateral compartments of the lower leg, driving foot dorsiflexion (via the tibialis anterior, extensor digitorum longus, and extensor hallucis longus) and foot eversion (via the peroneus longus and brevis). Additionally, it provides vital sensory innervation to the anterolateral lower leg and the vast majority of the dorsum of the foot and toes through its terminal superficial and deep branches.
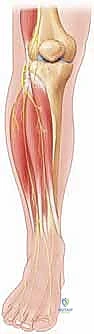
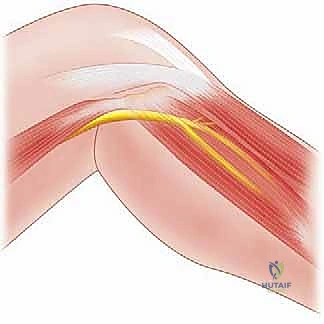
Pathology of the CPN classically and most frequently occurs as it wraps anteriorly around the neck of the fibula. In this highly vulnerable anatomical zone, the nerve passes through a tight fibro-osseous tunnel deep to the fascial origin of the peroneus longus muscle, just prior to its bifurcation into the deep and superficial peroneal nerves. This superficial and tethered position makes the CPN highly susceptible to direct blunt trauma to the lateral knee, stretch injuries from severe knee dislocations or varus stress, iatrogenic injury during proximal fibular osteotomies or lateral knee arthrotomies, and external compression from tight casts or prolonged immobilization.
When evaluating potential neuropathies in the lateral knee or lower leg, the astute surgeon must remember that while the CPN is the primary peripheral nerve affected, neuromas of smaller, adjacent sensory nerves can also develop and confound the clinical picture. These include the lateral retinacular nerve, medial retinacular nerves, the articular branch of the common peroneal nerve (transmitting deep joint pain), and the infrapatellar branch of the saphenous nerve. These smaller, often-overlooked nerves can be a profound source of chronic postoperative knee pain, frequently masking CPN involvement or being mistakenly diagnosed as isolated CPN neuropathy. A thorough understanding of this complex local neural network is essential for accurate diagnosis and comprehensive surgical management.
General Nerve Anatomy and Vascularity Principles
A fundamental principle of peripheral nerve surgery is the recognition that a peripheral nerve possesses a highly sophisticated, intrinsic and extrinsic blood supply. This rich vascular network is what allows us, as surgeons, to safely mobilize the nerve from its fibrotic anatomical bed, perform epineurotomy, and even execute delicate interfascicular neurolysis when strictly necessary. The extrinsic vascular system consists of regional segmental blood vessels that enter the nerve through a delicate, areolar connective tissue sling known as the mesoneurium. Once these vessels pierce the epineurium, they transition into the intrinsic system, forming an extensive, highly anastomotic network of longitudinal microvessels within the epineurial, perineurial, and endoneurial layers.
Extensive microvascular studies have demonstrated that a healthy peripheral nerve can be safely elevated and isolated from its extrinsic segmental vascular supply for a length of approximately 60 times its outer diameter without suffering irreversible ischemic damage. This physiological tolerance is critical, as it means we can often mobilize the nerve sufficiently to transpose it into a healthy, well-vascularized soft tissue bed free from external compression.
However, extreme caution and surgical judgment must be exercised, particularly in the setting of repeat neurolysis. Repeated surgical dissection and circumferential mobilization strip the nerve of its mesoneurium, risking profound devascularization. The surgeon must constantly balance the mechanical need for complete decompression and transposition with the biological imperative to preserve the nerve's delicate intrinsic vascularity. Overzealous skeletonization of the nerve will inevitably lead to ischemic fibrosis, exacerbating the patient's neuropathy and leading to a catastrophic surgical failure.
Exhaustive Indications and Contraindications
The decision to proceed with surgical decompression of a peripheral nerve must be based on a rigorous, evidence-based evaluation. Surgery is rarely the first line of treatment, save for acute, severe traumatic injuries with documented loss of nerve continuity. For chronic entrapment syndromes of the LFCN and CPN, a meticulous period of conservative management is mandatory. Surgical intervention is generally reserved for patients who have failed exhaustive non-operative modalities and present with clear, progressive clinical deficits or intractable neuropathic pain that severely degrades their quality of life.
Conservative management typically spans a minimum of 12 weeks and should encompass a multimodal approach. This includes strict activity modification to eliminate inciting biomechanical stressors, targeted physical and occupational therapy to address muscle imbalances and fascial tightness, and aggressive pharmacological management utilizing membrane-stabilizing agents (such as gabapentin or pregabalin), selective serotonin-norepinephrine reuptake inhibitors (SNRIs), and appropriate analgesics. Diagnostic and therapeutic image-guided nerve blocks with local anesthetics and corticosteroids can also provide both symptomatic relief and critical diagnostic information. If a patient fails to achieve a clinically meaningful recovery after this comprehensive 12-week regimen, nonoperative management is deemed a failure, and surgical evaluation is formally indicated.
For the Common Peroneal Nerve, indications for surgery are often more urgent due to the devastating functional consequences of a foot drop. Progressive motor weakness in the anterior or lateral compartments of the leg, particularly if accompanied by a rapidly deteriorating electromyography (EMG) profile showing active denervation, is a strong indication for prompt surgical exploration. In contrast, LFCN entrapment (meralgia paresthetica) is purely sensory; thus, the primary indication for surgery is intractable, severe dysesthesia and pain that is refractory to all conservative measures. In both scenarios, the presence of a strongly positive Tinel’s sign at the anatomical site of suspected compression serves as an excellent clinical predictor of a favorable surgical outcome following decompression.
| Parameter | Lateral Femoral Cutaneous Nerve (LFCN) | Common Peroneal Nerve (CPN) |
|---|---|---|
| Primary Indications | Intractable anterolateral thigh pain/dysesthesia failing >12 weeks of conservative care; Positive diagnostic nerve block; Severe restriction of ADLs due to pain. | Progressive or profound motor deficit (foot drop); Intractable lateral leg/dorsal foot pain; Positive EMG/NCS indicating focal entrapment at the fibular head; Failure of conservative management. |
| Relative Indications | Recurrent symptoms after previous successful block; Intolerance to neuropathic pain medications. | Static, incomplete motor deficit with severe pain; Impending nerve compression from expanding local mass (e.g., intraneural ganglion cyst). |
| Absolute Contraindications | Active local or systemic infection; Uncorrected severe coagulopathy; Pain entirely non-dermatomal or widespread. | Active infection at the surgical site; Severe, advanced peripheral neuropathy (e.g., end-stage diabetic neuropathy) where the nerve lacks regenerative capacity; Complete, long-standing (>18-24 months) flaccid paralysis where motor endplates are irreversibly lost (tendon transfer indicated instead). |
| Relative Contraindications | Severe psychiatric comorbidities confounding pain assessment; Obesity precluding safe surgical access (relative). | Concomitant severe L5 radiculopathy mimicking CPN palsy (Double Crush Syndrome requires careful staging); Poor soft tissue envelope over the fibular head. |
Pre-Operative Planning, Templating, and Patient Positioning
The preoperative phase is the foundation upon which surgical success is built. It begins with a meticulous, highly detailed patient history and physical examination. The neurologic assessment is paramount and often suggests the diagnosis by demonstrating a strict dermatomal distribution of pain, paresthesia, or motor weakness. The surgeon must perform a thorough manual muscle test, carefully differentiating between an L5 radiculopathy (which affects foot inversion via the tibialis posterior) and a CPN palsy (where foot inversion is spared). A precise mapping of the sensory deficit and the exact location of the Tinel's sign must be documented.
Diagnostic challenges are rampant in peripheral nerve surgery. Traditional imaging modalities, including plain radiographs, CT scans, and radiolabeled bone scans, have extremely limited utility in definitively confirming the diagnosis of a malfunctioning nerve. They are primarily deployed to exclude other confounding pathologies, such as occult fractures, osteoarthritis, or bony tumors. High-resolution Magnetic Resonance Neurography (MRN) and high-frequency neuromuscular ultrasound are emerging as the gold standards, capable of visualizing nerve cross-sectional area, intraneural edema, and exact sites of anatomical tethering. However, these modalities are highly operator-dependent.
Electrodiagnostic studies (EMG/NCS) are frequently utilized but must be interpreted with profound caution. It is a well-documented fact in the literature that EMG/NCS can have a false-negative rate as high as 33% in cases of symptomatic peripheral nerve entrapment, particularly in purely sensory nerves like the LFCN or in dynamic compression scenarios. Similarly, while diagnostic nerve blocks can confirm the pain generator, significant anatomic variations can lead to misplaced injectate and false-negative results. Therefore, the surgeon must rely heavily on clinical acumen, utilizing diagnostics to eliminate other etiologies for postoperative pain, such as deep infection, loosened orthopedic hardware, mechanical limb misalignment, or spinal stenosis, before committing to peripheral nerve decompression.
Operating Room Setup and Patient Positioning
The execution of flawless peripheral nerve surgery requires a meticulously organized operating room. Ensure adequate space for the surgical team, specialized instrument tables, and the potential introduction of a surgical microscope. Excellent, shadowless overhead lighting is crucial, and this must be supplemented by a high-intensity fiber-optic headlight for the primary surgeon. Magnification is non-negotiable; wide-field surgical loupes with a minimum magnification of 3.5x (preferably 4.5x or higher) are absolutely essential for identifying microvascular structures and fascial bands. A dedicated set of fine microsurgical instruments—including delicate Adson forceps with teeth, smooth jeweler's forceps, right-angle nerve hooks, fine tenotomy scissors, and precise bipolar electrocautery—must be opened and ready on the Mayo stand.
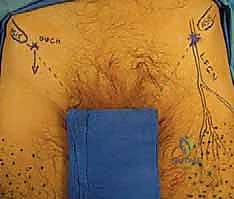
For Lateral Femoral Cutaneous Nerve (LFCN) Decompression, the patient is placed in the strict supine position on a radiolucent operating table, though fluoroscopy is rarely required. Both arms should be carefully padded and tucked to the patient's side or abducted on arm boards to prevent brachial plexus traction. The affected leg should be positioned to allow for unencumbered access to the anterolateral thigh and groin region. Placing a small gel bump under the ipsilateral hemipelvis can slightly externally rotate the hip and make the ASIS more prominent, facilitating easier identification of the primary surgical landmark. Preoperatively, the surgeon must palpate and mark the ASIS, trace the anticipated course of the inguinal ligament, and mark the planned incision line.
For Common Peroneal Nerve (CPN) Decompression, the patient is also placed in the supine position. A well-padded thigh tourniquet is applied to the proximal affected limb, provided the patient has no history of peripheral vascular disease or previous vascular bypass grafting in the extremity. The affected leg is gently flexed at the knee and appropriately rotated (often utilizing a bump under the ipsilateral hip to internally rotate the limb, or positioning the limb in a figure-of-four position depending on surgeon preference) to maximally expose the lateral aspect of the knee and the fibular head. Preoperatively, the surgeon must identify and mark the prominent fibular head, the course of the biceps femoris tendon, and the anticipated trajectory of the peroneus longus muscle. The incision is planned obliquely, approximately 1 to 2 fingerbreadths distal and posterior to the fibular head.
Step-by-Step Surgical Approach and Decompression Technique
Alright, team, let's get scrubbed in. We have exhaustively reviewed the anatomy, optimized the patient's medical status, and our OR is perfectly set. Peripheral nerve decompression is a game of millimeters. Precision, meticulous hemostasis, and immense patience are our watchwords today. We respect the tissue, we respect the vascularity, and we release every single point of fascial tethering.
Lateral Femoral Cutaneous Nerve Decompression Technique
Our primary objective during this procedure is to definitively decompress the LFCN from any extrinsic compression—most commonly located at, under, or just distal to the inguinal ligament—and to ensure its completely free, unhindered gliding through its anatomical tunnel during full hip extension.
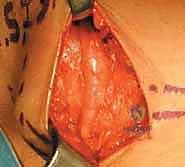
With the patient supine and the surgical field meticulously prepped and draped, we begin by making a roughly 6 cm longitudinal or slightly oblique incision. The incision starts approximately 2 cm medial and 2 cm distal to the Anterior Superior Iliac Spine (ASIS), following the natural Langer’s lines of the groin when possible. We carry the dissection sharply through the skin and subcutaneous tissue, utilizing bipolar electrocautery to maintain an absolutely bloodless field. We deepen the dissection through Scarpa’s fascia until the gleaming white surface of the fascia lata is identified.
At this stage, we transition to blunt dissection and careful spreading with fine tenotomy scissors. We are looking for the LFCN as it pierces the fascia lata to become superficial. Remember, the nerve often branches before or immediately after piercing the fascia; we must identify all branches. Once the main trunk of the LFCN is identified, we secure it with a soft vessel loop—never crushing it with forceps. We then meticulously trace the nerve proximally toward the inguinal ligament. Using fine scissors, we divide the fascia lata and the superficial layer of the inguinal ligament directly over the nerve.
We must visually confirm that the nerve is entirely free as it dives into the pelvis. Frequently, there is a dense, deep fascial band from the iliacus fascia that creates a sharp "kink" in the nerve; this must be completely released. During this deep, retroperitoneal portion of the dissection, we must remain hyper-vigilant. We retract the internal oblique and transversalis fascia superiorly and carefully sweep medially to identify and protect the deep circumflex iliac artery, which often crosses directly over or under the nerve in this space. Once the nerve is completely liberated and glides freely with passive hip motion, we irrigate the wound copiously and close the deep dermal and epidermal layers. If the nerve is found to be severely damaged, scarred beyond repair, or if this is a revision for a painful neuroma, a formal neurectomy with proximal transposition of the stump deep into the retroperitoneal pelvic musculature may be indicated over simple decompression.
Common Peroneal Nerve Decompression Technique
For the CPN, our goal is a complete release of the nerve from the distal thigh, around the fibular neck, and down into the anterior and lateral compartments of the leg, ensuring both the deep and superficial branches are entirely free of fascial compression.
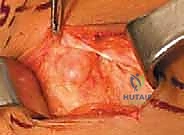
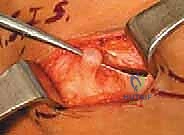
We begin with an oblique incision, roughly 6 to 8 cm in length, centered over the fibular neck and tracking along the posterior border of the biceps femoris tendon, curving gently anteriorly toward the anterior compartment. We incise the skin and subcutaneous tissues, maintaining strict hemostasis. Our first major landmark is the biceps femoris tendon. We incise the deep fascia just posterior to the tendon in the distal thigh. Using blunt finger dissection or a right-angle clamp, we locate the large, unmistakable trunk of the Common Peroneal Nerve in the popliteal fossa, proximal to the zone of compression.
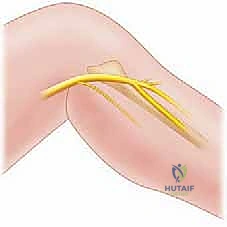

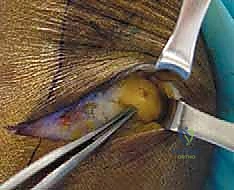
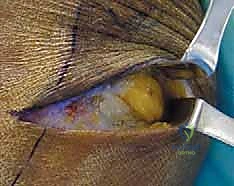
Once identified proximally, we place a soft vessel loop around the CPN for gentle handling. We then trace the nerve distally as it courses obliquely across the lateral head of the gastrocnemius and approaches the fibular neck. Here, the nerve dives beneath the sharp, tendinous leading edge of the peroneus longus muscle. This is the critical zone of entrapment. Using dissecting scissors, we carefully incise the superficial fascia of the peroneus longus, retracting the muscle belly anteriorly.
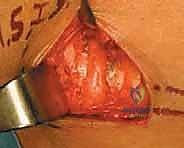
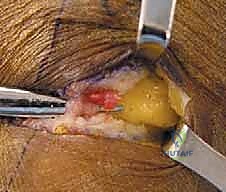
Deep to the muscle belly, we will encounter a thick, transverse fibrous band—the fibular tunnel—that tightly binds the nerve against the periosteum of the fibular neck. This band must be completely divided. We insert a blunt nerve hook or the lower blade of our tenotomy scissors deep to this fascial band and sharply divide it, taking great care not to injure the underlying epineurium. We continue this release distally until we clearly visualize the bifurcation of the CPN into its deep and superficial branches. We must follow both branches for several centimeters into their respective muscular compartments (the deep branch diving through the anterior intermuscular septum) to ensure no secondary distal
