Mastering Distal Upper Extremity Nerve Tumor Excision & Reconstruction
Key Takeaway
This masterclass guides fellows through surgical management of distal upper extremity nerve tumors. We cover meticulous dissection, distinguishing schwannomas from neurofibromas, and precise enucleation or resection. Emphasis is placed on nerve preservation, microdissection, and reconstruction techniques using autografts or conduits, ensuring optimal functional outcomes while navigating complex anatomy and potential intraoperative challenges.
Comprehensive Introduction and Patho-Epidemiology
Fellows and colleagues, welcome to the operating theater and to this definitive exploration of peripheral nerve pathology. Today, we are tackling a fascinating, intricate, and often unforgiving area of upper extremity surgery: the surgical excision and reconstruction of nerve tumors in the distal upper extremity. These lesions, while relatively uncommon in the grand scheme of hand and wrist pathology, demand a highly nuanced understanding of microanatomy, meticulous surgical technique, and a strategic, forward-thinking approach to preserve, restore, or reconstruct vital neural function. The margin for error is razor-thin; a millimeter of misplaced dissection can mean the difference between full functional recovery and devastating, permanent iatrogenic paralysis.
Peripheral nerve sheath tumors (PNSTs) represent an exclusive subset of neoplasms, comprising less than 5% of all tumors we encounter in the hand, wrist, and forearm. The vast majority of these lesions are benign, with schwannomas (neurilemmomas) and neurofibromas making up the lion's share of our clinical volume. However, the specter of malignancy, particularly in patients harboring the Neurofibromatosis type I (NF1) genetic mutation, always looms large in our differential diagnosis. Our primary surgical and oncologic goals are tripartite: obtaining a definitive histopathologic diagnosis, achieving durable symptom control, and, crucially, executing the preservation or reconstruction of neurological function.
To master this domain, we must first understand the fundamental pathology and natural history of the enemy. Malignant peripheral nerve sheath tumors (MPNSTs) are aggressive, high-grade sarcomas that can arise de novo, but critically, they frequently evolve from a pre-existing benign peripheral nerve sheath tumor (BPNST), specifically a plexiform neurofibroma. This malignant transformation is a paramount concern in our NF1 patient population, where approximately half of all MPNSTs occur. While the sporadic incidence of MPNST in the general population is vanishingly rare (around 0.001%), the lifetime risk for an NF1 patient escalates dramatically to 8% to 13%. This staggering statistic underscores the absolute necessity for extreme vigilance, serial examinations, and a low threshold for advanced imaging and biopsy in this high-risk cohort.
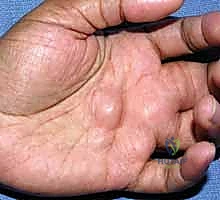
Conversely, the most frequent nerve tumors we encounter in the distal upper extremity are benign schwannomas. They typically present as solitary, encapsulated lesions in middle-aged adults, growing insidiously and often painlessly over several years. While generally small at presentation—usually measuring less than 2.5 cm—even these benign tumors can precipitate significant nerve dysfunction. This dysfunction is rarely due to the inherent pathology of the tumor invading the axons; rather, it is secondary to focal, mechanical impingement and microvascular compromise of adjacent normal fascicles confined within the unyielding epineurium. This is a critical clinical distinction: the tumor itself may not be destroying neural tissue, but its mass effect within a closed space certainly will. Therefore, the neural origin of a mass is often not anticipated by the referring provider. Always discuss the distinct possibility of a nerve tumor preoperatively with your patient, as unexpected, transient, or even permanent loss of function may occur after surgery, even for benign lesions. Informed patient counseling is paramount to managing postoperative expectations.
Detailed Surgical Anatomy and Biomechanics
Before we make any incision, a profound, almost microscopic dive into the anatomy of peripheral nerves is non-negotiable. Remember, these tumors arise from the supporting cells of the nerve sheath, not the conducting axons themselves. Understanding the spatial relationship between the tumor and the functional fascicles dictates our entire surgical approach.
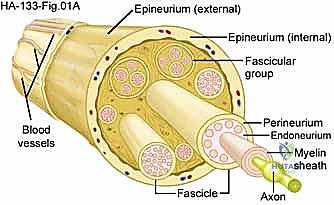
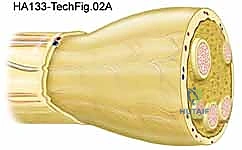
Let us meticulously trace the histological layers, progressing from superficial to deep. The outermost layer is the Epineurium, a robust layer of dense connective tissue. It is subdivided into the epifascicular epineurium, which surrounds the entire nerve trunk, and the interfascicular epineurium, which separates individual fascicles. The epineurium is highly vascularized, containing the vasa nervorum—the extrinsic blood supply to the nerve. During our initial dissection and mobilization, we must respect these longitudinal vessels to prevent ischemic injury to the nerve trunk.
Beneath the epineurium lies the Perineurium. This is a strong, metabolically active, and highly cellular layer that encapsulates individual fascicles (bundles of axons). The perineurium is a critical structure; it forms the blood-nerve barrier via tight junctions between perineurial cells, maintaining the privileged intrafascicular milieu required for action potential propagation. It also provides the primary tensile strength of the peripheral nerve. When a benign schwannoma grows, it typically arises from a single fascicle and expands eccentrically, displacing and compressing the remaining normal fascicles against the perineurial and epineurial envelopes.
Deep within each fascicle is the Endoneurium, a delicate matrix of loose connective tissue that surrounds each individual axon and its associated Schwann cells. It is these Schwann cells—the myelin-producing glial cells of the peripheral nervous system—or occasionally perineurial fibroblasts, that serve as the cell of origin for the vast majority of nerve sheath tumors. In a schwannoma, the tumor is generally encapsulated and separable from the main nerve trunk. In contrast, a neurofibroma is unencapsulated and intimately intertwines with the axonal fibers, expanding the nerve centrally and making function-sparing enucleation substantially more difficult, if not impossible.
In patients with NF1, the microanatomy is often grossly distorted. We frequently encounter a complex, chaotic picture: multiple schwannomas, diffuse neurofibromas, or a combination of both, affecting major upper extremity nerve trunks simultaneously. A particular oncologic and surgical concern is the plexiform neurofibroma. These present as thick, tortuous, "bag of worms" masses that infiltrate and expand long segments of a nerve and its branches. They are pathognomonic for NF1, possess a significantly higher risk of malignant progression to MPNST, and require careful, lifelong clinical and radiographic monitoring, as their complete surgical extirpation without profound neurological morbidity is rarely feasible.
Exhaustive Indications and Contraindications
The decision to operate on a distal upper extremity nerve tumor is rarely an emergency, but it requires a calculated assessment of the tumor's natural history, the patient's symptomatology, and the inherent risks of microsurgery. An isolated mass in the distal upper extremity warrants surgical intervention for three primary reasons: definitive histopathologic diagnosis, mechanical control of progressive symptoms, and the exclusion of malignancy.
| Category | Specific Clinical Scenarios | Rationale / Considerations |
|---|---|---|
| Absolute Indications | Rapid, unexplained growth of a previously stable mass. | High suspicion for malignant transformation (MPNST). Requires urgent biopsy/resection. |
| Absolute Indications | Intractable, progressive rest pain or severe nocturnal pain. | Suggests aggressive expansion, ischemia, or malignancy. |
| Absolute Indications | Progressive, documented motor weakness or profound sensory loss. | Indicates severe axonal compression or infiltration. Decompression/resection is required to prevent irreversible Wallerian degeneration. |
| Relative Indications | Mild to moderate paresthesias or mechanical irritation (e.g., carpal tunnel syndrome secondary to a tumor). | Surgery aims to improve quality of life and prevent further axonal damage. |
| Relative Indications | Patient anxiety or cosmetic deformity from a visible mass. | Must be weighed against the risk of iatrogenic nerve injury during excision. |
| Absolute Contraindications | Diffuse, asymptomatic plexiform neurofibroma in an NF1 patient without focal signs of malignancy. | Attempted resection will result in catastrophic, widespread paralysis. Observation is mandatory. |
| Relative Contraindications | Small, entirely asymptomatic, incidentally discovered mass with classic benign MRI features. | The risk of iatrogenic neuropraxia during excision outweighs the benefits. Serial observation is preferred. |
When considering surgical intervention, the surgeon must adopt a philosophy balanced between "tissue is the issue" (the need for diagnosis) and "primum non nocere" (first, do no harm). In the absence of rapid growth, intractable pain, or progressive nerve dysfunction, it is perfectly reasonable to observe a distal upper extremity mass, especially if high-resolution MRI features are entirely consistent with a benign BPNST and show no aggressive features.
However, if symptoms progress, surgical management becomes our call to action. For instance, if a patient presents with classic carpal tunnel syndrome caused by a small, benign-appearing tumor of the palmar cutaneous branch or within the canal itself, a limited surgical approach involving an open carpal tunnel release and a definitive excisional biopsy of the small branch may be sufficient. If the main median nerve is involved by an asymptomatic, benign-appearing lesion, one might opt for decompression alone without attempting high-risk enucleation, provided the diagnosis of a benign lesion is virtually certain.
Pre-Operative Planning, Templating, and Patient Positioning
The Diagnostic Puzzle: History and Physical Examination
A meticulous history and a highly specific physical examination are our first, and arguably most important, steps in anticipating a nerve tumor. During the history, focus heavily on the duration of the mass, its growth velocity (slow and insidious versus rapid and explosive), and any local or distal effects. While mild, vague discomfort is common, frank paresthesias or sharp neuropathic pain are surprisingly the exception rather than the rule in early benign lesions. This means we must maintain a high index of suspicion and consider a nerve tumor even in the absolute absence of classic compressive neuropathy symptoms.
The physical examination requires a nuanced touch. Palpation is the key diagnostic maneuver. A peripheral nerve tumor will typically be highly mobile in the transverse plane (side-to-side) but firmly tethered longitudinally along the anatomical course of the host nerve. This specific biomechanical behavior distinguishes it from many other soft tissue masses like lipomas or superficial cysts.
Sensory testing must be objective and reproducible. We utilize Semmes-Weinstein monofilaments to assess light touch and pressure thresholds. Early mechanical nerve compression often increases this threshold long before two-point discrimination (which measures innervation density) becomes abnormal. Motor assessment involves looking for subtle visible atrophy in the intrinsic musculature innervated by the affected nerve. However, manual strength testing (e.g., 5/5 grading) is often completely normal, even with significant tumor burden, due to the compensatory nature of motor units. Applying direct, firm pressure over the nerve just proximal to the mass can sometimes elicit sensitivity or paresthesias in the nerve's distal distribution. Percussing the nerve immediately adjacent to the mass may produce a Tinel sign; however, remember that a positive Tinel sign primarily indicates an injured nerve attempting to regenerate. Most established, slow-growing benign nerve tumors do not exhibit a strongly positive Tinel sign unless there is concurrent mechanical entrapment.
Differential Diagnosis: Ruling Out the Imposters
The distal upper extremity is fraught with soft tissue masses that can perfectly mimic a nerve tumor. A comprehensive differential diagnosis is crucial to avoid misdiagnosis, inappropriate patient counseling, and disastrous surgical planning.
Ganglia are the most common masqueraders. They arise from joint capsules or tendon sheaths, typically in characteristic locations (e.g., the volar radiocarpal joint, dorsal scapholunate ligament). They usually transilluminate and can be confirmed by aspirating viscous mucinoid material.
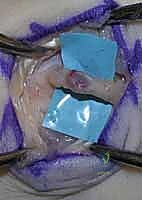
A patient presenting with localized hypesthesia of the thenar eminence caused by an apparent tumor compressing the palmar cutaneous branch (PCB) of the median nerve.
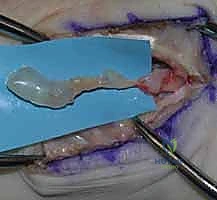
Careful intraoperative dissection reveals the mass is not a nerve tumor, but a ganglion cyst arising deep from the radioscaphoid articulation, compressing the overlying nerve.
Giant Cell Tumors (GCTs) of the tendon sheath are reactive synovial lesions that commonly occur in the palm and volar digits. They are often palpably firm and multi-nodular, which is a key differentiator from the smooth, fusiform margins of most nerve tumors. Lipomas, while common, are usually more superficial, doughy, and highly mobile in all planes. Rarely, they can grow within confined anatomical spaces like the carpal canal or Guyon's canal, causing compressive neuropathy. Nodular fasciitis is a firm, rapidly growing reactive soft tissue proliferation, often found on the volar forearm. Its aggressive, rapid growth can clinically mimic a sarcoma, but palpation reveals dense adhesions to adjacent subcutaneous tissue, distinguishing it from the relative transverse mobility of a nerve tumor.
Advanced Imaging: Peeking Inside the Neural Landscape
Plain radiographs are a standard initial step to look for intralesional calcification or signs of adjacent osseous invasion. Intralesional calcification is exceedingly rare in BPNSTs; its presence should immediately pivot your differential toward synovial chondromatosis, calcific tendonitis, myositis ossificans, or a synovial sarcoma.
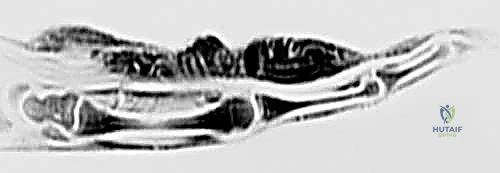
Magnetic Resonance Imaging (MRI) with and without intravenous contrast is our most valuable, indispensable diagnostic tool. It helps evaluate intrinsic tumor characteristics, delineate surrounding vascular and tendinous anatomy, and critically, plan our surgical approach.
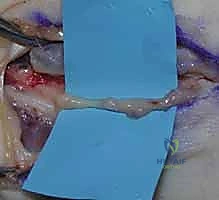
Cross-sectional T2-weighted imaging of a benign peripheral nerve sheath tumor reveals the hyperintense mass to be perfectly contiguous with the normal, hypointense axonal bundles proximally and distally.
Further MRI sequencing confirms the fusiform dilation typical of a peripheral nerve sheath tumor, demonstrating the "split-fat" sign at the proximal and distal poles.
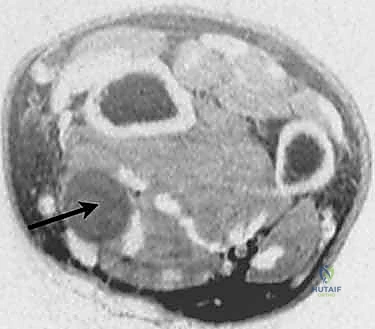
Localization of the tumor to the direct vicinity of a known large nerve trunk strongly suggests a peripheral nerve tumor. On T1-weighted images, nerve tumors often show intermediate signal intensity, sometimes with a high fat content, particularly in lipofibromatous hamartomas.
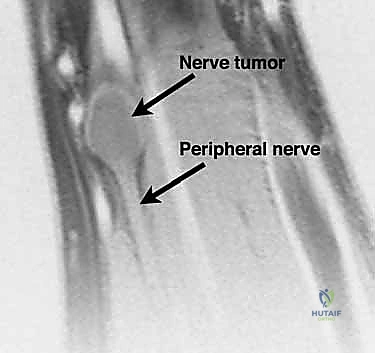
MRI of a benign nerve sheath tumor often demonstrates a high fat content, seen best as hyperintense signal on T1-weighted images.
On T2-weighted images, BPNSTs are typically brightly hyperintense. They may exhibit a "target sign"—a hyperintense rim (myxoid tissue) with a hypointense center (fibrocollagenous tissue)—which is highly suggestive of a neurofibroma or schwannoma. However, while these MRI features are strongly suggestive, they are not pathognomonic and can occasionally be seen with other soft tissue neoplasms. Signs of malignancy on MRI include irregular, invasive margins, size greater than 5 cm, rapid interval growth, peritumoral edema, and central tumor necrosis.
Step-by-Step Surgical Approach and Fixation Technique
Preoperative Preparation and Initial Exposure
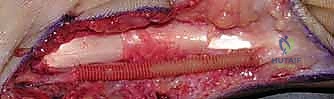
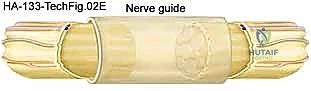
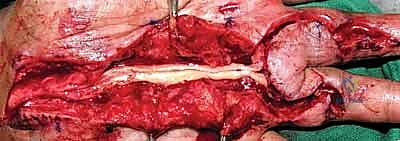
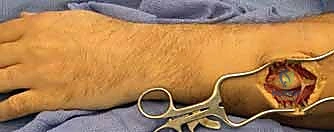
Surgical excision of a nerve tumor demands a meticulous, unhurried environment. The patient is positioned supine with the operative arm extended on a radiolucent hand table. A well-padded pneumatic tourniquet is applied to the proximal arm. While the initial exposure can be performed under tourniquet control to ensure a bloodless field, the tourniquet must be deflated prior to final hemostasis and occasionally during the procedure if intraoperative nerve stimulation is required, as prolonged ischemia will blunt the nerve's electrical responsiveness. Loupe magnification (minimum 3.5x) is mandatory for the initial approach, and an operating microscope should be immediately available in the room for intraneural dissection and potential grafting.
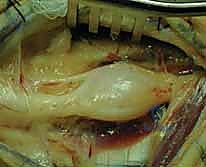
The surgical incision must be extensile. We utilize a generous, longitudinal incision centered over the mass, incorporating Brunner-type zig-zags if crossing flexion creases, to allow for adequate proximal and distal exposure of the normal nerve trunk.
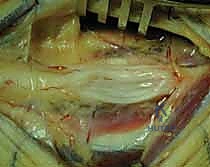
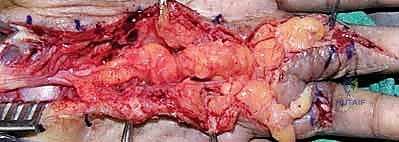
Once the subcutaneous tissues are divided, the normal nerve trunk is identified both proximally and distally to the tumor in virgin, unscarred tissue beds. Vessel loops are passed around the nerve for gentle, atraumatic handling.
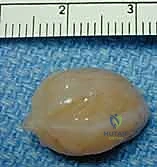
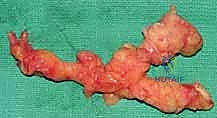
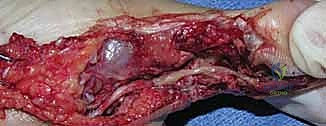
The dissection proceeds toward the mass. Here, we see the tumor coming into view, completely enveloped by the epineurium of the host nerve.