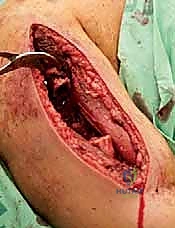
Humeral Metastasis Management: An Intraoperative Masterclass in Resection & Reconstruction
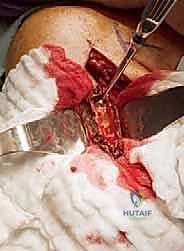
Key Takeaway
This masterclass guides fellows through the intricate surgical management of humeral metastases. We cover comprehensive anatomy, meticulous preoperative planning, and granular, real-time intraoperative execution for Type I, II, and III lesions. Emphasizing precision, neurovascular protection, and advanced reconstructive techniques, this session also delves into critical pearls, pitfalls, and postoperative care to optimize patient function and outcomes.
Comprehensive Introduction and Patho-Epidemiology
Welcome, esteemed colleagues and fellows, to the operating theater and to this definitive exploration of oncologic orthopaedics. Today, we are undertaking a critical, technically demanding, and profoundly impactful aspect of our specialty: the surgical management of metastatic bone disease in the humerus. This endeavor transcends the mere mechanical extirpation of a neoplastic lesion; it is a complex orchestration of restoring vital biomechanical function, alleviating intractable, life-altering pain, and significantly elevating our patients' quality of life during their most vulnerable clinical chapters. The humerus represents the second most common long bone affected by metastatic dissemination—surpassed only by the femur—and its involvement, particularly in the dominant upper extremity, demands our utmost surgical precision, anatomical mastery, and oncologic understanding.
To master the surgical management of these lesions, we must first understand the pathophysiology driving the bone destruction. Metastatic cells do not typically destroy bone directly; rather, they hijack the host's physiological bone remodeling machinery. Through the secretion of factors such as Parathyroid Hormone-Related Protein (PTHrP) and interleukins, tumor cells stimulate osteoblasts to upregulate Receptor Activator of Nuclear Factor Kappa-Β Ligand (RANKL). This, in turn, drives massive osteoclastogenesis and subsequent osteolysis. This "vicious cycle" of bone metastasis results in profound structural weakening of the cortical and cancellous architecture, predisposing the humerus to catastrophic mechanical failure under normal physiological loads. Understanding this osteoclastic hyperactivity is fundamental, as it dictates the necessity for rigid structural reinforcement rather than relying on biological bone healing, which is invariably compromised in these patients.
Unlike primary bone sarcomas, which often present with massive, invasive soft tissue extensions requiring radical en bloc resections, metastatic carcinomas typically present with a relatively constrained soft tissue component, even amidst extensive, permeative bone destruction. This critical oncologic characteristic frequently permits us to perform intralesional curettage or marginal bone-only resections. By meticulously sparing extracortical structures—such as the glenohumeral joint capsule, overlying muscular envelopes, and their tendinous attachments—we preserve the essential biological engines required for functional reconstruction. We must remember a fundamental tenet of upper extremity reconstruction: a few centimeters of humeral shortening, while biomechanically undesirable, has a far less significant impact on overall patient independence compared to a similar limb-length discrepancy in the lower extremities, where it invariably leads to a debilitating, energy-consuming limp.
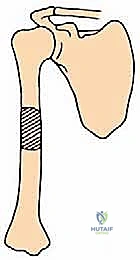
To systematize our surgical approach, we universally classify humeral metastases into three distinct types based on their precise anatomical location. Each zone presents unique biomechanical challenges and dictates highly specific surgical reconstructive algorithms:
* Type I: Proximal humerus lesions, extending from the articular surface across the anatomic neck and surgical neck, often compromising the rotator cuff insertions and the proximal metadiaphysis.
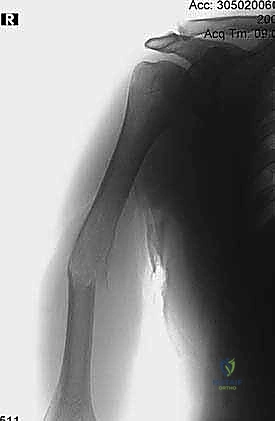
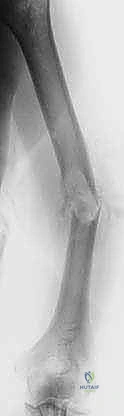
* Type II: Humeral diaphysis lesions, located strictly between the surgical neck proximally and the supracondylar ridges distally, characterized by high torsional stress profiles.
* Type III: Distal humerus lesions, extending from the supracondylar ridges down to the complex articular condyles of the elbow joint.
Detailed Surgical Anatomy and Biomechanics
Fellows, a superficial anatomical review is insufficient for the oncologic orthopaedic surgeon. A profound, three-dimensional understanding of humeral anatomy and its local biomechanical environment is absolutely non-negotiable. We must navigate these structures with absolute certainty to achieve tumor clearance while preventing catastrophic iatrogenic morbidity.
Proximal Humerus and Shoulder Girdle
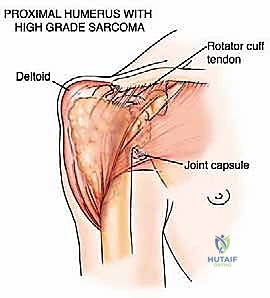
The proximal humerus is an anatomically dense and biomechanically complex region, serving as the fulcrum for the greatest range of motion of any joint in the human body. The osteology features the humeral head, anatomic neck, surgical neck, the greater and lesser tuberosities, and the bicipital groove. The muscular envelope is dominated anteriorly, laterally, and posteriorly by the thick, multipennate deltoid muscle. Deep to the deltoid, the rotator cuff muscles—supraspinatus, infraspinatus, teres minor, and subscapularis—coalesce to form a dynamic capsular stabilization mechanism. The long head of the biceps brachii tendon intra-articularly traverses the bicipital groove, serving as a critical landmark during anterior approaches.

The neurovascular relationships in the proximal humerus are unforgiving. The axillary nerve and the posterior circumflex humeral artery are particularly vulnerable. These structures exit the axilla through the quadrangular space and wrap transversely around the surgical neck of the humerus, directly deep to the deltoid muscle. They provide essential motor innervation to the deltoid and teres minor, as well as sensory innervation to the lateral shoulder. Iatrogenic injury to the axillary nerve results in catastrophic deltoid paralysis, rendering the shoulder functionally severely impaired. Furthermore, the brachial plexus lies superiorly and medially, with its terminal branches descending into the anterior and posterior compartments of the arm.
When executing a deltoid-splitting approach, you must adhere strictly to the "5-centimeter rule." The axillary nerve traverses the deep surface of the deltoid approximately 5 to 7 centimeters distal to the lateral edge of the acromion. Splitting the deltoid muscle fibers distally beyond this critical threshold virtually guarantees transection of the nerve's anterior branches. Biomechanically, the proximal humerus is subjected to massive compressive and shear forces, particularly during abduction and forward elevation, making secure fixation of tuberosity fragments to any endoprosthetic reconstruction the most challenging and critical step for restoring active elevation.
Humeral Diaphysis
The humeral shaft is a relatively cylindrical cortical tube that acts as a lever arm for hand positioning, but it is surrounded by distinct muscular compartments and highly critical neurovascular bundles. The upper half of the diaphysis serves as the insertion site for major axillary and chest wall muscles: the teres major, latissimus dorsi, and pectoralis major medially and anteriorly, and the deltoid laterally at the deltoid tuberosity. The lower half provides the broad origin for the brachialis muscle anteriorly and the medial and lateral heads of the triceps posteriorly. The lateral intermuscular septum separates the anterior and posterior compartments and serves as a critical surgical landmark.
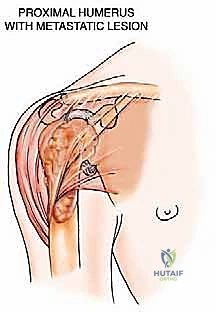
The radial nerve is the absolute sovereign structure of the mid-arm, and its preservation dictates our entire diaphyseal surgical strategy. It spirals posteriorly from medial to lateral, exiting the triangular interval to lie directly against the periosteum of the bone within the spiral groove (radial groove), deep to the lateral head of the triceps. At the junction of the middle and distal thirds of the humerus, it pierces the lateral intermuscular septum to enter the anterior compartment, running distally between the brachialis and brachioradialis muscles. It is highly susceptible to traction neuropraxia, compression, or outright transection during diaphyseal tumor curettage, osteotomy, or the passage of intramedullary reamers and nails.
Biomechanically, the humeral diaphysis is unique. Unlike the femur or tibia, which are primarily subjected to massive axial loading, the humerus is subjected to immense torsional (twisting) and bending forces. Simple activities of daily living, such as pushing oneself up from a chair, opening a heavy door, or wringing out a towel, generate massive torque across the shaft. Therefore, any reconstruction or prophylactic fixation of a Type II lesion must provide absolute rotational stability. A simple intramedullary nail without adequate proximal and distal interlocking screws will fail catastrophically under these torsional loads.
Distal Humerus and Elbow Articulation
The distal humerus transitions from a cylindrical diaphysis to a flattened, triangular metaphysis, terminating in the complex articular surfaces of the trochlea and capitellum. The bone stock here is remarkably thin, particularly at the olecranon and coronoid fossae, leaving very little room for standard internal fixation hardware when compromised by lytic metastases. The medial and lateral epicondyles serve as the robust origins for the common flexor and extensor tendon masses, respectively.
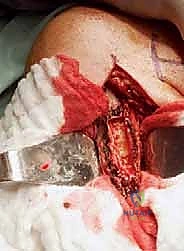
The neurovascular anatomy around the distal humerus is intricate and tightly packed. The primary neurovascular bundle (brachial artery and median nerve) courses along the medial aspect of the distal arm, nestled between the biceps and brachialis muscles, before crossing the anterior elbow capsule. The ulnar nerve diverges posteriorly, piercing the medial intermuscular septum to run directly posterior to the medial epicondyle within the cubital tunnel. It must be routinely identified, mobilized, and protected—often requiring anterior transposition—during any posterior approach to the distal humerus.
The radial nerve, having pierced the lateral intermuscular septum proximally, lies along the anterolateral aspect of the distal humerus, positioned between the brachialis and brachioradialis muscles, anterior to the lateral epicondyle. It subsequently divides into its superficial sensory and deep motor (posterior interosseous nerve) branches. Resection of Type III lesions often requires meticulous dissection of all three major nerves (radial, median, ulnar) to ensure safe tumor extirpation and the placement of a distal humeral replacement or total elbow arthroplasty.
Exhaustive Indications and Contraindications
The decision-making process in oncologic orthopaedics requires a delicate, highly individualized calculus. We must constantly balance the patient's overall oncologic prognosis and life expectancy against the anticipated surgical morbidity, rehabilitation time, and the immediate need for mechanical stabilization. Our primary goal is not always curative; it is to maximize the patient's functional independence and minimize suffering for the duration of their remaining life.
A completed pathological fracture is an absolute and obvious indication for surgical intervention, demanding immediate stabilization to provide pain relief and restore limb continuity. However, the true art of our specialty lies in prophylactic intervention for impending pathological fractures. We rely heavily on the Mirels' scoring system, which evaluates four variables: site of the lesion, nature of the lesion (lytic, blastic, mixed), size of the lesion relative to bone diameter, and the presence of mechanical pain. A Mirels' score of 9 or greater strongly dictates prophylactic fixation. Generally, any lytic lesion destroying more than 50% of the diaphyseal cortex, or a lesion greater than 2.5 cm in a high-stress area, warrants surgical reinforcement to prevent the catastrophic pain and complex reconstruction associated with a completed fracture.
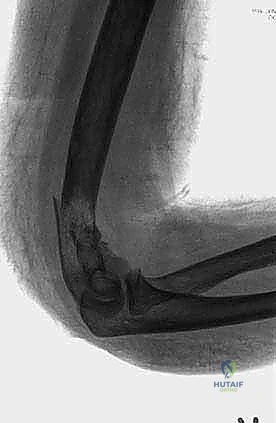
Beyond mechanical instability, intractable pain is a primary surgical indication. When localized, progressive metastatic disease causes severe, mechanical-type pain that is unresponsive to maximal systemic narcotic therapy, bisphosphonates, and preoperative external beam radiation therapy, surgical debulking and stabilization can provide profound, immediate relief. Furthermore, in highly selected patients presenting with a solitary bone metastasis—particularly from favorable primary histologies like renal cell carcinoma or breast cancer—and a long anticipated life expectancy, wide surgical resection with negative margins can offer definitive long-term local control and occasionally contribute to a systemic cure.
Despite these indications, we must rigorously respect the contraindications to surgical intervention. Operating on a patient who will not survive the perioperative period or who will never complete the necessary rehabilitation is a disservice.
| Category | Specific Indications | Specific Contraindications |
|---|---|---|
| Mechanical | Completed pathological fracture with displacement. Impending fracture (Mirels' score $\ge$ 9). Cortical destruction > 50% of circumference. |
Fracture in a completely paralyzed/plegic limb (non-functional). Asymptomatic blastic lesions with low fracture risk. |
| Oncologic | Solitary metastasis with curative intent (e.g., RCC, Thyroid). Failure of local radiation therapy. Radio-resistant tumor histologies. |
Widespread, uncontrolled visceral crisis (liver/lung failure). Life expectancy strictly < 6 to 12 weeks. Rapidly progressing polymetastatic disease. |
| Clinical | Intractable, mechanical pain unresponsive to narcotics. Need for immediate restoration of upper limb ADLs. |
Severe medical comorbidities (ASA IV/V) precluding anesthesia. Active, uncontrolled systemic or local infection. Patient bedbound with no functional goals. |
Pre-Operative Planning, Templating, and Patient Positioning
Thorough, obsessive preoperative planning is the hallmark of a master surgeon. In oncologic orthopaedics, the preoperative plan is our blueprint; it minimizes intraoperative surprises, dictates our margin status, and ensures we have the correct reconstructive armamentarium available. Attempting to improvise during a tumor resection is a recipe for catastrophic failure.
Advanced Imaging and Staging Modalities
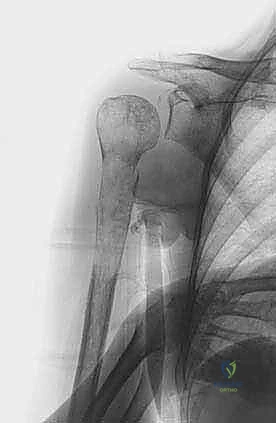
The imaging workup must be exhaustive. We begin with high-quality, orthogonal plain radiographs (AP and lateral) of the entire humerus, including the shoulder and elbow joints. This is critical to rule out synchronous "skip" metastases within the same bone that would necessitate altering our surgical plan from a short plate to a full-length intramedullary nail. Following plain films, a high-resolution, thin-slice Computed Tomography (CT) scan of the humerus is mandatory. The CT scan precisely defines the remaining cortical bone stock, the exact geometry of the osteolytic destruction, and helps us definitively choose between intralesional curettage with cementation versus a formal segmental resection.
Magnetic Resonance Imaging (MRI) with and without intravenous contrast of the entire humerus is indispensable for assessing the soft tissue and intramedullary extent of the tumor. The MRI meticulously delineates the tumor margins, identifying any extraosseous soft tissue mass and, crucially, its relationship to the major neurovascular bundles (especially the radial and axillary nerves). Furthermore, systemic staging using Total Body Bone Scintigraphy (Technetium-99m bone scan) or, preferably, a whole-body PET-CT is essential. This detects synchronous metastases elsewhere in the skeleton or visceral organs, directly influencing the patient's overall prognosis and, consequently, our surgical aggressiveness. If the primary tumor is unknown, a preoperative core needle biopsy is performed to establish the histology, which may dictate preoperative embolization (e.g., for highly vascular renal or thyroid metastases) or specific systemic therapies.
Surgical Templating and Construct Selection
Once imaging is complete, we transition to meticulous preoperative templating. Utilizing digital templating software, we calibrate the radiographs and overlay various implant templates. For prophylactic stabilization or curettage, we determine the precise diameter and length of intramedullary nails, ensuring we bypass the lesion by at least two cortical diameters proximally and distally. If utilizing plate osteosynthesis, we template the length of the reconstruction plate and the volume of polymethylmethacrylate (PMMA) bone cement required to fill the defect.
For formal resections of Type I or Type III lesions, templating is even more critical. We must calculate the exact length of the bone segment to be resected to achieve clean oncologic margins. We then template the corresponding modular endoprosthetic reconstruction system, selecting the appropriate stem diameters, segment lengths, and joint articulation sizes (e.g., hemiarthroplasty versus reverse total shoulder for the proximal humerus). Blood conservation strategies must be planned; cross-matching packed red blood cells is mandatory, and a cell saver is often kept on standby, though its use is controversial and strictly regulated in the presence of gross tumor spillage.
Patient Positioning and Operating Room Setup
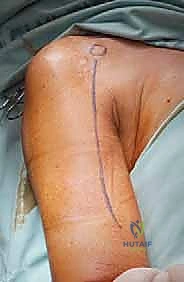
Proper patient positioning and operating room setup are the foundations of a smooth procedure. For Type I (Proximal) and Type II (Diaphyseal) lesions, the patient is typically placed in a semilateral decubitus or "beach chair" position. A radiolucent beanbag is utilized to stabilize the torso. The affected arm must be prepped and draped completely free from the fingertips to the base of the neck, allowing for unhindered, 360-degree manipulation of the limb during the procedure. We utilize an arm board for initial resting positioning, but the arm must remain mobile to allow for rotational adjustments during intramedullary nailing or prosthetic reduction.
For Type III (Distal) lesions requiring a posterior approach, the patient is typically placed in the lateral decubitus position with the operative arm draped over a well-padded L-bar or Mayo stand, allowing the elbow to flex freely to 120 degrees. Alternatively, a supine position with the arm draped across the chest can be utilized. Regardless of position, the fluoroscopy C-arm must be integrated into the setup seamlessly. The C-arm is positioned to allow for rapid, orthogonal AP and lateral views of the entire humerus without requiring the surgeon to break scrub or the patient to be dangerously repositioned.
Step-by-Step Surgical Approach and Fixation Technique
Alright, fellows, let us scrub in. The patient is positioned, the surgical site is marked, and the time-out is complete. We will now execute the surgical plan with meticulous attention to detail, oncologic principles, and anatomical preservation.
Proximal Humerus Resection and Reconstruction (Type I)
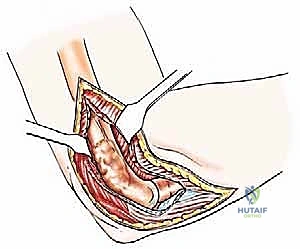
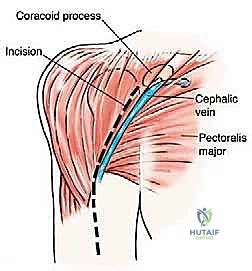
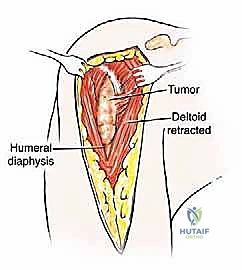
For Type I lesions requiring resection, we utilize an extended deltopectoral approach or an anterior utilitarian shoulder girdle incision. The incision begins at the coracoid process, follows the deltopectoral groove, and extends distally along the lateral border of the biceps. We identify the cephalic vein and typically retract it laterally with the deltoid. For purely metastatic lesions where the deltoid is uncompromised, we may opt for a direct longitudinal deltoid split to access the proximal humerus, strictly respecting the 5-centimeter rule to protect the axillary nerve.
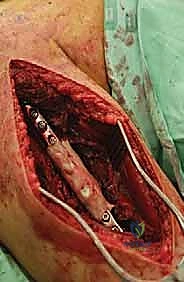
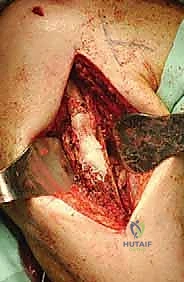
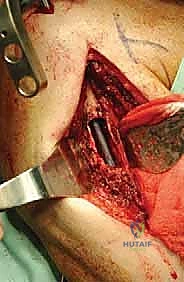
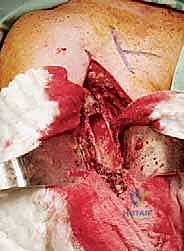
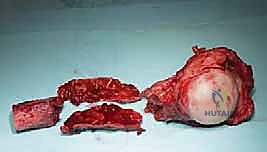
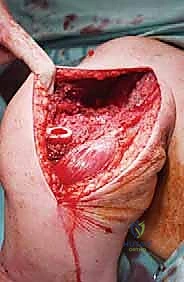
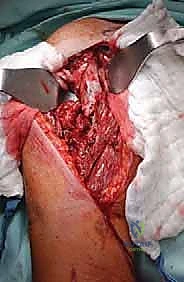
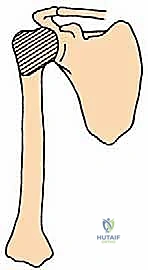
Once the proximal humerus is exposed, we identify and tag the rotator cuff tendons (subscapularis anteriorly; supraspinatus, infraspinatus, teres minor superiorly and posteriorly). If the tuberosities are free of tumor, we perform an osteotomy to preserve them; if involved, the tendons are sharply detached at their insertions. The long head of the biceps is tenodesed to the pectoralis major. The surgical neck or proximal diaphysis is then exposed, and the radial nerve is protected posteriorly. Using an oscillating saw, the humerus is transected at the pre-templated margin, at least 2-3 cm distal to the MRI-defined tumor extent. The proximal humerus is then mobilized and removed en bloc.
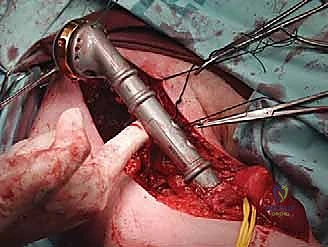
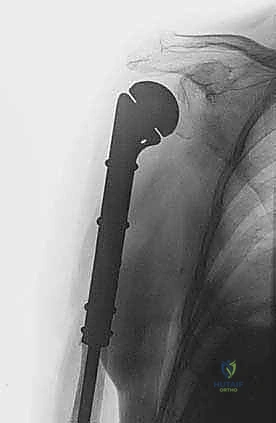
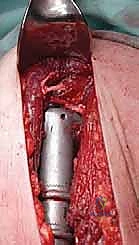
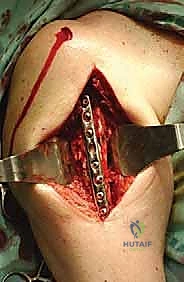
Reconstruction is typically achieved with a modular proximal humeral endoprosthesis. The distal humeral canal is sequentially reamed, and the prosthetic stem is cemented into place using antibiotic-loaded PMMA. We assemble the modular body to match the exact length of the resected specimen to restore myofascial tension. If the rotator cuff and tuberosities were preserved, they are meticulously repaired to the prosthesis using heavy non-absorbable sutures through specialized fenestrations. If the cuff is deficient, we may opt for a reverse total shoulder arthroplasty configuration to restore active elevation utilizing the preserved deltoid muscle.
Diaphyseal Management and Intramedullary Stabilization (Type II)
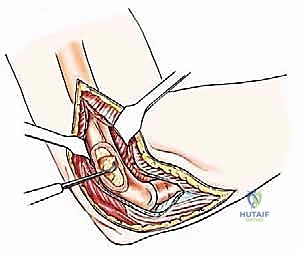
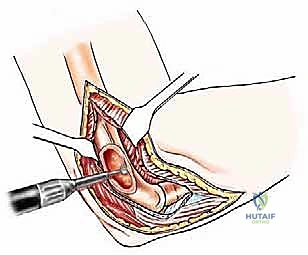
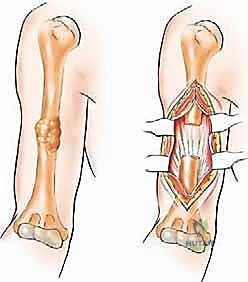
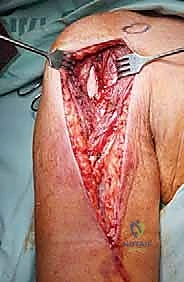
For Type II diaphyseal lesions, our approach depends on whether we are performing a closed intramedullary nailing or an open curettage and cementation. For open procedures, we utilize an anterolateral approach. The incision follows the lateral border of the biceps muscle. We identify the interval between the biceps and the brachialis. The brachialis muscle is then split longitudinally down to the periosteum. It is imperative to remember that the lateral half of the brachialis is innervated by the radial nerve; therefore, the split should be slightly medial to the midline of the muscle belly.
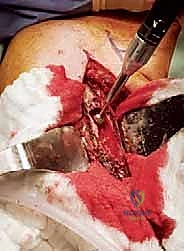
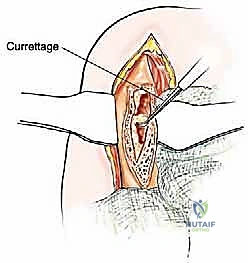
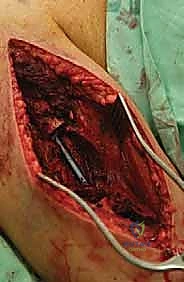
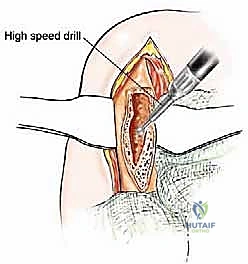
The most critical step in this approach is the definitive identification and protection of the radial nerve. We find the nerve as it pierces the lateral intermuscular septum and trace it proximally into the spiral groove. Once the nerve is safely retracted with vessel loops, we perform a large cortical window over the metastatic lesion. The tumor is aggressively removed using large curettes and a high-speed burr. We then utilize argon beam coagulation or chemical adjuvants (like phenol or hydrogen peroxide) to achieve local tumor necrosis and rigorous hemostasis within the cavity.
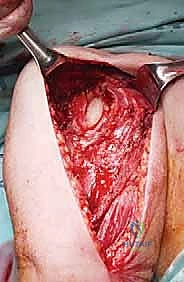
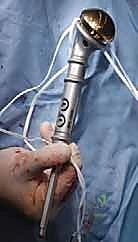
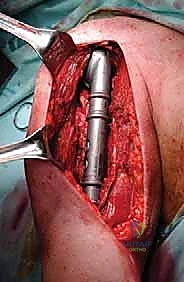
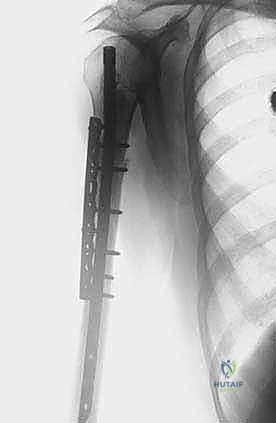
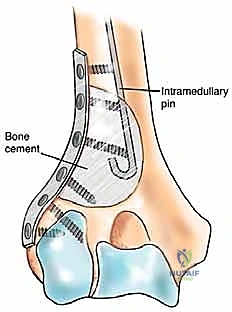
Following tumor extirpation, the massive cavitary defect must be reconstructed. We typically utilize a locked intramedullary nail to provide full-length load sharing and torsional stability. The nail is passed antegrade or retrograde. Once the nail is in place, the remaining cavitary defect is packed with liquid PMMA bone cement. The cement interdigitates with the remaining cancellous bone, provides immediate structural compressive strength, and the exothermic reaction of the curing cement provides an additional margin of thermal tumor necrosis. Finally, interlocking screws are placed proximally and distally to secure the construct.
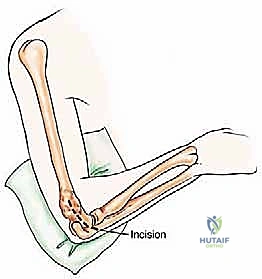
Distal Humerus Resection and Arthroplasty (Type III)
Type III lesions are approached via a universal posterior incision. The patient is lateral, and a midline longitudinal incision is made over the posterior arm, curving slightly radially around the olecranon tip. The ulnar nerve is immediately identified in the cubital tunnel, meticulously neurolysed, and transposed anteriorly to completely remove it from the zone of resection and subsequent reconstruction
Clinical & Radiographic Imaging Archive