Upper Extremity Anesthesia: An Orthopaedic Surgeon's Masterclass in Perioperative Management
Key Takeaway
Join us in the OR for a masterclass on upper extremity anesthesia. We'll meticulously review preoperative planning, intricate surgical anatomy, and real-time intraoperative execution of regional and general anesthesia techniques. Learn critical pearls, avoid pitfalls, and optimize patient outcomes from a surgeon's perspective. This guide covers everything from nerve blocks to managing complex patient comorbidities like rheumatoid arthritis.
Comprehensive Introduction and Patho-Epidemiology
Welcome, fellows and colleagues, to the operating theater. Today, we are not merely discussing surgical technique, but rather, the critical, often underappreciated, role of anesthetic considerations in ensuring successful outcomes for our upper extremity cases. As orthopaedic surgeons, our focus is naturally drawn to the osseous and soft tissue reconstruction, but the patient's entire surgical journey—from preoperative assessment to postoperative recovery—is profoundly influenced by the anesthetic plan. Understanding this symbiotic relationship between the orthopaedic surgeon and the anesthesiologist is paramount for any master surgeon aiming to optimize patient outcomes, minimize perioperative morbidity, and enhance hospital throughput.
The epidemiology of upper extremity surgery has shifted dramatically over the past two decades. With the advent of advanced microsurgical techniques, arthroscopic innovations, and robust osteosynthesis implants, procedures that once required prolonged inpatient stays are now routinely performed in the ambulatory setting. This paradigm shift has been largely facilitated by parallel advancements in anesthesiology, specifically the refinement of ultrasound-guided regional anesthesia and multimodal analgesia protocols. However, this transition is not without its patho-epidemiological challenges. The aging population presents with a higher burden of systemic comorbidities, including advanced rheumatoid arthritis, cardiopulmonary disease, and metabolic syndrome. Consequently, the perioperative stress response, characterized by neuroendocrine activation, hypercoagulability, and immunosuppression, must be meticulously managed.
Furthermore, the incidence of postoperative complications such as Postoperative Cognitive Dysfunction (POCD), Postoperative Nausea and Vomiting (PONV), and chronic regional pain syndrome (CRPS) remains a significant concern in upper extremity surgery. By actively participating in the formulation of the anesthetic strategy, the orthopaedic surgeon can directly mitigate these risks. Whether negotiating the necessity of profound muscle relaxation for a complex distal humerus fracture reduction or advocating for a specific peripheral nerve block to preemptively address tourniquet pain, our comprehensive understanding of these anesthetic modalities is a non-negotiable component of surgical mastery.
Detailed Surgical Anatomy and Biomechanics
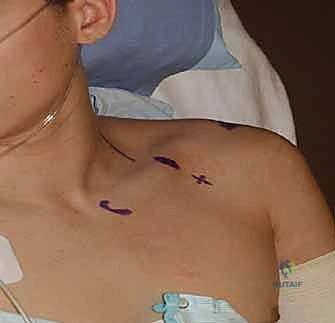
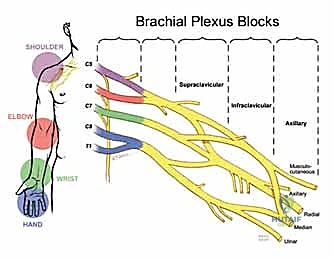
To effectively collaborate with our anesthesia colleagues, a profound understanding of the neurovascular anatomy and the biomechanics of patient positioning is essential. The brachial plexus, the primary target for upper extremity regional anesthesia, is a complex network of nerves originating from the ventral rami of the lower four cervical nerves (C5-C8) and the first thoracic nerve (T1). Understanding its anatomical progression—roots, trunks, divisions, cords, and branches—is critical for predicting the efficacy and potential limitations of various regional blocks.
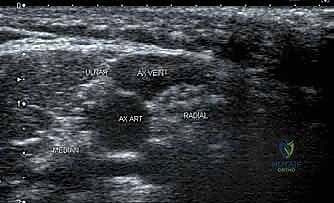
The roots emerge from the intervertebral foramina and pass between the anterior and middle scalene muscles, forming the trunks (superior, middle, and inferior) in the posterior triangle of the neck. This interscalene groove is the prime target for shoulder and proximal humerus surgery. However, the close proximity of the phrenic nerve (originating from C3-C5) and the recurrent laryngeal nerve mandates precise needle placement to avoid unintended diaphragmatic or vocal cord paralysis. As the plexus courses distally over the first rib and under the clavicle, the trunks divide into anterior and posterior divisions, which then form the cords (lateral, posterior, and medial) in the axilla, named according to their relationship with the axillary artery.

Biomechanically, patient positioning during upper extremity surgery significantly impacts both surgical access and anesthetic safety. The "beach chair" position, frequently utilized for shoulder arthroscopy and arthroplasty, presents unique hemodynamic challenges. The elevation of the head relative to the heart can lead to cerebral hypoperfusion if mean arterial pressure is not meticulously maintained, a phenomenon exacerbated by the vasodilatory effects of general anesthesia or interscalene blocks. Conversely, the lateral decubitus position requires careful padding of the dependent axilla to prevent compression neurapraxia of the contralateral brachial plexus, while the operative arm is subjected to precise traction vectors to optimize joint distraction without inducing stretch injuries to the neurovascular bundle.
Exhaustive Indications and Contraindications
The selection of an anesthetic modality—whether general, regional, or a combined approach—requires a nuanced evaluation of the patient's physiological status, the surgical requirements, and the anticipated postoperative recovery trajectory. There is rarely a uniform, "one-size-fits-all" algorithm; rather, each decision must be individualized.
General anesthesia remains a highly reliable modality, particularly indicated for prolonged procedures, cases requiring absolute patient immobility (e.g., complex microsurgical free tissue transfers), or when multiple surgical sites are involved (e.g., simultaneous upper extremity reconstruction and autologous bone graft harvest from the iliac crest). It is also frequently the modality of choice for pediatric patients or those with severe cognitive impairments who cannot cooperate with regional techniques. However, general anesthesia carries inherent risks, including airway trauma, cardiopulmonary depression, and a higher incidence of PONV and POCD.
Regional anesthesia, conversely, offers profound site-specific analgesia, blunts the surgical stress response, and facilitates rapid postoperative recovery. It is highly indicated for procedures where early discharge is desired, in patients with severe cardiopulmonary comorbidities where general anesthesia poses an unacceptable risk, and for preemptive analgesia to mitigate chronic pain pathways. Nevertheless, regional anesthesia is not without its absolute and relative contraindications, which must be rigorously respected to prevent catastrophic complications such as Local Anesthetic Systemic Toxicity (LAST) or permanent nerve injury.
Indications and Contraindications for Anesthetic Modalities
| Modality | Primary Indications | Absolute Contraindications | Relative Contraindications |
|---|---|---|---|
| General Anesthesia | Prolonged surgery, multiple operative sites, patient refusal of block, necessity for immediate post-op neuro exam, complex pediatric/trauma cases. | Lack of appropriate equipment/personnel, severe unoptimized cardiopulmonary failure (case-dependent). | Severe reactive airway disease, history of Malignant Hyperthermia (requires non-triggering agents), severe untreated GERD. |
| Regional Anesthesia (Brachial Plexus Blocks) | Outpatient surgery, severe cardiopulmonary comorbidities, preemptive analgesia, need for continuous post-op pain control, reduction of POCD. | Patient refusal, active infection at the injection site, true allergy to local anesthetics, severe uncorrected coagulopathy. | Pre-existing severe peripheral neuropathy (e.g., advanced diabetic neuropathy), inability to cooperate, contralateral phrenic nerve palsy (for interscalene blocks). |
| Intravenous Regional Anesthesia (Bier Block) | Short duration (< 60 mins) procedures of the hand/forearm (e.g., carpal tunnel, simple hardware removal). | Severe crush injury, ischemic limbs, patient inability to tolerate tourniquet. | Sickle cell disease, severe hypertension, Raynaud's phenomenon. |
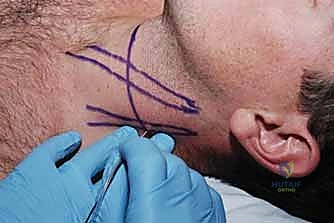
Pre-Operative Planning, Templating, and Patient Positioning
Before we even consider an incision, a meticulous preoperative evaluation is non-negotiable. This is the critical juncture where we identify potential physiological and anatomical challenges, collaborating directly with our anesthesiology colleagues to formulate a robust, fail-safe plan.
The Rheumatoid Arthritis Patient: A Systemic Challenge
Patients presenting with advanced rheumatoid arthritis (RA) embody a highly complex scenario that demands our utmost clinical vigilance. RA is not merely a localized articular disease; it is a relentless, systemic inflammatory process with widespread, potentially lethal perioperative implications.
A paramount concern is the potential for cervical spine instability, specifically atlantoaxial subluxation. This insidious instability arises from the inflammatory erosion of the transverse ligament at the odontoid process of C2, which normally tethers it to the anterior arch of C1. This C1-C2 instability can lead to dynamic subluxation during neck extension or flexion, placing the cervical spinal cord at imminent risk of compression, ischemia, and potentially irreversible paralysis. For any RA patient, especially those with advanced disease or long-standing steroid use, preoperative flexion and extension lateral cervical spine radiographs are absolutely mandatory. We must rigorously assess the anterior atlantodental interval (AADI) and the posterior atlantodental interval (PADI). If significant instability is identified (e.g., AADI > 3mm in adults, or a compromised PADI), a prophylactic cervical fusion—often extending from C1 to the occiput—may be required before we proceed with any elective orthopaedic procedure. The risk of a neurological catastrophe during endotracheal intubation or surgical positioning is simply too high to ignore.
Beyond the cervical spine, RA frequently induces severe anatomic alterations in the temporomandibular joint (TMJ) and the cricoarytenoid joints, leading to severely limited jaw opening, restricted neck mobility, and narrowing of the glottic opening. This triad can render standard direct laryngoscopy and endotracheal intubation extremely difficult, if not impossible. In such perilous airway scenarios, our anesthesiologists must be prepared to employ awake fiberoptic bronchoscope-guided intubation. This highly specialized, technically demanding technique secures the airway while the patient maintains spontaneous ventilation and protective reflexes. As surgeons, we must anticipate this extended induction time and ensure our OR schedule is appropriately adjusted. Furthermore, RA is characterized by accelerated atherosclerosis and restrictive pulmonary disease. A comprehensive preoperative workup, including a 12-lead ECG, echocardiogram, cardiac stress testing, and pulmonary function testing, is essential to quantify their physiological reserve and tailor the anesthetic approach accordingly.
Trauma Patients: Urgency and Unpredictability
Orthopaedic trauma cases inherently present as high-stakes surgical emergencies, demanding rapid, decisive action under immense pressure. The trauma patient's physiological status is often unknown or rapidly evolving, necessitating a highly conservative and structured anesthetic approach.
In the acute trauma setting, an uncleared cervical spine mandates strict adherence to in-line manual stabilization during endotracheal intubation, regardless of whether the patient is awake, obtunded, or undergoing rapid sequence induction (RSI). This is a critical safety measure to prevent iatrogenic spinal cord injury in the presence of an occult fracture. The urgency of the trauma—such as an expanding hematoma, compartment syndrome, or a mangled extremity—may sometimes dictate that general anesthesia be induced before complete radiologic evaluations can be finalized. This underscores the critical importance of a rapid, focused primary and secondary survey. General anesthesia is frequently the modality of choice for complex trauma due to the unpredictable duration of the procedure, the frequent involvement of multiple extremities, the necessity for profound neuromuscular blockade to aid in fracture reduction, and the imperative to assess postoperative neurologic function immediately upon emergence.

Pediatric Patients: A Special Population
Operating on the pediatric population requires a fundamentally unique approach, characterized by a deep understanding of developmental anatomy, physiology, and psychology. Children generally lack the emotional and intellectual maturity to remain conscious, calm, and cooperative during an invasive procedure, making general anesthesia the foundational choice for nearly all pediatric upper extremity surgeries.
While general anesthesia is prevalent, the integration of regional anesthesia offers profound benefits, including superior postoperative analgesia and reduced opioid consumption. However, pediatric regional anesthesia carries specific, highly nuanced considerations. Historically, the lack of appropriately sized pediatric equipment posed a challenge, though modern advancements have largely mitigated this. A primary concern is the loss of subjective patient feedback; performing a regional block in a child who is already under general anesthesia eliminates the ability to detect paresthesias or pain upon injection, theoretically increasing the risk of intraneural injection. Consequently, ultrasound-guided nerve blocks are considered the absolute standard of care in pediatrics. Direct, real-time visualization of the neural targets, the needle tip, and the spread of the local anesthetic significantly reduces the risk of iatrogenic nerve injury.
Furthermore, pediatric patients are highly susceptible to Local Anesthetic Systemic Toxicity (LAST). Their incomplete neural myelination permits greater local anesthetic penetration, but their higher cardiac output and altered protein binding capacities necessitate meticulous, weight-based dosing. For continuous peripheral nerve catheters, dilute local anesthetics (e.g., bupivacaine or ropivacaine 0.1%) are strictly recommended to decrease the risk of systemic accumulation. Recommended infusion rates typically commence at 0.15 mL/kg per hour of bupivacaine 0.25%. Strict adherence to preoperative fasting guidelines is also paramount to prevent catastrophic pulmonary aspiration during induction.
Fasting Guidelines for Pediatric Patients
| Age | Milk and Solids (hr) | Clear Liquids (hr) |
|---|---|---|
| < 6 mo | 4 | 2 |
| 6–36 mo | 6 | 3 |
| > 36 mo | 8 | 3 |
Pediatric Doses and Clinical Characteristics of Commonly Used Local Anesthetics*
| Local Anesthetic | Concentration (%) | Usual Doses (mg/kg) | Maximum Dose, Plain † (mg/kg) | Maximum Dose, with Epinephrine † (mg/kg) | Latency (min) | Duration of Effects (hr) |
|---|---|---|---|---|---|---|
| Lidocaine | 0.5–2.0 | 5 | 7.5 | 10 | 5–15 | 0.75–2.0 |
| Prilocaine | 0.5–1.5 | 5 | 7.0 | 10 | 15–25 | 0.75–2.0 |
| Mepivacaine | 0.5–1.5 | 5–7 | 8.0 | 10 | 5–15 | 1–1.25 |
| Bupivacaine | 0.25–0.5 | 2 | 2.5 | 3 | 15–30 | 2.5–6.0 |
| Ropivacaine | 0.2–10.0 | 3 | 3.5 | NA | 7–20 | 2.5–5.0 |
| *Data are not applicable to spinal anesthesia or intravenous regional anesthesia. † Maximum doses vary; free and unbound local anesthetic causes toxicity, not total dose. Do not apply if previously injected or if local anesthetic infusion is maintained. |
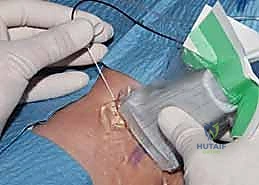
Anticoagulation Guidelines: A Team Approach
Managing patients on chronic anticoagulation or antiplatelet therapy requires precise, documented coordination between the orthopaedic surgeon, the anesthesiologist, and the primary care physician or cardiologist. The risk of catastrophic hematoma formation—particularly with neuraxial techniques or deep peripheral nerve blocks (e.g., infraclavicular blocks where the vessels are non-compressible)—must be carefully weighed against the profound benefits of regional anesthesia. We strictly adhere to the updated American Society of Regional Anesthesia and Pain Medicine (ASRA) guidelines to dictate the safe cessation and resumption of these potent medications.
American Society of Regional Anesthesia Guidelines for Anticoagulation
| Medication | Discontinuation Recommendation |
|---|---|
| Herbal medications: ginkgo, ginseng, and garlic | No discontinuation |
| Nonsteroidal anti-inflammatories and acetaminophen | No discontinuation |
| Ticlopidine and clopidogrel | 14 days (Clopidogrel often 5-7 days in updated literature, but 14 days for Ticlopidine) |
| Heparin SQ (Prophylactic) | No discontinuation (Wait 4-6 hours after dose for block) |
| IV Heparin (Therapeutic) | Stop 4-6 hours prior; check aPTT |
| Low-molecular-weight heparin (Prophylactic) | 12 hours after last dose |
| Low-molecular-weight heparin (Therapeutic) | 24 hours after last dose |
| Coumadin (Warfarin) | Discontinue 4-5 days; normalize INR < 1.2 |
| Thrombolytics | Avoid regional anesthesia |
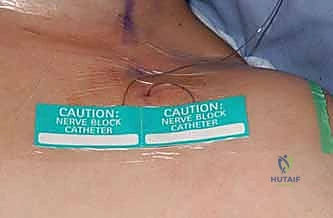
Step-by-Step Surgical Approach and Anesthetic Technique
The choice of anesthesia for upper extremity surgery is a carefully calculated intraoperative strategy. While general anesthesia provides complete unconsciousness, amnesia, and muscle relaxation, the integration of regional anesthesia has revolutionized our approach, particularly for complex reconstructions and arthroplasties.
The Power and Precision of Regional Anesthesia
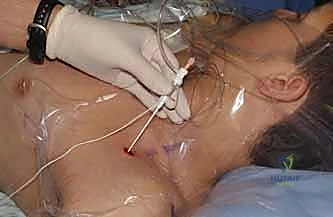
Regional anesthesia, specifically ultrasound-guided peripheral nerve blocks, offers unparalleled advantages. By directly anesthetizing the brachial plexus, we achieve profound, site-specific analgesia that significantly reduces intraoperative volatile anesthetic requirements and drastically curtails postoperative opioid consumption. This translates directly to a lower incidence of PONV, urinary retention, and postoperative ileus. Furthermore, regional blocks facilitate faster emergence, expediting PACU discharge and enhancing overall OR efficiency. Notably, by blunting the surgical stress response and providing superior pain control, regional anesthesia has been shown to reduce the incidence of Postoperative Cognitive Dysfunction (POCD) from 9.8% to 3.5% in vulnerable populations.
The specific approach to the brachial plexus is dictated by the surgical site:
- Interscalene Block: The gold standard for shoulder and proximal humerus surgery. It reliably targets the C5-C7 roots. However, it almost universally results in an ipsilateral phrenic nerve palsy (hemidiaphragmatic paresis), making it relatively contraindicated in patients with severe pulmonary disease or contralateral diaphragmatic dysfunction.
- Supraclavicular Block: Often termed the "spinal of the arm," this block targets the trunks/divisions as they cross the first rib. It provides dense, rapid-onset anesthesia for the entire upper extremity distal to the shoulder. The primary risk here, historically, was pneumothorax, though ultrasound guidance has reduced this incidence to near zero.
- Infraclavicular Block: Targeting the cords of the brachial plexus below the clavicle, this approach is excellent for elbow, forearm, and hand surgery. Its anatomical location makes it ideal for securing continuous perineural catheters, as the chest wall provides a stable fixation point.
- Axillary Block: Performed in the axilla targeting the terminal branches (radial, median, ulnar). It is highly effective for forearm and hand surgery. Crucially, the musculocutaneous nerve exits the plexus proximally and must be blocked separately within the coracobrachialis muscle to ensure complete coverage, particularly for tourniquet tolerance.
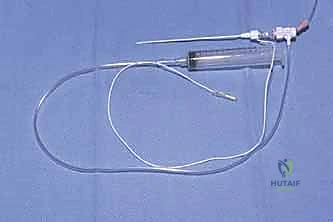
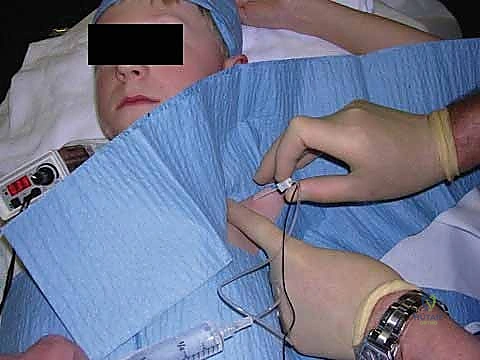
Continuous Regional Analgesia
For procedures anticipated to generate severe, prolonged postoperative pain (e.g., major joint arthroplasty, complex trauma reconstruction), single-shot blocks are insufficient. Here, we utilize continuous regional analgesia. By placing an indwelling perineural catheter under ultrasound guidance, we can connect the patient to an elastomeric pump that delivers a continuous infusion of dilute local anesthetic (e.g., ropivacaine 0.2%). This maintains a dense analgesic block for 48 to 72 hours, effectively bridging the patient through the most acute phase of the inflammatory pain cycle.

Complications, Incidence Rates, and Salvage Management
Despite the profound benefits of our advanced anesthetic techniques, complications can and do occur. As orthopaedic surgeons, we must be intimately familiar with the presentation and immediate management of these adverse events, as they often manifest while the patient is under our direct care in the OR or immediately postoperatively.
The most feared complication of regional anesthesia is Local Anesthetic Systemic Toxicity (LAST). This occurs due to inadvertent intravascular injection or rapid systemic absorption of the local anesthetic, leading to profound central nervous system excitation (seizures, perioral numbness, tinnitus) rapidly followed by cardiovascular collapse (refractory arrhythmias, asystole). Immediate salvage management requires halting the injection, securing the airway, and the rapid administration of 20% Intravenous Lipid Emulsion (Intralipid), which acts as a "lipid sink" to sequester the circulating local anesthetic molecules.
Nerve injury, ranging from transient neurapraxia to permanent axonotmesis, is another critical concern. While the incidence of severe, permanent injury is exceedingly low (<0.04% with ultrasound guidance), transient paresthesias can occur in up to 10% of patients. Management is initially expectant, with thorough neurological documentation. If deficits persist beyond the expected duration of the block, urgent electromyography (EMG) and neurology consultation are warranted to differentiate between block-induced injury, surgical stretch/compression, or pre-existing pathology.
Complications, Incidence Rates, and Salvage Management
| Complication | Estimated Incidence | Clinical Presentation | Salvage Management / Protocol |
|---|---|---|---|
| Local Anesthetic Systemic Toxicity (LAST) | < 0.01% | Tinnitus, metallic taste, perioral numbness, agitation, seizures, followed by cardiovascular collapse / arrhythmias. | 1. Stop injection. 2. Call for help/code cart. 3. Airway management (100% O2). 4. Administer 20% Lipid Emulsion (1.5 mL/kg bolus, followed by 0.25 mL/kg/min infusion). 5. Avoid vasopressin; use small doses of epinephrine. |
| Iatrogenic Nerve Injury | Transient: 3-10% Permanent: < 0.04% |
Persistent motor or sensory deficit beyond the expected pharmacological duration of the block. Severe pain upon injection. | Preventative: Ultrasound guidance, avoid injection against high resistance. Salvage: Detailed neuro exam, early EMG (at 3-4 weeks) to establish baseline, Gabapentinoids for neuropathic pain, Neurology consult. |
| Pneumothorax | < 0.1% (with US guidance) | Pleuritic chest pain, dyspnea, decreased breath sounds, hypoxemia. Most common with supraclavicular blocks. | Upright CXR or lung ultrasound. Small (<20%) asymptomatic: observation and supplemental O2. Large/symptomatic: Tube thoracostomy (chest tube) placement. |
| Phrenic Nerve Palsy | 100% (Interscalene) 10-20% (Supraclavicular) |
Dyspnea, orthopnea, elevated hemidiaphragm on CXR. | Preoperative screening: avoid in patients with severe COPD or contralateral palsy. Management is supportive; function typically returns as the block resolves. |
| Hematoma Formation | Rare (< 0.1%) | Expanding mass at injection site, sudden onset of severe pain, compressive neuropathy. | Preventative: Strict adherence to ASRA anticoagulation guidelines. Salvage: Manual compression, urgent vascular surgery consult if expanding rapidly or compromising distal perfusion. |
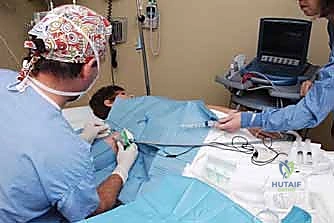
Phased Post-Operative Rehabilitation Protocols
The success of an upper extremity orthopaedic procedure is heavily reliant on the postoperative rehabilitation phase. The anesthetic plan plays a pivotal role in facilitating this transition. The primary goal is to provide a seamless bridge from intraoperative surgical anesthesia to manageable, outpatient oral analgesia, thereby enabling early, pain-free mobilization.
For patients discharged with continuous peripheral nerve catheters and elastomeric pumps, meticulous patient education is required. Patients must be instructed on how to protect the insensate limb to prevent secondary injury (e.g., resting the arm on a hot surface or sleeping in a position that causes profound nerve compression). They must also be educated on the signs of local anesthetic toxicity, catheter site infection, and the mechanics of pump removal, which is typically performed at home by the patient or a caregiver on postoperative day 2 or 3.
A critical phenomenon to anticipate and manage is "rebound pain"—a severe, disproportionate spike in pain that occurs as the regional block precipitously wears off. To mitigate this, a robust multimodal oral analgesic regimen must be initiated before the block resolves. This typically includes scheduled acetaminophen, a non-steroidal anti-inflammatory drug (NSAID) such as meloxicam or celecoxib (if not contraindicated by bone healing concerns), and a neuropathic agent like gabapentin. Opioids are reserved strictly for breakthrough pain. By overlapping these oral medications with the waning regional block, we ensure a smooth transition, allowing the patient to engage effectively with early physical therapy protocols, which is vital for preventing stiffness and ensuring optimal functional recovery.
Summary of Landmark Literature and Clinical Guidelines
The evolution of upper extremity anesthesia is deeply rooted in evidence-based medicine. Mastery of this domain requires familiarity with the landmark literature that shapes our current clinical guidelines.
The American Society of Regional Anesthesia and Pain Medicine (ASRA) guidelines on regional anesthesia in the patient receiving antithrombotic or thrombolytic therapy remain the definitive text for perioperative anticoagulation management. These guidelines are continuously updated and dictate our protocols to prevent catastrophic hemorrhagic complications.
Furthermore, literature regarding Enhanced Recovery After Surgery (ERAS) protocols for upper extremity procedures heavily emphasizes the transition away from opioid-centric general anesthesia towards multimodal, regional-based techniques. Studies have consistently demonstrated that the utilization of ultrasound-guided brachial plexus blocks not only provides superior pain scores in the first 48 hours postoperatively but also significantly reduces the incidence of PONV, decreases time to PACU discharge, and
Clinical & Radiographic Imaging Archive