Posterior Blade Plate Arthrodesis for Failed Total Ankle Arthroplasty: An Intraoperative Masterclass
Key Takeaway
Join us in the OR for an immersive masterclass on posterior blade plate arthrodesis for failed total ankle arthroplasty. We'll meticulously cover patient assessment, preoperative planning, detailed surgical anatomy, and a step-by-step intraoperative execution. Learn critical techniques for exposure, implant removal, bone grafting, and optimal hardware placement to achieve successful fusion and improve patient outcomes.
Comprehensive Introduction and Patho-Epidemiology
Welcome, colleagues, to the operating theater. Today, we are undertaking a highly demanding but increasingly indispensable surgical procedure: the salvage of a failed total ankle arthroplasty (TAA) utilizing a posterior blade plate arthrodesis. The landscape of foot and ankle reconstruction has evolved precipitously over the last two decades. With the advent of third-generation and fourth-generation TAA implants, surgical volume has expanded exponentially worldwide. Consequently, as orthopedic surgeons, we must anticipate and be technically proficient in managing the inevitable failures that arise from this increased utilization. Whether the etiology is aseptic osteolysis, catastrophic polyethylene wear, or complex septic complications, the end-stage presentation of a failed TAA demands a meticulous, biomechanically sound reconstructive strategy.
While the primary objective of a TAA is the restoration of pain-free motion and the preservation of adjacent joint kinematics, the reality of revision surgery often dictates a paradigm shift toward salvage via arthrodesis. This is primarily necessitated by the profound compromise of peri-articular bone stock, the disruption of the medial and lateral ligamentous complexes, and the distortion of native osseous anatomy that accompanies implant failure. The posterior approach, coupled with rigid blade plate fixation, offers unparalleled advantages in these complex revision scenarios. It allows for simultaneous access to pristine bone graft donor sites, avoids the often-compromised anterior soft tissue envelope, and capitalizes on the biomechanical superiority of tension-band plating.
The pathogenesis of TAA failure mirrors the well-documented mechanisms observed in hip and knee arthroplasty, yet it is uniquely influenced by the constrained anatomy and high contact stresses of the ankle joint. Aseptic failure remains the predominant etiology, driven largely by polyethylene wear and subsequent particulate disease. This macrophage-mediated inflammatory cascade leads to progressive, often "ballooning" osteolysis, which relentlessly destroys the limited metaphyseal bone stock of the distal tibia and the body of the talus. As osteolysis progresses, the structural integrity of the host-implant interface is compromised, culminating in component subsidence, angular malalignment, and ultimately, catastrophic mechanical failure.
Septic failure, though less common than aseptic loosening, presents a far more formidable challenge. Chronic periprosthetic joint infections (PJIs) in the ankle are notoriously difficult to eradicate due to the tenuous soft tissue envelope and the proximity of neurovascular structures. Biofilm formation on the implant surfaces necessitates complete hardware explantation. In these instances, salvage dictates a staged approach: aggressive radical debridement and placement of an antibiotic-eluting polymethylmethacrylate (PMMA) spacer, followed by definitive posterior blade plate arthrodesis only after normalization of serological markers and negative synovial aspirations. Understanding these distinct pathophysiological pathways is the critical first step in formulating a successful surgical blueprint.
Detailed Surgical Anatomy and Biomechanics
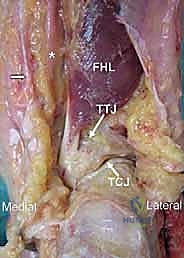
A profound understanding of the posterior ankle anatomy is the sine qua non of a successful posterior blade plate arthrodesis. The posterior approach exploits the internervous and intermuscular planes to access the posterior malleolus, the posterior facet of the subtalar joint, and the extra-articular posterior calcaneal tuberosity. The primary incision is typically placed longitudinally, either directly over the Achilles tendon for a trans-tendinous approach or, more commonly, just lateral to the Achilles tendon to exploit the interval between the Achilles and the peroneal tendons. When utilizing the posterolateral approach, the sural nerve and the lesser saphenous vein must be meticulously identified and protected in the subcutaneous tissues, as iatrogenic injury here leads to debilitating postoperative neuromas.
Deep to the crural fascia, the surgeon encounters the deep posterior compartment of the leg. The crucial anatomical landmark here is the flexor hallucis longus (FHL) muscle belly and tendon. The FHL serves as the primary protector of the posterior neurovascular bundle (the posterior tibial artery and the tibial nerve), which lies immediately medial to it. By mobilizing the FHL and retracting it medially, the surgeon safely isolates the neurovascular bundle from the operative field, gaining direct, unobstructed access to the posterior capsular structures of the tibiotalar and subtalar joints. This safe zone allows for aggressive debridement of the failed TAA components and the preparation of broad, flat osseous surfaces for arthrodesis.
Biomechanically, the posterior application of a rigid, fixed-angle blade plate is vastly superior to anterior or lateral plating in the context of salvage arthrodesis. The posterior aspect of the ankle and hindfoot represents the tension side of the biomechanical construct during the stance phase of gait. According to the principles established by the AO Foundation, applying a plate to the tension side converts the distracting forces of weight-bearing into compressive forces across the arthrodesis site. This dynamic compression is critical for achieving primary bone healing in the presence of massive structural allografts or autografts, which are universally required to fill the cavitary defects left by the explanted TAA.
Furthermore, the blade plate provides exceptional multi-planar stability. The blade itself, when driven into the dense trabecular bone of the calcaneal tuberosity (for a tibiotalocalcaneal fusion) or the remaining body of the talus (for a tibiotalar fusion), creates a robust, fixed-angle anchor that resists the powerful deforming forces of the Achilles tendon. The proximal portion of the plate is then secured to the posterior cortex of the tibia, which is typically thick and cortical, providing excellent screw purchase even in osteopenic bone. This rigid construct minimizes micromotion at the graft-host interfaces, thereby optimizing the biological environment for osteogenesis and significantly reducing the risk of nonunion compared to intramedullary nailing or isolated screw fixation.
Exhaustive Indications and Contraindications
The decision to proceed with a posterior blade plate arthrodesis for a failed TAA is complex and must be highly individualized. The primary indication is aseptic loosening with catastrophic bone loss that precludes the implantation of a revision TAA component. When ballooning osteolysis has destroyed the talar body or the distal tibial metaphysis, the remaining bone stock is often insufficient to support any articulating device. In these scenarios, arthrodesis is the only viable option to restore a stable, plantigrade, and painless limb.
Another critical indication is the salvage of an infected TAA. Following the successful eradication of a periprosthetic joint infection via a staged protocol with an antibiotic spacer, the posterior approach is ideal for definitive fusion. The posterior soft tissue envelope is typically pristine, having been spared from the anterior incisions used during the primary arthroplasty and the subsequent debridement surgeries. This healthy soft tissue bed is crucial for minimizing wound healing complications and providing adequate coverage for the bulky blade plate hardware. Furthermore, the posterior approach allows for simultaneous access to the posterior superior iliac spine (PSIS) for the harvesting of voluminous, high-quality autologous cancellous bone graft, which is essential for filling the massive defects left by the explanted components and the spacer.
Contraindications, while relatively few, must be strictly respected to avoid disastrous outcomes. Severe peripheral vascular disease is an absolute contraindication; adequate arterial inflow must be confirmed preoperatively via non-invasive vascular studies or angiography, as the extensive dissection required for this procedure demands a robust blood supply for wound healing and bone incorporation. Active, untreated infection is an absolute contraindication for a single-stage procedure; placing massive metallic hardware and bone graft into an infected bed guarantees failure. Finally, profound, uncorrectable immunosuppression or severe medical comorbidities that preclude a prolonged general or regional anesthetic may necessitate non-operative management, such as a custom-molded ankle-foot orthosis (AFO) or a patellar tendon-bearing brace.
| Factor | Indications for Posterior Blade Plate Arthrodesis | Contraindications (Absolute & Relative) |
|---|---|---|
| Aseptic Failure | Massive ballooning osteolysis precluding revision TAA; severe component subsidence; periprosthetic fracture with instability. | Lack of adequate tibial or calcaneal bone stock to achieve proximal/distal fixation (Relative - may require custom implants). |
| Septic Failure | Eradicated PJI (second stage of a two-stage protocol); chronic infection where amputation is refused. | Active, untreated periprosthetic joint infection (Absolute for single-stage); acute fulminant sepsis. |
| Soft Tissue | Compromised anterior soft tissue envelope (multiple prior anterior incisions, skin grafting, or flaps). | Severe peripheral vascular disease (Absolute); active posterior ulceration (Absolute). |
| Deformity | Severe coronal or sagittal plane malalignment secondary to TAA failure; combined ankle and subtalar pathology. | Uncorrectable equinus contracture that prevents plantigrade positioning (Relative - requires extensive release). |
| Patient Factors | High functional demand requiring absolute stability; failure of conservative bracing. | Severe medical comorbidities precluding major surgery; non-compliance with NWB protocols. |
Pre-Operative Planning, Templating, and Patient Positioning
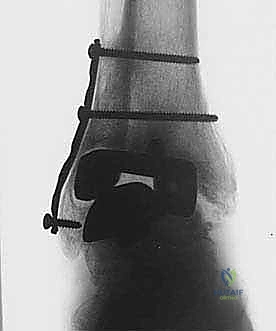
Meticulous preoperative planning is the bedrock upon which a successful posterior blade plate arthrodesis is built. This process begins with a rigorous evaluation of high-quality imaging. Weight-bearing, standing plain radiographs (anteroposterior, lateral, and mortise views) are essential for assessing the overall mechanical axis of the lower extremity, the degree of component subsidence, and the gross extent of osteolysis. However, plain films drastically underestimate the true volumetric bone loss behind metallic implants.

Therefore, a thin-slice computed tomography (CT) scan with metal artifact reduction sequence (MARS) is mandatory. The CT scan allows the surgeon to precisely quantify the cavitary defects in the distal tibia and the talus, evaluate the integrity of the medial and lateral malleoli, and assess the condition of the adjacent subtalar and transverse tarsal joints. If the subtalar joint exhibits significant arthritic changes or if the talar body is completely destroyed, the surgical plan must be upgraded from a tibiotalar (TT) arthrodesis to a tibiotalocalcaneal (TTC) arthrodesis. The CT scan also dictates the volume and type of bone graft required—whether massive structural allografts (e.g., femoral head) or copious autologous cancellous bone from the iliac crest will be necessary to restore limb length and achieve union.
Pre-contouring the blade plate is a critical, time-saving step that must be performed prior to the inflation of the tourniquet. Utilizing a sawbones model of the ankle and the preoperative CT templates, the surgeon selects the appropriate blade length and plate length. The blade is typically angled at approximately 90 to 95 degrees relative to the shaft of the plate to accommodate the native valgus and plantarflexion of the hindfoot. The plate itself must be contoured to match the posterior flare of the distal tibia and the posterior prominence of the calcaneus. This pre-contouring significantly reduces intraoperative frustration, minimizes tourniquet time, and ensures that the hardware will sit flush against the bone, optimizing compression and minimizing soft tissue irritation.
Patient positioning is exclusively prone for this procedure. The patient is carefully rolled onto a radiolucent Jackson table or a standard operating table with chest and pelvic rolls to ensure adequate pulmonary excursion. The ipsilateral lower extremity is prepped and draped in a sterile fashion from the mid-thigh to the toes. Crucially, the ipsilateral posterior superior iliac spine (PSIS) is also prepped and draped into the sterile field to allow for unhindered access for autologous bone graft harvesting. A pneumatic tourniquet is placed on the proximal thigh. The prone position not only provides unparalleled access to the posterior ankle and the PSIS but also allows gravity to assist in maintaining the foot in a plantigrade position during the final reduction and fixation steps. Intraoperative fluoroscopy must be positioned to allow for seamless anteroposterior, lateral, and axial views of the hindfoot without compromising the sterile field.
Step-by-Step Surgical Approach and Fixation Technique
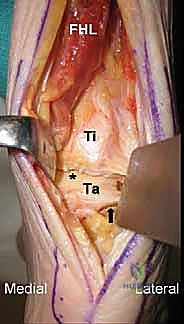
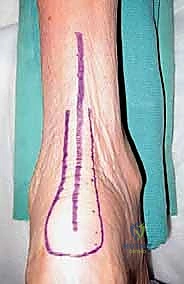
The surgical execution demands precision, patience, and a deep respect for the posterior soft tissue envelope. Following exsanguination and tourniquet inflation, a longitudinal incision is made approximately 15 centimeters in length, centered over the posterior ankle. The incision is typically placed slightly lateral to the Achilles tendon. The subcutaneous tissues are carefully dissected, and the sural nerve is identified, mobilized, and protected with a vessel loop. The crural fascia is incised in line with the skin incision. The interval between the Achilles tendon (medially) and the peroneal tendons (laterally) is developed. The deep posterior compartment is entered by incising the deep fascia covering the flexor hallucis longus (FHL) muscle.
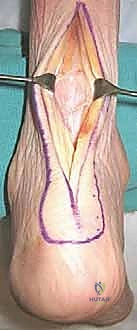
The FHL muscle belly and tendon are meticulously dissected and retracted medially. This is the most critical step in the exposure, as the FHL protects the posterior tibial artery and the tibial nerve. With the neurovascular bundle safely retracted medially, the posterior capsule of the ankle joint and the subtalar joint (if a TTC fusion is planned) are exposed. An aggressive posterior capsulotomy is performed. The failed TAA components are now visualized. Using a combination of osteotomes, high-speed burrs, and extraction devices, the tibial and talar components are carefully explanted. Extreme care must be taken to preserve as much host bone as possible during this extraction phase.
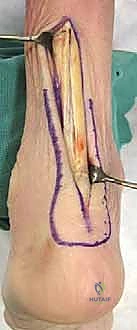
Once the hardware is removed, the true extent of the osteolysis is revealed. A radical debridement of all fibrous tissue, metallosis, and polyethylene debris is performed using curettes and a rongeur. The sclerotic bone surfaces of the distal tibia, the remaining talus, and the calcaneus are aggressively prepared using a high-speed burr and osteotomes to expose bleeding, healthy cancellous bone. If a structural defect exists, a massive femoral head allograft is contoured to fit the defect precisely, restoring limb length and providing structural support. Copious autologous cancellous bone graft, harvested simultaneously from the PSIS by a second surgical team, is packed tightly into all remaining cavitary defects and around the structural graft.
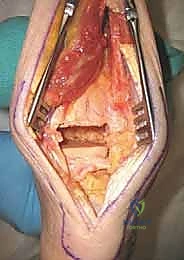
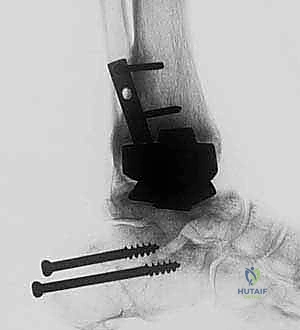
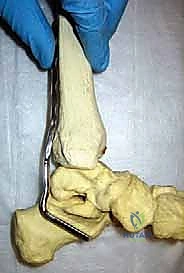
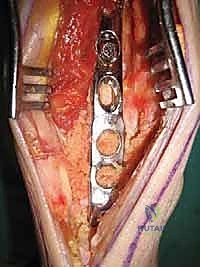
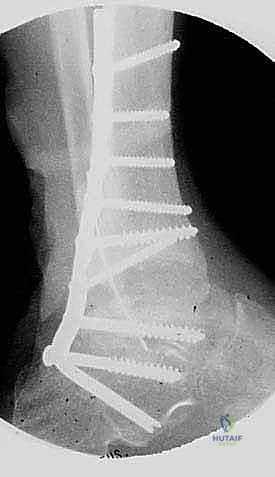
The fixation phase begins with the insertion of the pre-contoured blade plate. For a TTC arthrodesis, a seating chisel is used to create a channel in the posterior calcaneal tuberosity, aiming anteriorly and slightly medially toward the base of the second metatarsal. The blade is then impacted into the calcaneus. The foot is meticulously positioned in 0 degrees of dorsiflexion, 5 degrees of valgus, and 5 to 10 degrees of external rotation. The proximal portion of the plate is then clamped to the posterior tibia. An articulated tension device (ATD) is applied to the proximal end of the plate to generate massive compression across the arthrodesis sites.
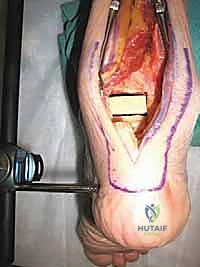
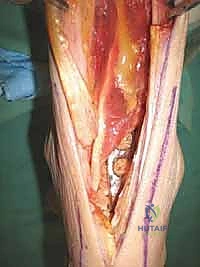
Once adequate compression is confirmed clinically and fluoroscopically, the plate is secured to the tibia using multiple bicortical locking and non-locking screws. Additional independent lag screws may be placed from the tibia into the talus or calcaneus outside the plate to augment the construct. The wound is copiously irrigated. The FHL is allowed to fall back into its native position, providing a vascularized muscle bed over the hardware. The crural fascia is closed loosely, and the subcutaneous tissues and skin are closed in layers over a closed-suction drain.
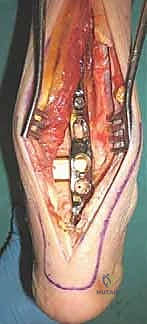
Complications, Incidence Rates, and Salvage Management
Despite meticulous surgical technique, the salvage of a failed TAA via posterior blade plate arthrodesis is fraught with potential complications. The most devastating, yet unfortunately common, complication is nonunion or delayed union. Given the massive bone loss, compromised vascularity, and the avascular nature of structural allografts, nonunion rates in the literature range from 10% to 25%. Nonunions are typically insidious, presenting with persistent, deep aching pain upon weight-bearing months after the initial surgery. Radiographic evaluation often reveals progressive radiolucent lines at the graft-host interface and hardware failure (e.g., broken screws or plate fracture). Management of a symptomatic nonunion requires a robust revision strategy: removal of hardware, aggressive re-debridement of the nonunion site, massive autogenous bone grafting (often utilizing a vascularized free fibula flap or bone morphogenetic proteins), and re-stabilization with an even more rigid construct, such as a custom-milled intramedullary nail or a circular external fixator.
Infection is another profound complication, particularly in cases where the index TAA failed secondary to sepsis. Even with a meticulously executed two-stage protocol, recurrent periprosthetic joint infection occurs in 5% to 15% of cases. The presence of massive metallic hardware and avascular bone graft creates an ideal environment for biofilm formation. Acute postoperative infections may be managed with aggressive surgical debridement, hardware retention (if the construct remains stable), and targeted intravenous antibiotic therapy. However, chronic, deep-seated infections invariably necessitate complete hardware explantation, removal of all bone graft, and placement of a new antibiotic spacer. In refractory cases, or in patients with severe host compromise, a below-knee amputation (BKA) becomes the definitive, life-saving salvage procedure.
Neurological complications, specifically iatrogenic injury to the sural nerve or the tibial nerve, represent a significant source of postoperative morbidity. Sural nerve injury, occurring in up to 10% of cases, typically results from vigorous retraction or direct transection during the posterolateral approach. This leads to painful neuromas and sensory deficits along the lateral border of the foot. Tibial nerve injury is less common but far more devastating, resulting in profound motor weakness and plantar numbness. Meticulous identification and protection of the neurovascular bundle behind the FHL is the only reliable method of prevention. Finally, hardware prominence can cause significant soft tissue irritation, particularly in thin patients. If the plate causes intractable pain or threatens skin integrity, hardware removal may be indicated, but only after solid, contiguous osseous fusion has been unequivocally confirmed on a CT scan, typically no earlier than 12 to 18 months postoperatively.
| Complication | Estimated Incidence | Pathogenesis / Risk Factors | Salvage Management Strategy |
|---|---|---|---|
| Nonunion / Delayed Union | 10% - 25% | Massive bone voids, avascular allograft, smoking, inadequate compression, micromotion. | Revision arthrodesis, biological augmentation (BMP, ICBG), rigid circular external fixation, vascularized fibula. |
| Deep Infection (Recurrent or De Novo) | 5% - 15% | Prior septic TAA, diabetes, prolonged operative time, massive hardware/allograft burden. | Radical I&D, hardware explantation, antibiotic spacer, prolonged IV antibiotics, possible BKA. |
| Sural Nerve Neurapraxia / Neuroma | 5% - 10% | Direct surgical trauma during posterolateral incision, aggressive retraction. | Gabapentinoids, targeted nerve blocks, surgical neuroma excision and burying into muscle belly. |
| Hardware Failure (Screw/Plate Breakage) | 5% - 8% | Fatigue failure secondary to nonunion, premature weight-bearing, inadequate construct rigidity. | Hardware removal, assessment of fusion mass, revision internal fixation if nonunion is present. |
| Wound Dehiscence / Necrosis | 5% - 10% | Poor vascular inflow, excessive soft tissue tension over bulky hardware, smoking. | Local wound care, negative pressure wound therapy (VAC), local or free tissue transfer (flap coverage). |
Phased Post-Operative Rehabilitation Protocols
The postoperative rehabilitation following a posterior blade plate arthrodesis for a failed TAA is a protracted, highly structured process that demands strict patient compliance. The primary objective in the early postoperative phase is the protection of the soft tissue envelope and the rigid stabilization of the arthrodesis site to facilitate initial osteogenesis. Immediately following surgery, the patient is placed in a bulky, well-padded, short-leg splint with the ankle strictly immobilized in a neutral, plantigrade position. The patient is made absolutely non-weight-bearing (NWB) on the operative extremity. Elevation of the limb above the level of the heart is critical during the first two weeks to mitigate the profound postoperative edema that inevitably accompanies this extensive surgical dissection.
At the two-week postoperative mark, the patient returns for the first clinical evaluation. The splint is removed, and the surgical incision is meticulously inspected for any signs of dehiscence or marginal necrosis. Sutures or staples are typically removed at this juncture, provided the wound is completely sealed. The limb is then transitioned into a rigid, custom-molded fiberglass cast or a locked controlled ankle motion (CAM) boot. The patient must maintain strict NWB status for a minimum of 6 to 8 weeks. During this critical period, the biological incorporation of the bone graft—particularly massive structural allografts—is in its most vulnerable nascent stage. Premature weight-bearing will inevitably lead to catastrophic hardware failure or graft collapse. Deep vein thrombosis (DVT) prophylaxis is maintained throughout this NWB period.
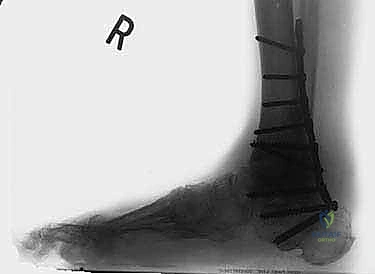
Radiographic and clinical evaluation dictates the progression of rehabilitation. At 8 to 10 weeks postoperatively, new plain radiographs are obtained to assess for early signs of trabecular bridging across the arthrodesis sites. If the clinical exam reveals no pain with manual stressing of the hindfoot and radiographs demonstrate early consolidation, the patient is permitted to initiate progressive, partial weight-bearing in the CAM boot. This typically begins at 25% of body weight, utilizing crutches or a walker, and advances by 25% every two weeks. Physical therapy is initiated at this stage, focusing not on the fused ankle, but on maximizing the range of motion and strength of the ipsilateral knee, hip, and the forefoot joints.
Full weight-bearing out of the CAM boot is rarely permitted before 12 to 16 weeks postoperatively, and often much later in cases involving massive structural grafts. A CT scan is highly recommended prior to authorizing unrestricted, unbraced ambulation, as plain radiographs notoriously overestimate the degree of solid osseous fusion behind the dense metallic shadow of the blade plate. Once solid fusion is confirmed, the patient is transitioned into regular footwear, often requiring a rocker-bottom sole modification or a custom orthotic to accommodate the loss of ankle and subtalar motion and to normalize the gait trajectory. Maximum medical improvement (MMI) is typically not achieved until 12 to 18 months following the salvage procedure.
Summary of Landmark Literature and Clinical Guidelines
The evolution of salvage strategies for failed TAA is heavily documented in the orthopedic literature, with the posterior blade plate arthrodesis emerging as a gold standard technique for complex, bone-deficient cases. Landmark studies by Myerson and colleagues have extensively detailed the biomechanical and clinical advantages of the posterior approach. Their cohort analyses consistently demonstrate that utilizing the posterior tension band principle yields superior union rates compared to anterior plating, particularly when massive structural allografts are required to reconstruct the talar body. The literature emphasizes that the posterior application of the blade plate effectively neutralizes the deforming forces of the Achilles tendon, converting them into beneficial compressive forces across the fusion mass.
Furthermore, the work of Hintermann and Coetzee has been pivotal in defining the indications for tibiotalocalcaneal (TTC) versus isolated tibiotalar (TT) arthrodesis in the salvage setting. Their clinical guidelines suggest that if the subtalar joint is arthritic, or if the remaining talar bone stock is less than 1.5 centimeters in height following thorough debridement, the arthrodesis must be extended to include the calcaneus to ensure adequate distal fixation. The literature universally cautions against attempting isolated TT fusions in the presence of severe talar avascular necrosis or ballooning osteolysis, as the risk of hardware cut-out and subsequent nonunion is unacceptably high.
Recent systematic reviews analyzing the outcomes of staged revisions for infected TAA further validate the posterior approach. These studies highlight that while the eradication of periprosthetic joint infection is achievable in the majority of cases using antibiotic spacers, the definitive reconstruction requires a robust, biologically friendly soft tissue envelope. The posterior approach, by avoiding the scarred and often compromised anterior skin, significantly reduces the incidence of postoperative wound breakdown—a complication that frequently leads to catastrophic failure and amputation. In conclusion, the current clinical guidelines strongly support the posterior blade plate arthrodesis as a highly reliable, biomechanically sound, and biologically advantageous technique for the definitive salvage of the failed total ankle arthroplasty.
