Masterclass: Extra-Articular Resection and Reconstruction of Supra-acetabular Metastatic Lesions
Key Takeaway
This masterclass details the surgical technique for resecting and reconstructing supra-acetabular metastatic lesions, preserving the hip joint. We cover meticulous preoperative planning, precise patient positioning, and a step-by-step intraoperative guide, emphasizing anatomical protection and optimal hardware application. Learn critical pearls, pitfalls, and comprehensive postoperative management to ensure immediate ambulation and superior patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
The management of metastatic bone disease in the pelvis represents one of the most formidable challenges in orthopedic oncology. Supra-acetabular metastatic lesions, in particular, threaten the critical weight-bearing axis of the human body, compromising the transfer of forces from the axial skeleton to the lower appendicular skeleton. When these lesions expand, they induce severe, intractable mechanical pain and pose an imminent risk of catastrophic pathological fracture. The primary objective of surgical intervention is not curative, but rather palliative and functional: to achieve immediate structural stabilization, alleviate debilitating pain, and restore ambulatory capacity, thereby maximizing the patient’s quality of life during their remaining survival period.
This masterclass focuses specifically on the extra-articular resection and reconstruction of supra-acetabular metastatic lesions. This highly specialized approach is meticulously tailored for Harrington Class I lesions. In 1981, Kevin Harrington proposed a classification system for peri-acetabular metastatic disease that remains the gold standard for surgical decision-making. Class I lesions are defined by extensive tumor invasion localized to the supra-acetabular region, with structurally intact medial and inferior walls, and critically, a preserved acetabular articular surface. By confirming the integrity of the subchondral bone and articular cartilage, the orthopedic oncologist can successfully perform a joint-preserving, extra-articular curettage and reconstruction, sparing the patient the immense physiological burden and morbidity associated with a complex total hip arthroplasty or hemipelvic replacement.
The pathophysiology of these lesions typically involves a vicious cycle of osteoclast-mediated bone resorption driven by tumor-secreted factors such as Parathyroid Hormone-related Protein (PTHrP). Carcinomas of the breast, prostate, lung, kidney, and thyroid are the most frequent culprits to metastasize to the periacetabular region. Lytic lesions, common in renal and thyroid carcinomas, destroy the trabecular architecture, leading to rapid mechanical failure. Blastic lesions, more typical of prostate cancer, produce disorganized, structurally inferior woven bone that is equally prone to mechanical insufficiency. Understanding the primary histology is essential, as it dictates the vascularity of the tumor, the expected response to adjuvant therapies, and the anticipated life expectancy, all of which heavily influence the surgical strategy.
Detailed Surgical Anatomy and Biomechanics
A profound understanding of the regional anatomy and the biomechanical forces acting upon the pelvis is the absolute foundation of a safe and durable reconstruction. The supra-acetabular corridor is the primary conduit for load transmission during the stance phase of gait. Forces equivalent to multiple times the patient's body weight are transmitted from the sacrum, across the sacroiliac joint, through the thick cortical bone of the sciatic buttress and the supra-acetabular ilium, and directly into the femoral head. A metastatic void in this region creates a massive stress riser, predisposing the acetabular roof to shear failure and superior migration of the femoral head.
The surgical approach to this region necessitates navigating a complex, multi-layered muscular envelope. The gluteus maximus, originating from the posterior ilium, sacrum, and sacrotuberous ligament, forms the superficial layer. Deep to the maximus lies the critical abductor mechanism, comprising the gluteus medius and minimus. These muscles originate from the outer table of the ilium and insert onto the greater trochanter. In the posterior approach utilized for supra-acetabular lesions, the interval between the gluteus maximus and the deeper abductors is exploited, requiring anterior retraction of the medius and minimus to expose the supra-acetabular ilium.
The neurovascular topography in this region is unforgiving and demands meticulous respect. The sciatic nerve, the largest nerve in the human body, exits the greater sciatic foramen typically inferior to the piriformis muscle. It courses distally over the short external rotators and must be identified and protected throughout the procedure. Equally critical, yet often underappreciated, is the superior gluteal neurovascular bundle. Exiting the greater sciatic foramen superior to the piriformis, it courses anteriorly between the gluteus medius and minimus. Vigorous or sustained anterior retraction of the abductor mass during exposure of the supra-acetabular ilium can cause traction injury or avulsion of these vessels, leading to devastating abductor weakness (Trendelenburg gait) and massive hemorrhage.
Exhaustive Indications and Contraindications
Patient selection is the most critical determinant of success in orthopedic oncology. The decision to proceed with major pelvic reconstruction must balance the anticipated mechanical benefits against the physiological toll of the surgery, taking into account the patient's overall oncologic prognosis.
Indications for Extra-Articular Reconstruction
Surgical intervention is indicated for patients presenting with intractable mechanical pain that is refractory to non-operative measures, including radiation therapy and pharmacological pain management. A key indication is an impending or completed pathological fracture of the supra-acetabular region where the joint space remains radiographically intact. The patient must have a reasonable life expectancy, generally accepted as greater than three to six months, to justify the surgical morbidity and the rehabilitation period. Furthermore, the lesion must be anatomically amenable to an extra-articular approach, strictly adhering to the criteria for a Harrington Class I defect.
Contraindications to Joint-Preserving Surgery
Absolute contraindications include Harrington Class II (medial wall destruction), Class III (lateral wall/acetabular rim destruction), and Class IV (massive pelvic discontinuity) lesions, all of which require complex arthroplasty or custom mega-prosthetic reconstruction. Direct tumor invasion through the subchondral plate into the hip joint mandates an intra-articular resection and total hip arthroplasty. Severe medical comorbidities that preclude general anesthesia or major blood loss, as well as an extremely poor overall oncologic prognosis (life expectancy less than three months), are strong relative contraindications where palliative care or less invasive stabilization techniques should be prioritized.
| Parameter | Indications for Extra-Articular Recon | Contraindications |
|---|---|---|
| Harrington Class | Class I (Intact medial wall & joint) | Class II, III, IV (Requires THA/Mega-prosthesis) |
| Joint Involvement | Cartilage and subchondral bone intact | Tumor invasion into the articular space |
| Pain Profile | Intractable mechanical pain | Pain manageable with radiation/medication |
| Life Expectancy | > 3 to 6 months | < 3 months (Consider non-operative palliation) |
| Structural Integrity | Impending/actual fracture of roof | Massive pelvic discontinuity |
Pre-Operative Planning, Templating, and Patient Positioning
Before a scalpel touches the skin, meticulous and exhaustive preoperative planning is paramount. The surgeon must construct a precise three-dimensional mental model of the tumor's relationship to the native bone, the hip joint, and the critical neurovascular "no-touch" zones.
Advanced Imaging Modalities
The diagnostic workup begins with high-quality orthogonal plain radiographs of the pelvis and the affected hip. These provide a baseline assessment of the lesion's lytic or blastic morphology and overall pelvic alignment. However, Computed Tomography (CT) is the definitive workhorse for assessing bone stock. We mandate fine-cut axial, coronal, and sagittal reconstructions. Three-dimensional CT modeling is increasingly utilized to template the exact volume of the defect, plan the trajectory of reinforcing pins, and determine the required volume of polymethylmethacrylate (PMMA) cement.

FIG 1 • A. Plain radiograph demonstrating a massive supra-acetabular metastatic lytic lesion. Note that the joint contour and medial wall appear radiographically preserved.
FIG 1 • B. Three-dimensional pelvic model illustrating the exact anatomical location and volume of the supra-acetabular defect.

FIG 1 • C. Preoperative templating on the 3D model demonstrating the planned reconstruction trajectory.
Magnetic Resonance Imaging (MRI) is indispensable for evaluating the soft tissue extent of the tumor and, crucially, the integrity of the articular cartilage. T1-weighted images delineate marrow replacement, while T2-weighted fluid-sensitive sequences are excellent for assessing the cartilage cap. If the MRI shows tumor breaching the subchondral bone and invading the joint space, the extra-articular plan must be aborted in favor of an arthroplasty.
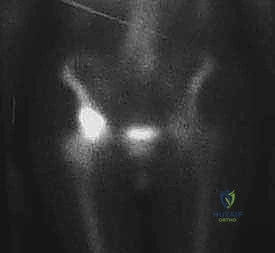
FIG 2 • A. T2-weighted MRI scan confirming a preserved cartilage border of the right hip joint, validating the extra-articular approach.
A whole-body Technetium-99m bone scan or a PET-CT is required to stage the skeletal disease. It is catastrophic to reconstruct a pelvis only to have the patient immediately fracture through an unrecognized distal femoral or subtrochanteric metastasis. Finally, for highly vascular tumors (e.g., renal cell or thyroid carcinoma), preoperative angiography with selective arterial embolization performed 24-48 hours prior to surgery is mandatory to mitigate life-threatening intraoperative hemorrhage.
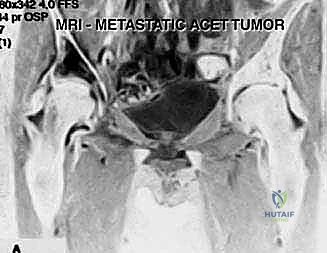
FIG 2 • B. Technetium Tc 99m bone scan showing intense uptake isolated to the supra-acetabular region, with no synchronous lesions in the ipsilateral femur.
Patient Positioning and Operating Room Setup
The patient is placed in the true lateral decubitus position on a radiolucent operating table. Rigid stabilization using a vacuum beanbag and anterior/posterior pelvic supports is critical to prevent shifting during aggressive curettage. All bony prominences, particularly the dependent axilla and the common peroneal nerve at the fibular head, must be meticulously padded. A sterile bump is placed between the knees to maintain neutral hip alignment and prevent excessive tension on the sciatic nerve. The fluoroscopy C-arm must be draped and positioned to allow unimpeded anterior-posterior, obturator oblique, and iliac oblique (Judet) views throughout the procedure without requiring patient repositioning.
Step-by-Step Surgical Approach and Fixation Technique
The surgical execution demands a delicate balance between aggressive oncologic clearance of the tumor and meticulous preservation of structural bone and neurovascular elements.
Incision and Deep Dissection
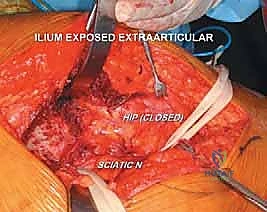
A curvilinear incision is utilized, beginning at the posterior third of the iliac crest and extending distally toward the greater trochanter, following the anterior border of the gluteus maximus. The subcutaneous tissues are divided, and the fascia lata is incised longitudinally. The gluteus maximus is bluntly split in line with its fibers or mobilized by detaching its anterior insertion from the iliotibial tract, allowing the muscle belly to be retracted posteriorly.
Attention is immediately directed to identifying and protecting the sciatic nerve. The nerve is located as it emerges inferior to the piriformis muscle in the deep retrogluteal space. It is carefully mobilized using blunt dissection, and a vessel loop is placed around it. This loop serves as a continuous visual marker, ensuring the nerve is protected from errant retractors or thermal injury from bone cement later in the procedure.

FIG 3 • Intraoperative exposure demonstrating the mobilization of the gluteal musculature and the identification of the deep neurovascular structures.
To expose the supra-acetabular ilium, the gluteus medius and minimus must be elevated from the outer table of the ilium. This is performed using an elevator, working from posterior to anterior. Broad, smooth Hohmann retractors are carefully placed to hold the abductor mass anteriorly. The surgeon must remain hyper-vigilant of the superior gluteal neurovascular bundle during this retraction; excessive tension will lead to irreversible abductor denervation.
Tumor Unroofing and Aggressive Curettage
Once the lateral cortex of the supra-acetabular ilium is exposed, the tumor cavity is unroofed. Depending on the degree of cortical destruction, this may be achieved with an osteotome, a high-speed burr, or simply by penetrating a lytic cortical shell with a curette. A wide cortical window is essential to visualize the entire extent of the tumor cavity.
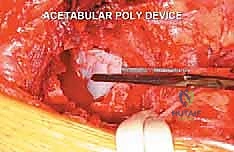
FIG 4 • A. Initial unroofing of the supra-acetabular lateral cortex to access the metastatic lesion.
Intralesional curettage is then performed systematically. Using a combination of straight and angled curettes, the gross tumor is evacuated. The curettage must be aggressive, extending to healthy, bleeding cancellous bone in all directions. The surgeon must exercise extreme caution when curetting inferiorly toward the acetabular dome; breaching the subchondral bone will convert an extra-articular procedure into an intra-articular disaster. Intraoperative fluoroscopy is utilized frequently to confirm the depth and trajectory of the curettes relative to the joint line.
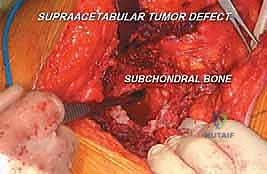
FIG 4 • B. Aggressive intralesional curettage being performed. Note the use of specialized angled curettes to reach the medial and anterior extents of the cavity.
Following gross tumor removal, high-speed burring of the cavity walls is performed to break down the reactive bone and expose the micro-trabecular architecture, which is vital for subsequent cement interdigitation. Adjuvant treatment of the cavity is highly recommended. This typically involves pulsatile lavage followed by the application of anhydrous alcohol, phenol, or an argon beam coagulator to induce necrosis of microscopic residual tumor cells and achieve meticulous hemostasis.
Structural Reconstruction and Cementation
The structural integrity of the supra-acetabular corridor must now be restored. For Harrington Class I lesions, this is achieved using a composite construct of metallic hardware and Polymethylmethacrylate (PMMA) bone cement. The hardware serves as internal rebar, transferring loads from the ilium to the sciatic buttress, while the PMMA provides immediate compressive strength and fills the massive void.
Threaded Steinmann pins (typically 4.5mm to 6.5mm) or specialized pelvic reconstruction screws are driven across the defect. The trajectory is critical: pins are typically anchored superiorly in the dense bone of the iliac crest and directed inferiorly and medially into the thick sciatic buttress and the anterior column, bridging the defect. Fluoroscopy is mandatory during pin placement to ensure they do not penetrate the hip joint or the sciatic notch.
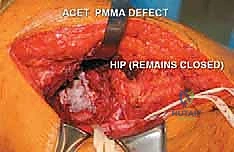
FIG 5 • A. Placement of threaded Steinmann pins across the defect to act as structural rebar, anchored securely into the iliac crest and sciatic buttress.
Once the hardware is positioned and confirmed radiographically, the cavity is thoroughly dried. PMMA bone cement, ideally mixed with antibiotics (such as tobramycin or vancomycin) and radiopaque contrast (barium sulfate), is prepared. In its doughy phase, the PMMA is pressurized into the cavity. Pressurization is critical to force the cement into the cancellous bone interstices, creating a robust micro-interlock. The cement encapsulates the metallic pins, forming a rigid, unified composite construct.
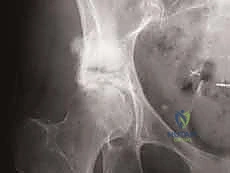
FIG 5 • B. The final composite reconstruction. The defect is completely filled with pressurized PMMA bone cement, which encapsulates the hardware and sits flush with the lateral cortex.
During the exothermic polymerization of the PMMA, the surgeon must continuously irrigate the surrounding soft tissues with cool saline to prevent thermal necrosis of the abductor muscles and the sciatic nerve. Once the cement has fully cured, a final fluoroscopic check is performed to confirm hardware placement, cement fill, and the absence of intra-articular cement extravasation. The wound is then closed in multiple layers over a closed-suction drain.
Complications, Incidence Rates, and Salvage Management
Surgical intervention in the metastatic pelvis carries a high risk of complications due to the compromised physiological state of the patient, the effects of prior radiation or chemotherapy, and the complex regional anatomy. Anticipation and rapid management of these complications are essential.
| Complication | Estimated Incidence | Etiology / Risk Factors | Prevention and Salvage Management |
|---|---|---|---|
| Surgical Site Infection (SSI) | 5% - 15% | Prior radiation, malnutrition, immunosuppression, large dead space. | Prevention: Pre-op optimization, antibiotic-loaded PMMA, meticulous hemostasis. Management: Aggressive surgical debridement, targeted IV antibiotics, vacuum-assisted closure (VAC). |
| Sciatic Nerve Palsy | 2% - 8% | Direct trauma, excessive traction, thermal injury from PMMA. | Prevention: Routine identification and tagging, careful retraction, cool irrigation during cement curing. Management: AFO bracing for foot drop, gabapentinoids, physiotherapy. Rarely reversible if transected. |
| Superior Gluteal N/A Injury | 3% - 10% | Overzealous anterior retraction of gluteus medius/minimus. | Prevention: Gentle, intermittent retraction; release of tendinous insertions if necessary. Management: Gait aids (cane/walker) for resultant Trendelenburg gait; physical therapy. |
| Hardware Failure / Construct Collapse | 5% - 12% | Inadequate pin purchase, poor cement interdigitation, disease progression. | Prevention: Strict adherence to biomechanical principles, bridging to healthy bone. Management: Revision surgery, potentially requiring conversion to complex total hip arthroplasty (Harrington Class II/III management). |
| Venous Thromboembolism (VTE) | 10% - 20% | Hypercoagulable state of malignancy, pelvic surgery, immobility. | Prevention: Mechanical prophylaxis, early mobilization, chemical prophylaxis (LMWH or DOACs) per guidelines. Management: Therapeutic anticoagulation, IVC filter if anticoagulation is contraindicated. |
Phased Post-Operative Rehabilitation Protocols
The primary goal of this procedure is immediate functional restoration. Unlike biological reconstructions that require prolonged periods of restricted weight-bearing for osseous integration, a PMMA-hardware composite construct achieves maximal mechanical strength immediately upon cement polymerization.
Phase I: Immediate Post-Operative (Days 0-14)
Rehabilitation begins on post-operative day one. The patient is typically allowed Weight Bearing As Tolerated (WBAT) with the assistance of a walker or crutches. The focus is on early mobilization to prevent pulmonary complications and VTE. Active and active-assisted range of motion (ROM) of the hip, knee, and ankle are initiated. Hip precautions may be implemented depending on the extent of the posterior soft tissue dissection, though the joint capsule itself is usually not violated.
Phase II: Intermediate Rehabilitation (Weeks 2-6)
As incisional pain subsides and wound healing is confirmed, therapy transitions to gait normalization and muscle re-education. Strengthening of the hip abductors (gluteus medius/minimus) is heavily emphasized, as these muscles undergo significant surgical trauma during the exposure. Patients are transitioned from a walker to a cane, and eventually to independent ambulation as tolerated.
Phase III: Long-Term Maintenance (Weeks 6 and beyond)
The focus shifts to maximizing independence in Activities of Daily Living (ADLs) and integrating the patient back into their oncologic treatment pathway. Routine radiographic surveillance is required every 3 to 6 months to monitor the integrity of the construct and screen for local disease recurrence or progression.
Summary of Landmark Literature and Clinical Guidelines
The foundation of periacetabular metastatic reconstruction was laid by Harrington in 1981 (Harrington KD. The management of acetabular insufficiency secondary to metastatic malignant disease. J Bone Joint Surg Am. 1981). His classification system remains the cornerstone of surgical decision-making, differentiating lesions that can be managed with extra-articular cementation (Class I) from those requiring complex arthroplasty rings or custom implants (Class II-IV).
Modern clinical guidelines, such as those published by the Musculoskeletal Tumor Society (MSTS) and the National Comprehensive Cancer Network (NCCN), reinforce the multidisciplinary approach to these lesions. While Mirels' criteria are predominantly utilized for long bone impending fractures, the underlying principle—that large, painful, lytic lesions in high-stress areas require prophylactic stabilization—applies directly to the supra-acetabular corridor.
Recent literature has focused on the integration of intraoperative navigation and robotic assistance to improve the precision of curettage and the trajectory of hardware placement, minimizing the risk of inadvertent joint penetration. Furthermore, while PMMA remains the standard for Harrington Class I defects, the advent of custom 3D-printed titanium implants is revolutionizing the management of more advanced Class II and III defects, offering highly porous surfaces for biological fixation in patients with extended life expectancies. Nonetheless, for the isolated supra-acetabular defect with an intact joint, the extra-articular curettage and PMMA composite reconstruction remains a highly effective, time-tested masterclass technique in orthopedic oncology.
