Radial Head Replacement: Solutions for Complex Elbow Injuries

Key Takeaway
This article provides essential research regarding Radial Head Replacement: Solutions for Complex Elbow Injuries. Radial head replacement is a surgical procedure indicated for complex radial head fractures not amenable to internal fixation, especially when accompanied by valgus instability due to medial collateral ligament insufficiency. It is also used for radial head fractures with concurrent distal radioulnar joint injury (Essex-Lopresti injury) or for instability following previous radial head resection.
Comprehensive Introduction and Patho-Epidemiology
The Evolution of Radial Head Arthroplasty
The management of complex radial head fractures has undergone a paradigm shift over the last several decades, evolving from a historical preference for radial head excision to the contemporary standard of radial head arthroplasty. Historically, isolated radial head excision was considered a benign procedure with acceptable functional outcomes for isolated injuries. However, as our understanding of elbow biomechanics and the devastating consequences of longitudinal forearm instability expanded, it became unequivocally clear that the radial head is not a vestigial structure but a critical stabilizer. The recognition of complex instability patterns, such as the "terrible triad" of the elbow and Essex-Lopresti lesions, catalyzed the development and refinement of radial head implants. Modern orthopedic surgery now dictates that in the setting of complex elbow trauma where the radial head is not amenable to stable internal fixation, replacement is mandatory to restore radiocapitellar contact and prevent proximal radial migration.
The evolution of implant design has mirrored our growing biomechanical comprehension. Early silastic implants, while temporarily maintaining joint space, were fraught with catastrophic failure, particulate synovitis, and inability to withstand the compressive loads across the radiocapitellar joint. This led to the advent of metallic implants, which have since diversified into monoblock, modular, bipolar, and anatomic designs. Each iteration has sought to perfectly replicate the complex, non-circular geometry of the native radial head while providing modularity to address varying degrees of bone loss and ligamentous incompetence. Today, radial head replacement is a sophisticated, highly technical procedure that demands a profound understanding of both the implant's design rationale and the patient's specific pathoanatomy.



Pathophysiology of Complex Elbow Trauma
Complex elbow injuries involving the radial head rarely occur in isolation; they are typically the result of high-energy trauma or low-energy falls in osteoporotic individuals, characterized by a cascade of osteoligamentous failures. The classic mechanism involves an axial load applied to a partially flexed, supinated forearm, resulting in a valgus and posterolateral rotatory force vector. This force sequentially disrupts the lateral collateral ligament (LCL) complex, fractures the radial head, and, if the energy is sufficient, shears the coronoid process, culminating in the dreaded terrible triad injury. In this pathophysiological state, the elbow is rendered acutely unstable, and the native bony constraints must be restored to allow the ligamentous structures to heal at their isometric origins.
Furthermore, the pathophysiology of an Essex-Lopresti injury highlights the radial head's role in the longitudinal axis of the forearm. When a severe axial load ruptures the interosseous membrane (IOM) and the distal radioulnar joint (DRUJ) ligaments, the radial head becomes the sole restraint against proximal migration of the radius. If the radial head is fractured and subsequently excised without replacement in this setting, the radius will migrate proximally, leading to ulnocarpal impaction, chronic wrist pain, and profound loss of grip strength. Therefore, the pathophysiology of these injuries dictates that the radial head must be viewed not merely as an articular surface, but as a keystone in both the coronal and longitudinal stability of the upper extremity.


Epidemiological Considerations in Radial Head Fractures
Radial head fractures are the most common fractures of the adult elbow, accounting for approximately 30% to 33% of all elbow fractures and up to 4% of all fractures in the human body. The epidemiological distribution is bimodal, with a peak in young males sustaining high-energy trauma (such as motor vehicle collisions or falls from height) and a second peak in older, predominantly female patients sustaining low-energy falls from a standing height. While Mason Type I (nondisplaced) and Type II (partially displaced, reconstructable) fractures make up the vast majority of these injuries, complex Mason Type III (comminuted, entire head involved) and Type IV (associated with elbow dislocation) fractures present the primary cohort requiring arthroplasty.
The incidence of complex fracture-dislocations requiring radial head replacement has seen a relative increase, partly due to an aging population maintaining active lifestyles and partly due to heightened surgical recognition of instability patterns. Epidemiological studies indicate that failure to recognize associated ligamentous injuries—present in up to 80% of displaced radial head fractures—leads to a significantly higher rate of late complications, including chronic instability and post-traumatic arthrosis. Consequently, the threshold for performing a radial head replacement has lowered in cases of severe comminution, particularly when associated with any clinical or radiographic signs of medial collateral ligament (MCL) or interosseous membrane disruption.


Detailed Surgical Anatomy and Biomechanics
Osteology and Articular Relationships
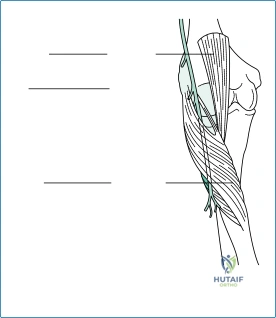
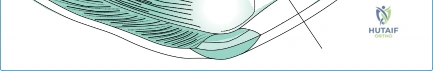
The radial head is a complex, asymmetric structure that articulates with both the capitellum of the distal humerus and the lesser sigmoid notch of the proximal ulna. Contrary to simplified anatomical models, the radial head is not perfectly cylindrical; it is elliptical, with its major and minor axes varying significantly among individuals. The articular dish (fovea) is concave and matches the convexity of the capitellum, forming the radiocapitellar joint, which is responsible for load transfer across the lateral column of the elbow. The peripheral articular margin, which articulates with the ulna, spans approximately 240 degrees, leaving a non-articular "safe zone" of about 120 degrees laterally where hardware can be safely placed during fixation, or where the implant must be carefully oriented to avoid impingement.
Understanding the normal range of motion (ROM) is critical for evaluating the success of arthroplasty. The normal anatomical ROM of the elbow is 0° of extension to 150° of flexion, with 80° of pronation and 80° of supination. However, the functional arc of motion required for most activities of daily living (ADLs) is significantly less: 30° to 130° of flexion, and 50° of both pronation and supination. In catastrophic cases where salvage arthrodesis is required, there is no single fixed position of arthrodesis that is universally ideal. Many authors recommend 90° of flexion; however, 110° may be best for ADLs (such as feeding oneself), whereas 60° may better suit specific heavy labor or work activities. The goal of radial head replacement is to restore the native osteology precisely enough to achieve the functional arc without impinging on these complex articular relationships.



Ligamentous Anatomy and the Stabilizing Role of the Radial Head
The stability of the elbow relies on a delicate interplay between static bony congruency and dynamic ligamentous tension. The primary static stabilizers are the ulnohumeral articulation, the medial collateral ligament (MCL) complex (specifically the anterior bundle), and the lateral collateral ligament (LCL) complex (specifically the lateral ulnar collateral ligament, LUCL). The radial head acts as a crucial secondary stabilizer. In the coronal plane, the radial head provides approximately 30% of the resistance to valgus stress in extension; however, if the MCL is compromised, the radial head becomes the primary restraint, transmitting up to 70% of the valgus load. This biomechanical reality dictates that in the presence of an MCL tear, the radial head must be preserved or replaced to prevent catastrophic valgus instability and rapid capitellar wear.
Furthermore, the radial head is intimately associated with the annular ligament, which encircles the head and stabilizes the proximal radioulnar joint (PRUJ) during pronation and supination. During surgical exposure for arthroplasty, the annular ligament must often be incised and subsequently repaired or reconstructed to prevent postoperative subluxation of the implant. The lateral ligamentous complex originates from the lateral epicondyle and its isometric fibers must drape appropriately over the replaced radial head. If an implant is "overstuffed" (too thick), it will abnormally tension the LCL complex, leading to stiffness, lateral compartment pain, and accelerated wear of the capitellar cartilage.



Kinematics and Normal Range of Motion
The kinematics of the elbow joint are governed by a complex, multi-axial hinge mechanism. As the elbow flexes and extends, the center of rotation remains relatively constant within the center of the capitellum and the trochlea. However, during forearm rotation, the radius must spin within the annular ligament and the lesser sigmoid notch while simultaneously gliding along the capitellum. This creates a highly dynamic contact area. Biomechanical studies have demonstrated that radiocapitellar contact pressures are highest in extension and pronation. When a metallic radial head implant is introduced, the modulus of elasticity is significantly higher than that of native bone, which can alter these contact pressures and lead to capitellar erosion if the implant is malpositioned.
Restoring the exact length of the radius is paramount to restoring normal kinematics. A discrepancy of as little as 2 to 3 millimeters can significantly alter joint tracking. If the implant is too short, the radiocapitellar joint will not engage, placing excessive stress on the IOM and ulnohumeral joint, potentially leading to longitudinal instability. Conversely, an overstuffed implant will severely restrict the functional arc of motion (30° to 130° flexion, 50° pronation/supination) and cause a fixed flexion contracture. Therefore, precise intraoperative assessment of kinematics, including fluoroscopic evaluation of the lateral ulnohumeral joint space (the "drop sign"), is a mandatory step in confirming appropriate implant sizing.


Exhaustive Indications and Contraindications
Primary Indications for Arthroplasty
The primary indication for radial head replacement is a severely comminuted fracture that is not amenable to stable internal fixation, typically defined as having more than three articular fragments or severe metaphyseal bone loss. In the setting of an isolated Mason Type III fracture, the decision between open reduction internal fixation (ORIF) and replacement can be nuanced; however, evidence strongly favors replacement when stable fixation cannot permit early, active range of motion. Prolonged immobilization to protect tenuous fixation is antithetical to modern elbow rehabilitation and inevitably results in profound stiffness. Thus, if the surgeon cannot confidently achieve rigid fixation, arthroplasty is the definitive solution.
Beyond isolated injuries, the indications for arthroplasty expand significantly in the context of complex instability. Radial head replacement is absolutely indicated in "terrible triad" injuries (elbow dislocation, radial head fracture, coronoid fracture) where the radial head cannot be perfectly reconstructed. Similarly, in Monteggia variant fracture-dislocations and trans-olecranon fracture-dislocations with associated radial head comminution, the radial head must be replaced to restore the anterior buttress against ulnohumeral subluxation. Furthermore, the presence of an Essex-Lopresti injury (concomitant distal radioulnar joint disruption) makes radial head replacement non-negotiable, as excision would guarantee proximal radial migration, ulnar plus variance, and debilitating wrist arthrosis.


Absolute and Relative Contraindications
While radial head arthroplasty is a powerful tool, it is not without contraindications. Absolute contraindications include active local or systemic infection, which precludes the implantation of any foreign hardware. Additionally, a neurologically flail upper extremity or severe, unmanageable psychiatric conditions that prevent adherence to strict postoperative rehabilitation protocols are considered absolute contraindications, as the success of the procedure relies heavily on patient compliance. Skeletal immaturity is also a strict contraindication; in pediatric patients with open physes, every effort must be made to preserve the native radial head to prevent severe growth disturbances and angular deformities of the forearm.
Relative contraindications require careful surgical judgment. Severe, pre-existing arthrosis or destruction of the capitellum is a significant relative contraindication. Placing a hard metallic implant against a denuded, eburnated capitellum will inevitably lead to persistent pain and accelerated wear. In such scenarios, particularly in older, low-demand patients, alternative salvage procedures such as isolated radial head excision (if ligaments are stable) or total elbow arthroplasty (TEA) may be more appropriate. Furthermore, a highly osteoporotic proximal radius that cannot support the stem of the implant may necessitate the use of cemented stems or alternative reconstructive strategies, complicating the standard arthroplasty approach.



Summary of Indications and Contraindications
| Category | Specific Clinical Scenarios | Rationale for Management |
|---|---|---|
| Absolute Indications | Essex-Lopresti injuries with comminuted radial head | Prevents proximal radial migration and DRUJ impaction. |
| Absolute Indications | Terrible Triad injuries with >3 fragments | Restores valgus and posterolateral rotatory stability. |
| Absolute Indications | Unreconstructable fracture with MCL tear | Radial head is the primary valgus stabilizer when MCL is out. |
| Relative Indications | Isolated Mason III fractures (highly comminuted) | Allows for early active ROM compared to tenuous ORIF. |
| Absolute Contraindications | Active joint infection or osteomyelitis | High risk of deep periprosthetic joint infection (PJI). |
| Absolute Contraindications | Skeletally immature patients (open physes) | Arrests longitudinal growth, causing severe deformity. |
| Relative Contraindications | Severe capitellar cartilage destruction | Metal-on-bone articulation causes severe pain and wear; consider TEA. |


Pre-Operative Planning, Templating, and Patient Positioning
Advanced Imaging Modalities
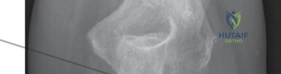
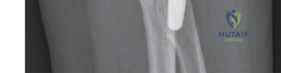
Meticulous preoperative planning begins with high-quality imaging. Standard anteroposterior (AP) and lateral radiographs of the elbow are mandatory, but they are often insufficient for fully characterizing complex fracture geometry. A computed tomography (CT) scan with 3D reconstructions is now considered the gold standard for any complex elbow trauma. The CT scan allows the surgeon to assess the exact number of radial head fragments, the presence of articular impaction, and critical associated injuries such as coronoid fractures or subtle marginal capitellar shearing fractures. Furthermore, CT imaging helps in identifying the "bare area" of the proximal radius, which is crucial if any fixation is to be attempted alongside arthroplasty.
In cases where an Essex-Lopresti injury is suspected, imaging must extend beyond the elbow. Dedicated bilateral posteroanterior (PA) and lateral radiographs of the wrists are required to assess for ulnar variance. A discrepancy in ulnar variance compared to the contralateral, uninjured side is highly suggestive of longitudinal radioulnar dissociation. Magnetic resonance imaging (MRI) is rarely used in the acute trauma setting for the radial head itself, but it can be an invaluable adjunct if the status of the MCL, LCL, or interosseous membrane remains clinically ambiguous and will dictate the extent of the surgical reconstruction.



Implant Selection and Templating
The selection of the appropriate implant is a critical phase of preoperative planning. Modern systems offer modularity, allowing independent selection of stem diameter, neck length, and head size. Templating should ideally be performed using radiographs of the contralateral, uninjured elbow to determine the native diameter and thickness of the radial head. The goal is to restore the exact distance from the capitellum to the lesser sigmoid notch. Surgeons must be familiar with the specific implant system available at their institution, noting whether the stems are designed to be fixed (press-fit or cemented) or loose-fit (smooth stems designed to act as a spacer and allow micro-motion).
Smooth, loose-fitting stems are currently favored by many high-volume elbow surgeons because they self-align with the capitellum during the arc of motion, potentially reducing asymmetric edge-loading and capitellar wear. Conversely, fixed stems offer rigid stability but require perfect anatomical alignment; any slight malversion can lead to catastrophic joint incongruency. Templating also involves anticipating the need for additional procedures, such as coronoid fixation or ligamentous repair. The surgeon must ensure that anchors, plates, and the radial head stem will not occupy the same physical space within the proximal ulna and radius.



Patient Positioning and Operating Room Setup
Proper patient positioning is paramount for achieving adequate exposure and facilitating intraoperative fluoroscopy. The procedure can be performed with the patient in the supine or lateral decubitus position. The supine position, with the arm draped across the chest or resting on a radiolucent hand table, is highly versatile. It allows excellent access to both the lateral and medial sides of the elbow, which is essential if an MCL repair or a separate medial approach for coronoid fixation is anticipated. A sterile tourniquet is applied high on the brachium to ensure a bloodless field, which is critical for identifying fine articular fragments and delicate neurovascular structures.
Alternatively, the lateral decubitus position with the arm suspended over a bolstered post offers unparalleled visualization of the posterior and lateral compartments. Gravity assists in retracting the triceps, and the elbow can be easily flexed and extended to assess stability. However, accessing the medial side in the lateral position can be ergonomically challenging. Regardless of the position chosen, the fluoroscopy unit (C-arm) must be brought in from the head or the opposite side of the table, and the surgeon must confirm that perfect AP and lateral views can be obtained without compromising the sterile field before the incision is made.



Step-by-Step Surgical Approach and Fixation Technique
Choosing the Optimal Surgical Approach
The deep surgical approaches to the elbow may be achieved through a single ‘utility’ posterior incision or through separate medial and lateral incisions. A universal posterior skin incision is highly recommended in complex trauma, as it mitigates the risk of creating narrow skin bridges if multiple windows are required, and it provides extensile access to the entire joint. From this posterior skin incision, full-thickness fasciocutaneous flaps are elevated. To access the radial head, two primary deep fascial intervals are utilized: the Kocher approach and the Kaplan approach.
The Kocher approach utilizes the internervous plane between the extensor carpi ulnaris (ECU, innervated by the posterior interosseous nerve) and the anconeus (innervated by the radial nerve). This approach provides excellent access to the radial head and the LUCL. However, it requires detachment of the LCL origin if extending proximally, which must be meticulously repaired. The Kaplan approach utilizes the interval between the extensor digitorum communis (EDC) and the extensor carpi radialis brevis (ECRB). This approach provides a more direct, anterior view of the radial head and coronoid but places the posterior interosseous nerve (PIN) at significantly higher risk, requiring careful pronation of the forearm to protect the nerve during deep retraction.



Deep Dissection and Radial Head Excision
Once the chosen interval is developed, the joint capsule is incised. In the setting of trauma, the capsule and LUCL are often already torn, and the surgeon can simply follow the rent in the tissue. The fractured fragments of the radial head are meticulously excised. It is imperative to piece the excised fragments together on the back table ("puzzle piecing") to ensure that no intra-articular loose bodies remain in the joint space. The native radial head also serves as a crucial template for sizing the definitive implant, making its careful reconstruction on the back table a critical step.
Following excision, the proximal radial neck is prepared. A clean, flat osteotomy is performed at the level of the intact neck. The surgeon must avoid resecting too much bone; the osteotomy should be just distal to the fracture lines but proximal to the bicipital tuberosity to preserve supination power. The medullary canal of the radius is then sequentially broached or reamed. Care must be taken not to perforate the radial cortex, particularly in osteoporotic bone. The forearm should be maintained in pronation during this step to move the PIN away from the surgical field and the retractor blades.



Implant Sizing, Trialing, and Implantation
Implant sizing is arguably the most unforgiving step of the procedure. The goal is to avoid "overstuffing" the joint. The trial stem is inserted, followed by the trial neck and head. The diameter of the prosthetic head should be slightly smaller than the native head to prevent impingement on the lesser sigmoid notch during rotation. The thickness (height) of the implant is evaluated by articulating it with the capitellum. A perfectly sized implant will allow smooth tracking without opening the lateral ulnohumeral joint space.
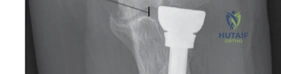
To assess for overstuffing, the surgeon performs the "drop sign" test under fluoroscopy. With the trial implant in place, a lateral radiograph is taken. If the ulnohumeral joint space is widened laterally, the implant is too thick and must be downsized. Additionally, the surgeon should be able to pass a small elevator between the radial head and the capitellum with minimal resistance. Once the perfect size is confirmed, the definitive implant is assembled and impacted into
Clinical & Radiographic Imaging Archive