Mastering Cryosurgical Ablation for Bone Tumors: An Intraoperative Guide
Key Takeaway
This masterclass provides an immersive, step-by-step guide to cryosurgical ablation of bone tumors. Fellows will learn intricate surgical anatomy, precise intraoperative techniques for both direct pour and closed argon systems, meticulous tumor removal, and robust reconstruction methods. We cover crucial pearls, potential pitfalls, and comprehensive postoperative management, ensuring successful outcomes for challenging benign-aggressive and low-grade malignant lesions.
Comprehensive Introduction and Patho-Epidemiology
Welcome to the operating theater and to this definitive exploration of a fascinating, highly effective technique in musculoskeletal oncology: cryosurgical ablation of bone tumors. As orthopedic surgeons, we are constantly navigating the delicate balance between achieving absolute local tumor control and preserving maximum biomechanical function and native anatomy. Historically, many locally aggressive benign tumors and low-grade malignancies required morbid en bloc resections, leading to complex reconstructions, allografts, or endoprostheses with finite lifespans. Cryosurgical ablation, when executed with meticulous precision, represents a paradigm shift. It is not merely the indiscriminate freezing of tissue; it is a highly controlled, targeted intralesional approach that extends our surgical margin microscopically while preserving the macro-structural integrity of the host bone, often circumventing the need for extensive resections or amputation.
To master this technique, one must first deeply understand the underlying patho-epidemiology of the lesions we treat and the rigorous science of cryobiology. Cryoablation serves as a potent physical adjuvant to meticulous mechanical curettage. It is primarily deployed against benign-aggressive lesions, most notably Giant Cell Tumor of Bone (GCT), Aneurysmal Bone Cysts (ABC), and Chondroblastoma, as well as select low-grade malignancies like grade 1 chondrosarcoma, and specific metastatic deposits where palliation and structural preservation are paramount. The epidemiological behavior of these tumors—specifically their propensity to form microscopic satellite nodules or "skip" lesions within the reactive zone of the surrounding cancellous bone—is precisely why mechanical curettage alone yields unacceptably high recurrence rates. Cryoablation effectively sterilizes this 1 to 2-centimeter reactive margin.
The mechanisms of tissue necrosis induced by extreme cold in situ are broadly categorized into immediate cytotoxic effects and delayed progressive necrosis. Immediate cytotoxicity is governed by the rapid formation of ice crystals. As liquid nitrogen (boiling point -196°C) or argon gas is applied, ice crystals nucleate rapidly in the extracellular spaces. This creates a hyperosmotic extracellular environment, drawing water out of the cells and leading to severe cellular dehydration and toxic intracellular electrolyte concentrations. Furthermore, the rapid freezing rate crucial to this technique promotes simultaneous intracellular ice crystal formation, which physically shears and disrupts cell membranes and vital organelles. The sudden, drastic drop in temperature induces profound thermal shock, leading to irreversible denaturation of cellular proteins and immediate cell death.
Beyond these immediate cellular destructive forces, delayed mechanisms ensure the durability of the ablation. Cryoablation inflicts catastrophic damage to the microvasculature within the frozen tissue zone. Endothelial cells are exquisitely sensitive to cold injury. The resulting endothelial disruption leads to profound vascular stasis, microthrombosis, and eventual widespread infarction of the treated tissue bed. This microvascular failure deprives any surviving peripheral tumor cells of oxygen and nutrients, culminating in progressive, delayed necrosis over the ensuing days to weeks. Histologically, this manifests as a well-demarcated 1 to 2 cm rim of extensive bone marrow necrosis, followed by gradual liquefaction and dense fibrosis, characterized by large, permanently thrombosed vessels. Crucially, repeated freeze-thaw cycles exponentially enhance this tissue necrosis. The initial freeze alters the thermal conductivity of the tissue, allowing subsequent cycles to penetrate deeper and freeze faster. Therefore, our standard protocol mandates multiple cycles of rapid freezing followed by spontaneous, slow thawing to maximize the lethal intracellular recrystallization effect.
Detailed Surgical Anatomy and Biomechanics
Executing a flawless cryosurgical ablation requires a profound appreciation of local surgical anatomy, specifically the neurovascular proximity, and the altered biomechanics of the bone post-ablation. The thermal conductivity of bone varies significantly between the dense cortical shell and the porous cancellous medullary canal. Cortical bone, being denser, conducts cold more efficiently but is also more susceptible to thermal necrosis and subsequent structural failure. When we apply a cryogen to a curetted cavity, the thermal front propagates radially. We must acutely understand what lies immediately beyond the thin cortical shell we are preserving.
The concept of the "heat sink" effect is a critical anatomical and physiological consideration. Major blood vessels adjacent to the tumor site—such as the popliteal artery in distal femur lesions, the axillary vessels in proximal humerus lesions, or the radial artery in distal radius tumors—act as massive thermal conduits. The constant flow of core-temperature blood rapidly dissipates the cold energy, creating a thermal barrier that can protect adjacent tumor cells from reaching lethal temperatures (typically -20°C to -40°C for complete necrosis). Conversely, this heat sink effect can be protective of the vessels themselves, though direct freezing of a major neurovascular bundle will still cause catastrophic intimal damage and thrombosis. Therefore, meticulous dissection, mobilization, and physical protection of these structures using insulating materials (like warm saline-soaked sponges or Gelfoam) are non-negotiable steps in the anatomical approach.
Biomechanically, the bone is significantly compromised by both the tumor's osteolysis and our surgical intervention. Creating a large cortical window for access inherently creates a stress riser. The geometry of this window is paramount; it must be elliptical or oval, with its long axis parallel to the diaphyseal shaft, and devoid of sharp corners that concentrate stress. Furthermore, the cryoablation process itself induces avascular necrosis of the remaining host bone rim. While this rim acts as a biological scaffold for eventual creeping substitution, it is mechanically brittle in the immediate postoperative period. The tensile and compressive strengths of the frozen bone are temporarily altered, and the subsequent necrotic phase significantly increases the risk of postoperative pathological fracture.
Because of this profound biomechanical degradation, prophylactic internal fixation and robust defect reconstruction are almost universally required. The cavity is typically filled with polymethylmethacrylate (PMMA) bone cement, which provides immediate structural stability, acts as a secondary thermal adjuvant during its exothermic polymerization phase, and provides a radiopaque background for detecting future radiolucent recurrences. If the subchondral bone plate is exceedingly thin (less than 3-5 mm), the heat of the PMMA and the cold of the cryogen can cause full-thickness cartilage necrosis and joint collapse. In such cases, a composite reconstruction using a layer of structural bone graft adjacent to the joint surface, backed by PMMA and internal fixation (plates and screws or intramedullary nails), is biomechanically necessary to span the defect and bypass the stress risers.
Exhaustive Indications and Contraindications
Patient selection and rigorous adherence to histologic and morphologic criteria are the cornerstones of successful cryosurgical ablation. This technique is a powerful tool, but it is not a panacea for all musculoskeletal tumors. The primary indication remains the locally aggressive benign bone tumor that exhibits a high propensity for local recurrence following simple curettage. Giant Cell Tumor of Bone (GCT) is the classic paradigm for this technique. Despite its benign histology, GCT can be locally devastating, destroying the articular surface and surrounding cortex. Cryoablation allows for joint salvage in periarticular GCTs where wide resection would necessitate a massive endoprosthesis and significant functional morbidity.
Other benign-aggressive lesions highly amenable to this approach include Aneurysmal Bone Cysts (ABCs), particularly recurrent ABCs or those in anatomically challenging locations where complete marginal resection is hazardous. Chondroblastoma, an epiphyseal lesion, is another excellent candidate, though extreme care must be taken to protect the adjacent articular cartilage and the physis in skeletally immature patients. Simple Bone Cysts (SBCs), Fibrous Dysplasia, Enchondromas, Eosinophilic Granuloma, Osteoblastoma, and Chondromyxoid Fibroma can also be treated with cryoablation, particularly when they present with aggressive features, cortical breakthrough, or have recurred after initial conservative surgical management.
In the realm of malignant disease, cryoablation is generally reserved for specific, low-grade scenarios or palliative intent. Low-grade (Grade 1) chondrosarcoma, particularly secondary chondrosarcomas arising from pre-existing enchondromas in the appendicular skeleton, can often be managed with extended intralesional curettage and cryoablation, avoiding the morbidity of wide en bloc resection. Furthermore, cryoablation is an invaluable tool in the management of metastatic bone disease. In patients with oligometastatic disease or those requiring local tumor control to alleviate severe pain and prevent impending pathological fractures, cryoablation provides excellent local control while preserving the bone stock necessary for stable palliative reconstruction.
However, strict contraindications exist and must be respected. High-grade sarcomas (e.g., Osteosarcoma, Ewing Sarcoma, high-grade Chondrosarcoma) are absolute contraindications for intralesional therapy; these require wide, en bloc resection with negative oncologic margins. Furthermore, if the tumor has caused such extensive cortical destruction that a circumferential rim of host bone cannot be maintained to contain the liquid cryogen or support the reconstruction, en bloc resection is favored. Active local infection is also an absolute contraindication, as the necrotic bone produced by cryoablation will serve as a massive nidus for persistent sepsis.
Indications and Contraindications Summary
| Category | Specific Conditions / Scenarios | Rationale / Clinical Note |
|---|---|---|
| Primary Indications (Benign-Aggressive) | Giant Cell Tumor (GCT), Aneurysmal Bone Cyst (ABC), Chondroblastoma, Osteoblastoma, Chondromyxoid Fibroma. | High local recurrence rate with simple curettage; cryo sterilizes the 1-2cm reactive margin, preserving joint function. |
| Primary Indications (Low-Grade Malignant) | Low-grade (Grade 1) Chondrosarcoma (appendicular skeleton). | Avoids morbid en bloc resection; provides adequate oncologic control for non-metastasizing, slow-growing lesions. |
| Palliative / Metastatic Indications | Solitary or oligometastatic bone lesions (e.g., Renal Cell, Thyroid, Breast). | Achieves local tumor control, relieves pain, and preserves bone stock for durable orthopedic reconstruction. |
| Absolute Contraindications | High-grade primary sarcomas (Osteosarcoma, Ewing, Grade 2/3 Chondrosarcoma). | Intralesional margins are oncologically unacceptable; high risk of local recurrence and systemic dissemination. |
| Morphologic Contraindications | Massive cortical destruction lacking a circumferential rim; extensive soft tissue extension. | Cannot contain the cryogen (risk of massive soft tissue necrosis); insufficient remaining bone for stable reconstruction. |
| Medical Contraindications | Active osteomyelitis or severe local soft tissue infection. | Cryo-induced osteonecrosis provides an avascular nidus, guaranteeing persistent, uncontrollable infection. |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough, exhaustive preoperative planning is non-negotiable in musculoskeletal oncology. A cryosurgical ablation is not a procedure one improvises at the scrub sink; it requires a meticulous, three-dimensional understanding of the tumor's extent, its relationship to critical anatomy, and a precise reconstructive strategy. High-resolution, multi-planar magnetic resonance imaging (MRI) is the gold standard and absolute prerequisite. T1-weighted, T2-weighted STIR, and T1 post-gadolinium contrast sequences are essential for defining the true intraosseous extent of the tumor, identifying any subtle cortical breaches, and mapping the extraosseous soft tissue component. The MRI also delineates the proximity of the tumor to major neurovascular bundles and the exact distance to the articular cartilage.
Complementing the MRI, high-resolution computed tomography (CT) is invaluable for assessing the integrity of the cortical shell and the subchondral bone plate. CT provides the best architectural map for planning the cortical window and assessing the risk of postoperative fracture. Baseline plain radiographs in orthogonal planes remain crucial for initial assessment, overall bone morphometry, and long-term postoperative comparison.
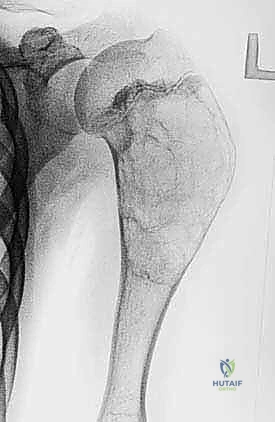
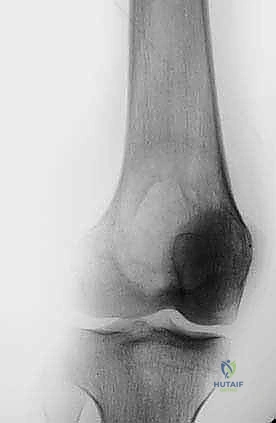
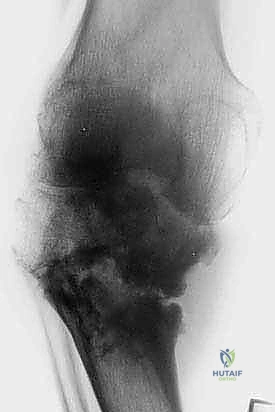
Example: Plain radiograph demonstrating an expansile, lytic lesion of the proximal humerus, consistent with an aneurysmal bone cyst, requiring careful assessment of cortical thinning.
Example: Radiographic evaluation of a giant cell tumor of the proximal tibia, highlighting the subchondral involvement and the necessity for joint-preserving techniques.
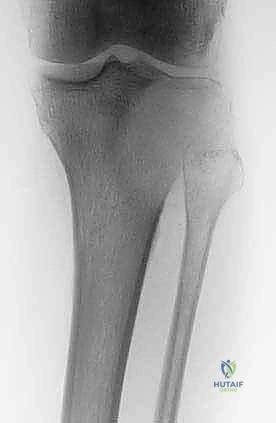
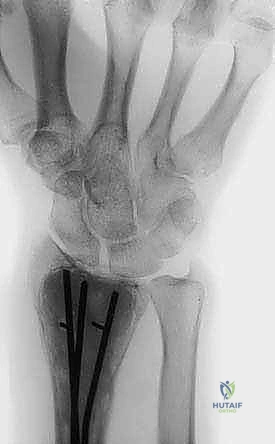
Examples: Advanced imaging of a recurrent low-grade chondrosarcoma of the distal radius, noting precise anatomical location, cortical integrity, and intramedullary extension.
Templating is the next critical phase. Using our advanced imaging, we must digitally or physically template the planned cortical window. We determine the exact dimensions required to access the farthest reaches of the tumor cavity with our curettes and burrs. Simultaneously, we must template the hardware placement for the prophylactic reconstruction—selecting the appropriate plates, screws, or intramedullary devices that will bypass the stress riser of our window and the ablated bone segment. Anticipating the volume of PMMA or structural bone graft required is also done at this stage.
Patient positioning and operating room setup are dictated entirely by the tumor's anatomical location, with the dual goals of achieving unhindered surgical exposure and facilitating gravity-dependent application of the cryogen (if using the direct pour technique). C-arm fluoroscopy must be draped into the sterile field and pre-positioned to ensure perfect anteroposterior (AP) and lateral views of the target lesion without the need to awkwardly reposition the patient mid-procedure. For extremity lesions, a pneumatic tourniquet is universally applied proximally. The tourniquet is critical not only for visualization during curettage but also for temporarily eliminating the local blood flow, which, as previously discussed, acts as a massive heat sink that can thwart the efficacy of the freeze cycle.
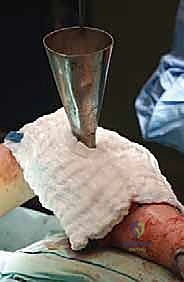
The cryoablation system must be rigorously checked before the patient enters the room. If utilizing the traditional direct pour liquid nitrogen technique, the insulated Dewar flask, stainless steel funnels, and insulated delivery cans must be sterile and immediately available.

Setup for direct pour technique, ensuring all funnels and insulated delivery systems are prepped and sterile.
If utilizing a modern closed argon gas system, the console must be tested, and the appropriate array of cryoprobes and conductive gel medium must be selected based on the cavity volume. Temperature monitoring is paramount; multiple sterile thermocouple probes must be calibrated and ready for percutaneous insertion to monitor the thermal spread in real-time, preventing catastrophic damage to adjacent nerves or skin.
Preparation of the closed argon gas cryoablation system, confirming probe functionality and console settings prior to incision.
Specific positioning examples highlight these principles. For a proximal humerus lesion, the patient is placed in a modified beach chair position with the arm draped free, allowing for an extensile deltopectoral approach. For a distal femur GCT, the patient is supine with a bump under the ipsilateral hip and the knee flexed over a sterile triangle to 90 degrees, allowing gravity to assist in pooling the liquid nitrogen in the condylar cavity. Distal radius tumors require the patient supine with the arm extended on a radiolucent hand table, allowing for precise dorsal or volar approaches while protecting the complex neurovascular anatomy of the wrist.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution of a cryoablation is a highly orchestrated, multiphase procedure. We divide this into five distinct, unforgiving stages. Meticulous attention to detail in each stage is what separates a durable oncologic cure from a disastrous local recurrence or structural failure.
Stage 1: Tumor Exposure and Cortical Window Creation
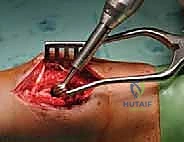
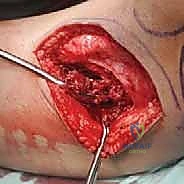
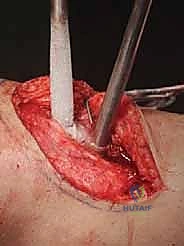
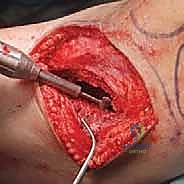
The initial goal is wide, safe exposure of the involved bone, ensuring clear margins for the cortical window while rigorously protecting the soft tissues. The skin incision is placed directly over the planned window, utilizing standard extensile approaches. Fasciocutaneous flaps must be raised meticulously and kept thick to maintain their viability, as they will be exposed to significant thermal stress.
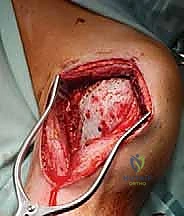
Extensile exposure of the proximal humerus, demonstrating meticulous handling of the soft tissue envelope.
Neurovascular structures are identified early, mobilized gently, and protected with vessel loops and moist laparotomy sponges. Once the bone is exposed, the tourniquet is inflated. Using fluoroscopic guidance, we confirm our anatomical landmarks. The planned cortical window is marked with a sterile pen or electrocautery.
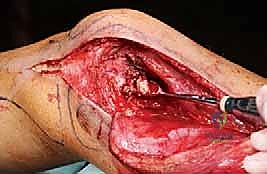
Marking the elliptical cortical window, ensuring dimensions are adequate for complete intralesional access.
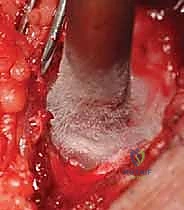
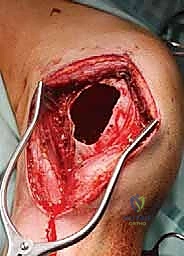
The window must be elliptical to minimize stress risers. We typically outline the window using a high-speed burr or multiple drill holes, which are then carefully connected with sharp osteotomes. The cortical lid is removed and can sometimes be sterilized and replaced later, though it is often discarded if heavily involved with tumor.
Creation of the cortical window using drill holes connected by osteotomes, preserving the structural integrity of the surrounding shaft.
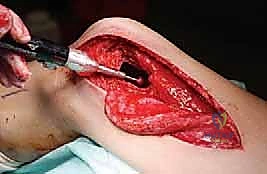
Stage 2: Aggressive Intralesional Curettage
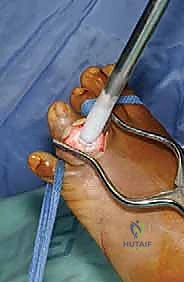
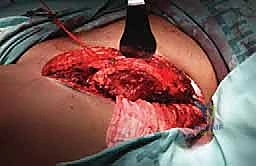
Fellows, hear me clearly: cryoablation is an adjuvant. It cannot overcome poor mechanical curettage. This stage is the most critical determinant of local control. We begin with large curettes, systematically removing the gross tumor volume. As we approach the cortical margins, we switch to progressively smaller, angled curettes to scrape every recess, ridge, and contour of the intramedullary cavity.
Aggressive mechanical curettage, systematically clearing the gross tumor volume from the medullary cavity.
Once gross tumor is removed, we utilize a high-speed power burr (often a large round diamond or cutting burr) to systematically break down the bony ridges and extend our margin 1-2 mm into normal-appearing cancellous bone. This breaks open the marrow spaces, allowing the cryogen to penetrate deeply. Copious pulsatile lavage is used repeatedly to clear debris and visualize the cavity. We do not stop until we have a completely clean, white bony cavity circumferentially.
High-speed burring of the cavity walls, a mandatory step to break down bony ridges and prepare the bed for thermal ablation.
Stage 3: Cryogen Application and Thermal Monitoring
With the cavity prepared and completely dry, we proceed to the ablation phase. The surrounding soft tissues, skin edges, and neurovascular bundles are heavily protected. We pack the periphery of the cortical window with warm, saline-soaked laparotomy sponges or continuous warm saline irrigation to create a thermal barrier. Gelfoam is often packed tightly around the bony edges to prevent the cryogen from spilling into the soft tissues. Thermocouple probes are placed percutaneously adjacent to critical nerves or just deep to the skin to ensure these tissues do not drop below 10°C.
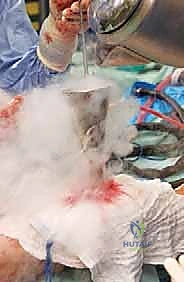
If using the direct pour technique, a sterile stainless steel funnel is placed securely into the window. Liquid nitrogen is poured slowly and continuously.
Direct pour technique: Liquid nitrogen is carefully introduced into the cavity through a sterile funnel.
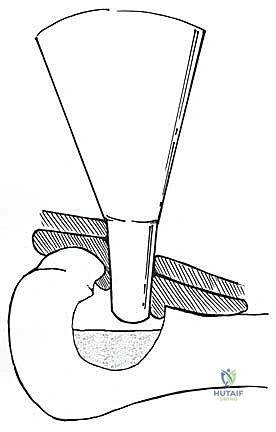
The cavity fills with liquid nitrogen, initiating the rapid freeze cycle and profound thermal shock to the microscopic disease.
We observe the rapid freezing of the cavity walls. The bone turns stark white. We maintain the pool of liquid nitrogen for a predetermined time (usually 2 to 5 minutes, depending on the cavity size and proximity to articular cartilage).
Visualization of the frozen cortical rim, demonstrating the extensive thermal penetration achieved during the cycle.
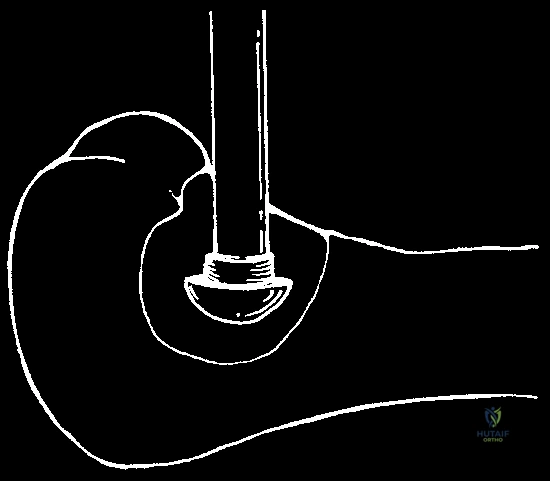
Alternatively, if using a closed argon system, the probes are inserted into the cavity, which may be filled with a conductive gel to ensure uniform thermal transfer.
Application of a closed argon gas probe system, utilizing conductive gel for precise, monitored freezing.
Iceball formation using the closed argon system, allowing for highly controlled, predictable margins of necrosis.
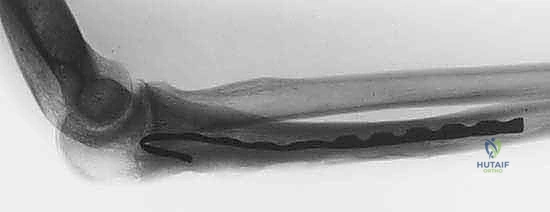
Stage 4: Thawing and Lavage
Following the rapid freeze, we allow for a spontaneous, slow thaw. Do not irrigate with warm saline to speed this up. The slow thaw allows for intracellular ice recrystallization, a process that is highly lethal to any surviving tumor cells. The thaw phase typically takes 10 to 15 minutes. Once the ice has completely melted and the bone returns to its normal color, the cavity is vigorously irrigated to remove the necrotic cellular debris. This entire freeze-thaw cycle is then repeated at least once, and often twice (a total of two to three cycles). The subsequent cycles freeze faster and penetrate deeper due to the altered thermal conductivity of the already-frozen bone.
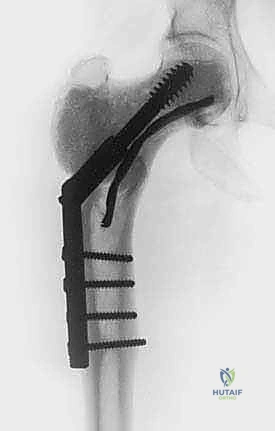
Stage 5: Defect Reconstruction and Prophylactic Fixation
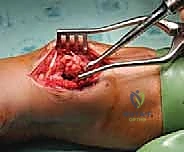
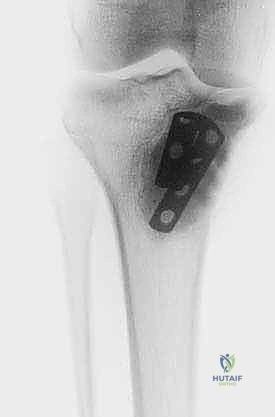
Following the final thaw and lavage, the cavity is meticulously dried. The bone is now structurally compromised and functionally dead. Reconstruction is mandatory. The cavity is typically packed with PMMA bone cement. The cement is pressurized into the cancellous interstices, providing immediate structural support and acting as a secondary thermal and chemical adjuvant.
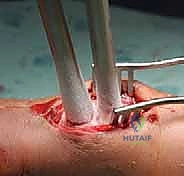
Preparation for reconstruction: The ablated cavity is ready to receive PMMA or structural bone graft.
Application of PMMA bone cement into the defect, providing immediate structural stability and a radiopaque background.
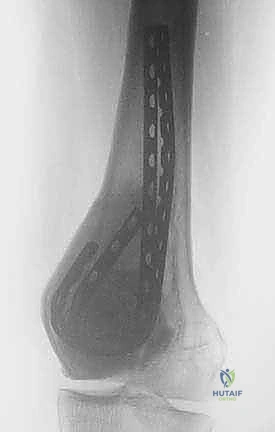
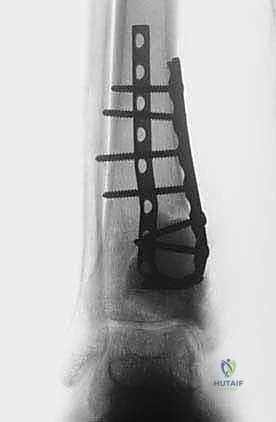
If the subchondral plate is paper-thin, a layer of cancellous or structural allograft/autograft may be placed against the cartilage before cementing to prevent thermal injury to the joint from the exothermic curing of the PMMA. Finally, to bypass the stress risers of the cortical window and the necrotic bone segment, rigid internal fixation is applied. This usually involves locking plates spanning the defect.
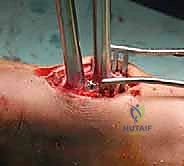

Intraoperative fluoroscopy and clinical images confirming the placement of robust internal fixation spanning the reconstructed defect.
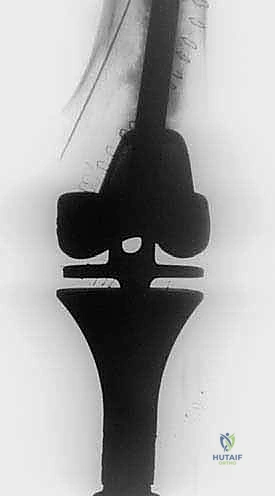
Final radiographic verification of the reconstruction, demonstrating excellent alignment, hardware placement, and complete filling of the defect.
Complications, Incidence Rates, and Salvage Management
Despite meticulous technique, cryosurgical ablation carries a distinct and significant complication profile. As academic surgeons, we must counsel our patients transparently and be prepared to manage these complex sequelae. The most common and devastating complication is a postoperative pathological fracture. The incidence of fracture ranges from 5% to 15%, depending heavily on the anatomical location, the size of the cortical window, and the adequacy of prophylactic fixation. The profound thermal necrosis weakens the bone significantly, and the necrotic phase peaks between 4 to 8 weeks postoperatively, making this the highest-risk window for structural failure. Salvage management for a fracture typically involves revision open reduction and internal fixation (ORIF), often requiring longer spanning plates, structural allografts, or in severe cases of joint collapse, conversion to a megaprosthesis or total joint arthroplasty.
Infection is another dreaded complication, occurring in 2% to 5% of cases. The massive volume of necrotic bone created by the cryoablation serves as an ideal, avascular nidus for bacterial colonization. If a deep infection occurs, it is rarely eradicated with suppressive antibiotics alone. Salvage requires aggressive surgical debridement, removal of all hardware and PMMA, placement of antibiotic-loaded cement spacers, and long-term intravenous antibiotics, often followed by a complex, multi-staged reconstruction once the infection is cleared.
Neurologic injury, primarily due
Clinical & Radiographic Imaging Archive