Non-Ossifying Fibroma vs. Metastasis: A Diagnostic Dilemma in Oncology Patients
Key Takeaway
Differentiating Non-Ossifying Fibroma (NOF) from metastatic lesions in oncology patients is crucial. NOF typically presents with a well-defined, eccentric lytic lesion, often multiloculated with a sclerotic rim on radiographs and CT. MRI shows peripheral enhancement and less surrounding marrow edema, aiding distinction from aggressive metastasis.
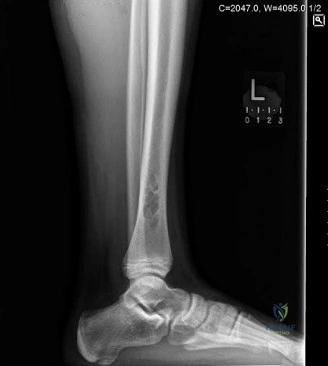
The clinical presentation detailed above concludes with the necessity to rule out impending pathological fracture. The MRI demonstrated a T1 hypointense, T2 hyperintense lobulated lesion with a distinct hypointense rim. Minimal perilesional edema was noted, and there was no soft tissue mass or cortical breakthrough. Contrast enhancement was peripheral and septal, lacking the solid, aggressive, and diffuse enhancement pattern typically associated with metastatic adenocarcinoma. However, given the patient's advanced age and oncologic history, a definitive tissue diagnosis was mandatory to differentiate between an atypical presentation of a benign entity, such as an adult Non-Ossifying Fibroma (NOF) or Benign Fibrous Histiocytoma, and a solitary lytic metastasis.

Introduction and Epidemiology
The diagnostic evaluation of a solitary lytic bone lesion in an older adult with a known history of malignancy represents a significant clinical challenge in orthopedic oncology. Statistically, a destructive bone lesion in a patient over the age of 40 with a history of carcinoma must be considered metastatic until proven otherwise. Prostate carcinoma is one of the most common primary malignancies to metastasize to bone, alongside breast, lung, renal, and thyroid carcinomas. While prostate metastases are classically osteoblastic, resulting in sclerotic lesions, mixed lytic-blastic or purely osteolytic variants can occur, particularly in advanced or heavily treated disease.
Conversely, Non-Ossifying Fibromas are benign, self-limiting developmental defects of bone primarily seen in the pediatric and adolescent populations. They are estimated to occur in up to 30-40% of all children, typically arising in the metaphyses of long bones, most frequently the distal femur and proximal tibia. The natural history of an NOF involves gradual migration away from the physis into the diaphysis, followed by progressive ossification and resolution by skeletal maturity. The persistence of an NOF into the seventh decade of life is exceedingly rare. When such histologically identical lesions are symptomatic in adults, they are often reclassified as Benign Fibrous Histiocytomas.
The dilemma in this specific patient population lies in the overlapping radiographic features of a healing, sclerotic-rimmed benign lesion and a slow-growing, perhaps radiation-altered, metastatic deposit. An erroneous assumption of benignity can lead to catastrophic pathological fracture and systemic tumor dissemination, whereas an overly aggressive surgical resection for a benign fibroma introduces unnecessary morbidity. Therefore, a rigorous, algorithmic approach focusing on biopsy, biomechanical evaluation, and appropriate surgical stabilization is paramount.
Surgical Anatomy and Biomechanics
Understanding the surgical anatomy and biomechanics of the distal femur is critical for both the diagnostic biopsy and the potential prophylactic stabilization of the limb.
Osteology and Vascularity
The distal femur transitions from a cylindrical diaphyseal shaft to a flared, metaphyseal region, terminating in the medial and lateral condyles. The cortical bone of the diaphysis is thick and provides substantial resistance to bending and torsional forces. As the femur flares distally, the cortex thins significantly, and the structural integrity relies heavily on the dense cancellous bone network. The vascular supply to this region is robust, derived primarily from the perforating branches of the profunda femoris artery, the descending genicular artery, and the superior genicular branches of the popliteal artery. The primary internervous plane utilized for approaching the lateral distal femur is between the vastus lateralis (femoral nerve) and the lateral intermuscular septum, which separates the anterior compartment from the posterior compartment (sciatic nerve).
Biomechanical Considerations
The distal femur is subjected to immense biomechanical forces during normal ambulation, including axial compression, bending, and torsion. The mechanical axis of the lower extremity passes through the center of the femoral head, the center of the knee, and the center of the ankle, placing the lateral cortex of the distal femur under tension and the medial cortex under compression.
The presence of a lytic lesion creates a significant stress riser. According to the principles of solid mechanics, the torsional rigidity of a tubular bone is proportional to the fourth power of its radius. A cortical defect reduces the load-bearing capacity disproportionately. An eccentric lytic lesion, such as the one described in the patient presentation, compromises the tensile strength of the lateral cortex. Mirels' criteria are widely utilized to quantify the risk of pathological fracture. The scoring system evaluates four variables: site of the lesion (upper limb, lower limb, peritrochanteric), nature of the lesion (blastic, mixed, lytic), size of the lesion (<1/3, 1/3-2/3, >2/3 of cortical diameter), and pain (mild, moderate, functional). A Mirels' score of 8 or higher generally indicates a high risk of impending fracture, warranting prophylactic internal fixation.
Indications and Contraindications
The management of a solitary lytic lesion in an oncology patient hinges on obtaining an accurate histological diagnosis and preventing pathological fracture. The decision to proceed with operative intervention must balance the oncologic prognosis, the mechanical stability of the bone, and the patient's overall physiological status.
Operative vs Non Operative Management
| Management Strategy | Primary Indications | Key Considerations |
|---|---|---|
| Core Needle Biopsy | All solitary lesions in oncology patients without definitive prior histology; atypical radiographic features. | Must be performed in line with the planned definitive surgical incision. |
| Non-Operative (Observation) | Confirmed benign asymptomatic NOF; Mirels' score < 7; poor surgical candidate; extremely short life expectancy (< 1 month). | Requires strict non-weight-bearing if pain is present; serial radiographic monitoring. |
| Intralesional Curettage and Grafting | Confirmed benign symptomatic lesion (e.g., Benign Fibrous Histiocytoma) compromising bone strength. | May require prophylactic hardware if the resulting cortical window is large. |
| Curettage Cementation and Fixation | Confirmed metastatic lesion; Mirels' score > 8; intractable pain; life expectancy > 6 weeks. | Polymethylmethacrylate (PMMA) provides immediate structural support and local thermal necrosis. |
| En Bloc Resection and Reconstruction | Solitary metastasis with curative intent (e.g., renal cell carcinoma); radioresistant tumors; massive bone destruction precluding fixation. | High morbidity; utilizes modular megaprostheses or massive allografts. |
Contraindications to Prophylactic Fixation
Absolute contraindications to surgical stabilization include patients who are medically unfit for anesthesia, those with an active systemic or local infection (unless the fixation is part of an infection control strategy), and patients with an extremely limited life expectancy (days to weeks) where the surgical intervention would not meaningfully improve the quality of remaining life. Relative contraindications include highly vascular tumors (e.g., metastatic renal cell or thyroid carcinoma) that have not undergone preoperative embolization, as intraoperative hemorrhage can be life-threatening.
Pre Operative Planning and Patient Positioning
Thorough preoperative planning is the cornerstone of successful orthopedic oncology surgery. The planning phase dictates the biopsy trajectory, the extent of resection, and the choice of reconstructive hardware.
Imaging and Staging Review
Standard orthogonal radiographs of the entire femur must be reviewed to assess for synchronous lesions and to determine the appropriate length of the implant. Advanced imaging is mandatory. A computed tomography (CT) scan provides superior detail regarding cortical integrity, the exact dimensions of the defect, and the presence of occult microfractures. Magnetic Resonance Imaging (MRI) is essential for delineating the intramedullary extent of the tumor, identifying any soft tissue extension, and visualizing the proximity of the lesion to the neurovascular bundle. Systemic staging, including a CT of the chest, abdomen, and pelvis, alongside a whole-body bone scintigraphy or PET-CT, is required to rule out disseminated disease, which would shift the treatment paradigm strictly toward palliation.
Implant Selection
For a distal femoral metadiaphyseal lesion, the surgeon must choose between an intramedullary device and an extramedullary plate. Retrograde intramedullary nails offer excellent biomechanical load-sharing properties and protect the entire length of the femur from subsequent metastatic deposits. However, they violate the knee joint and can potentially seed the entire medullary canal with tumor cells. Extramedullary fixation utilizing a lateral locking plate avoids the intra-articular approach and limits medullary seeding but may create a stress riser at the proximal end of the plate. The choice is dictated by the lesion's exact location, the presence of other femoral lesions, and the histological diagnosis.
Patient Positioning and Setup
The patient is positioned supine on a radiolucent operating table. A bump is placed beneath the ipsilateral hemipelvis to prevent the natural external rotation of the lower extremity, ensuring the patella faces directly anteriorly. This orientation is critical for accurate assessment of alignment and hardware trajectory. The entire lower extremity is prepped and draped freely to allow for full intraoperative range of motion. A sterile tourniquet may be applied to the proximal thigh but is often not inflated during oncologic procedures to allow for the identification and ligation of feeding vessels, and to prevent the potential systemic embolization of tumor cells upon tourniquet deflation. The fluoroscopy unit (C-arm) is positioned on the contralateral side of the table, ensuring unobstructed orthogonal views of the femur from the hip to the knee.
Detailed Surgical Approach and Technique
The surgical execution for a suspected metastatic lesion of the distal femur involves a sequential approach: biopsy, frozen section analysis, tumor extirpation, and biomechanical stabilization. The following details the technique for an open biopsy followed by intralesional curettage, cementation, and lateral locked plating.
Biopsy and Surgical Exposure
The skin incision is planned along the lateral aspect of the distal femur, incorporating the biopsy tract. A longitudinal incision is made through the skin and subcutaneous tissues. The iliotibial band is incised longitudinally in line with its fibers. The vastus lateralis is identified and elevated anteriorly off the lateral intermuscular septum. Perforating vessels from the profunda femoris are meticulously identified, clamped, and ligated to prevent postoperative hematoma.
If a preoperative core needle biopsy was not definitive, an open incisional biopsy is performed at this stage. A small cortical window is created over the lesion using a high-speed burr or osteotome. Tissue samples are sent immediately to pathology for frozen section analysis. If the frozen section confirms a benign entity such as an NOF, the surgeon may proceed with curettage and bone grafting. If it confirms metastasis, the procedure pivots to aggressive intralesional curettage and cementation.
Intralesional Curettage and Adjuvant Therapy
Once the diagnosis of metastasis is established and intralesional treatment is deemed appropriate, the cortical window is enlarged to allow complete access to the tumor cavity. The window must be oval or elliptical to minimize stress risers at its corners.
Aggressive intralesional curettage is performed using sequentially sized curettes. The goal is to remove all macroscopic tumor tissue until healthy, bleeding cortical bone is encountered. A high-speed burr is then utilized to extend the curettage by an additional 1-2 millimeters into the surrounding reactive bone, breaking down the sclerotic rim and ensuring thorough tumor removal.
Following gross resection, local adjuvant therapies are employed to extend the oncologic margin microscopically and reduce the risk of local recurrence. Pulsatile lavage with sterile saline is used to clear debris. Hydrogen peroxide or phenol may be applied to the cavity walls to induce chemical necrosis of residual tumor cells, followed by thorough irrigation. Alternatively, argon beam coagulation can be used to achieve thermal necrosis and excellent hemostasis of the cavity walls.
Reconstruction and Prophylactic Fixation
The resultant cavitary defect severely compromises the structural integrity of the distal femur. Polymethylmethacrylate (PMMA) bone cement is utilized to fill the defect. PMMA provides immediate structural support, resists compressive forces, and the exothermic reaction during polymerization provides an additional layer of thermal necrosis to any microscopic residual disease. The cement is pressurized into the cavity in a doughy state, ensuring interdigitation with the surrounding cancellous bone.
Before the cement fully hardens, the definitive fixation construct is applied. A pre-contoured distal femoral locking plate is slid submuscularly along the lateral cortex. The plate must be of sufficient length to span the lesion, ideally bypassing the defect by at least three cortical diameters proximally. The plate is provisionally fixed with K-wires. Fluoroscopy is utilized to confirm anatomical alignment in both the coronal and sagittal planes.
Locking screws are then inserted. Distally, multiple locking screws are placed into the femoral condyles to achieve a robust distal anchor. Proximally, bicortical locking or non-locking screws are utilized depending on bone quality. It is crucial to ensure that screws passing through the metadiaphyseal region traverse the PMMA cement mantle, creating a mechanically sound composite construct of bone, cement, and metal. The surgical site is profusely irrigated, a subfascial closed suction drain is placed, and the iliotibial band, subcutaneous tissues, and skin are closed in a layered fashion.
Complications and Management
Surgical intervention for impending or actual pathological fractures in oncology patients carries a higher complication rate than standard orthopedic trauma procedures due to the systemic burden of the disease, prior radiation therapy, and compromised local tissue healing.
Common Complications and Salvage Strategies
| Complication | Estimated Incidence | Etiology and Risk Factors | Management and Salvage Strategy |
|---|---|---|---|
| Hardware Failure / Construct Breakage | 5% - 10% | Disease progression causing further osteolysis; nonunion; inadequate initial fixation length. | Revision surgery utilizing a longer spanning implant, dual plating, or conversion to a modular distal femoral replacement (megaprosthesis). |
| Deep Surgical Site Infection | 3% - 8% | Immunosuppression from chemotherapy; prior local radiation; prolonged operative time; malnutrition. | Aggressive surgical debridement; retention of stable hardware; targeted intravenous antibiotic therapy; soft tissue coverage with local or free flaps if necessary. |
| Local Tumor Recurrence | 10% - 20% | Incomplete initial curettage; aggressive tumor biology; failure of local adjuvant therapy. | Postoperative external beam radiation therapy (EBRT); repeat curettage and cementation; en bloc resection if limb salvage is threatened. |
| Venous Thromboembolism (VTE) | 5% - 15% | Hypercoagulable state of malignancy; prolonged immobility; endothelial damage during surgery. | Preoperative optimization; mechanical prophylaxis; chemical prophylaxis (LMWH or DOACs) initiated postoperatively and continued for 4-6 weeks. |
| Periprosthetic Fracture | 2% - 5% | Stress riser at the proximal end of the plate; new metastatic deposit adjacent to hardware. | Revision internal fixation bypassing the new fracture site; intramedullary nailing if anatomically feasible. |
Post Operative Rehabilitation Protocols
The primary goal of prophylactic stabilization in the oncologic setting is the immediate restoration of function and pain relief. Unlike procedures relying on biological bone healing (such as bone grafting for benign lesions), the use of PMMA cement and rigid locked plating allows for immediate structural stability.
Patients are typically permitted immediate weight-bearing as tolerated on the operative extremity. Early mobilization is critical to prevent the sequelae of prolonged bed rest, including deep vein thrombosis, pulmonary embolism, and pulmonary atelectasis. Physical therapy is initiated on postoperative day one, focusing on active and active-assisted range of motion of the hip, knee, and ankle.
If the final histopathology confirms a metastatic lesion, coordination with the radiation oncology team is essential. Postoperative external beam radiation therapy (EBRT) is often indicated to achieve local tumor control and prevent disease progression that could compromise the hardware construct. Radiation therapy is typically delayed for 2 to 3 weeks postoperatively to allow for adequate incisional wound healing, minimizing the risk of wound dehiscence and deep infection.
Summary of Key Literature and Guidelines
The management of metastatic bone disease and the differentiation from benign entities is guided by a robust body of orthopedic oncology literature.
The foundational framework for prophylactic fixation remains the Mirels' criteria, originally published in Clinical Orthopaedics and Related Research in 1989. Mirels demonstrated that lesions with a score of 8 or higher have a fracture risk exceeding 15%, justifying the morbidity of prophylactic surgery. Subsequent validation studies have confirmed the high sensitivity of this scoring system, although some modern literature suggests it may lack specificity, occasionally leading to overtreatment.
For the definitive management of bone metastases, the National Comprehensive Cancer Network (NCCN) guidelines provide evidence-based algorithms. The guidelines strongly advocate for a multidisciplinary approach, emphasizing that surgical stabilization should be followed by local radiation therapy for optimal local control.
Regarding the diagnostic dilemma presented in this case, literature on adult presentations of Non-Ossifying Fibromas and Benign Fibrous Histiocytomas emphasizes the necessity of biopsy. Studies published in the Journal of Bone and Joint Surgery (JBJS) and Skeletal Radiology highlight that while radiographic features such as a sclerotic rim and lack of cortical destruction strongly suggest a benign process, the incidence of atypical lytic metastases in older adults mandates histological confirmation before definitive treatment is initiated. The integration of advanced imaging, meticulous surgical technique, and adherence to oncologic principles ensures optimal patient outcomes in these complex scenarios.
