Patient Presentation & History
We present the case of a 68-year-old male, a retired mechanic with a history of hypertension well-controlled on medication, and Type 2 Diabetes Mellitus managed with oral agents. He presented with a chief complaint of a progressively enlarging, largely painless mass in his right posterior thigh over the preceding 6 months. Initially, he dismissed it as a fatty lump or a muscle strain, but its gradual yet relentless growth, reaching an estimated 8x6 cm by presentation, prompted him to seek medical attention.
The patient denied any specific mechanism of injury, trauma, or preceding inflammatory episode. He reported no significant constitutional symptoms such as fever, chills, night sweats, or unintentional weight loss. He did not describe any sensory deficits or motor weakness in the ipsilateral lower extremity initially, though he noted a recent onset of mild, dull ache in the region, particularly after prolonged standing or walking. His social history included a 40 pack-year smoking history, which he quit 5 years prior, and occasional alcohol consumption. There was no family history of sarcomas, but his mother had a history of breast cancer. His past surgical history was limited to an appendectomy in his youth. The absence of trauma, the deep-seated nature of the mass, and its progressive, relatively painless growth raised immediate suspicion for a neoplastic process rather than a benign etiology such as a hematoma, abscess, or lipoma, particularly given the patient's age and the size of the lesion. The slight discomfort upon presentation, rather than being an initial symptom, suggests local mass effect or early infiltration of adjacent structures.
Clinical Examination
Upon physical examination, the patient was generally well-appearing, with no signs of cachexia or systemic illness. Vitals were within normal limits.
Local Examination (Right Lower Extremity):
*
Inspection:
A large, ovoid mass was visible on the posterior aspect of the right thigh, approximately 15 cm proximal to the popliteal crease. The overlying skin appeared normal, without erythema, discoloration, ulceration, or prominent venous engorgement. No dimpling or tethering of the skin was noted. The mass appeared to cause a subtle contour deformity of the posterior thigh musculature.
*
Palpation:
The mass was firm, non-tender, and deeply seated within the musculature. Its approximate dimensions were 8 cm (craniocaudal) x 6 cm (mediolateral) x 5 cm (anteroposterior). It exhibited limited mobility in relation to the deep fascial layers and underlying bone, suggesting potential fixation or deep origin, but some slight mobility relative to the superficial skin. There was no warmth or pulsatility detected. No regional lymphadenopathy was palpable in the inguinal or popliteal regions.
*
Range of Motion (ROM):
Hip and knee ROM were full and painless on the affected side. However, passive and active flexion of the knee against resistance, particularly hamstring activation, elicited a mild, deep ache within the mass itself, indicative of its probable intramuscular or intermuscular location.
*
Neurological Assessment:
Sensation to light touch and pinprick was intact throughout the right lower extremity in all dermatomal distributions (L2-S2). Motor strength was 5/5 for all major muscle groups (hip flexion/extension, knee flexion/extension, ankle dorsiflexion/plantarflexion), though sustained hamstring contraction was mildly uncomfortable. Deep tendon reflexes (patellar and Achilles) were 2+ bilaterally and symmetrical. No pathological reflexes were elicited. This indicated no overt neural compression or infiltration at this stage.
*
Vascular Assessment:
Distal pulses (femoral, popliteal, dorsalis pedis, posterior tibial) were palpable, strong, and symmetrical bilaterally. Capillary refill was brisk (<2 seconds) in the toes. No bruits were audible over the mass or major vessels.
The clinical findings, particularly the large, deep-seated, firm, and relatively fixed nature of the mass in an elderly patient, strongly necessitated further urgent investigation to rule out a high-grade soft tissue sarcoma.
Imaging & Diagnostics
Initial Radiographs:
An anteroposterior and lateral radiograph of the right femur and knee were obtained. These revealed no overt signs of bone involvement, periosteal reaction, cortical erosion, or intralesional calcification. The soft tissue mass itself was radiographically dense but non-specific, consistent with a soft tissue lesion. The primary utility of initial plain films in this context is to rule out osseous tumors or significant cortical involvement by a soft tissue mass, which could complicate surgical planning.
Magnetic Resonance Imaging (MRI):
A dedicated MRI of the right thigh with and without intravenous gadolinium contrast was performed and was the definitive imaging modality for local staging.
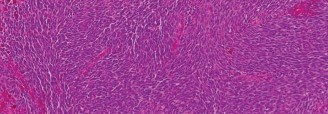
The MRI revealed a large, heterogeneous, infiltrative soft tissue mass measuring approximately 8.5 x 6.2 x 5.8 cm within the posterior compartment of the right thigh, predominantly involving the semimembranosus and semitendinosus muscles.
*
T1-weighted sequences:
The mass appeared largely isointense to muscle, with some areas of subtle hypointensity, suggestive of necrosis or fluid components. A poorly defined pseudocapsule was noted, but signs of extralesional extension were evident.
*
T2-weighted and STIR sequences:
The mass demonstrated marked heterogeneity and significant hyperintensity, indicating high water content, inflammation, or necrosis, typical of a high-grade malignancy. Central areas of lower signal intensity were suspicious for hemorrhage or necrosis.
*
Post-contrast T1-weighted sequences with fat saturation:
There was avid, heterogeneous enhancement throughout the mass, particularly at its periphery, further supporting a highly vascular and active neoplastic process.
*
Neurovascular Assessment:
The mass was in close proximity to the sciatic nerve and the popliteal neurovascular bundle, with a clear but narrow fat plane maintained in most areas, suggesting potential but not definite direct infiltration. The femoral artery and vein were unaffected. There was no evidence of bone marrow edema or direct cortical invasion of the femur. Intratumoral vascularity, characteristic of sarcomas, was noted.
Computed Tomography (CT) of Chest, Abdomen, and Pelvis:
As part of the systemic staging protocol, a CT scan of the chest, abdomen, and pelvis was performed to rule out distant metastases. This revealed no pulmonary nodules, hepatic lesions, or retroperitoneal lymphadenopathy. This indicated localized disease at presentation, or at least no overt metastatic disease identifiable by conventional imaging.
Positron Emission Tomography-Computed Tomography (PET-CT):
A PET-CT scan was performed to assess metabolic activity and identify any occult metastatic disease not picked up by conventional CT. The primary thigh lesion demonstrated intense FDG avidity (SUVmax 12.5), consistent with a high-grade metabolic tumor. No other sites of abnormal FDG uptake were identified, confirming the absence of detectable distant metastases and confirming localized disease, which is crucial for surgical planning and prognosis.
Biopsy:
Following imaging, an image-guided (ultrasound-guided) core needle biopsy was performed by the orthopedic oncology service. Three core samples were obtained from different parts of the mass, ensuring adequate tissue acquisition while minimizing contamination of surrounding tissues. The biopsy tract was carefully planned to be excised en bloc with the definitive resection specimen.
*
Histopathological Examination:
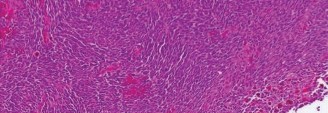
Microscopic examination revealed a highly pleomorphic spindle cell neoplasm with abundant nuclear atypia, prominent nucleoli, and numerous atypical mitotic figures (including atypical forms). There were large areas of necrosis and hemorrhage. No specific line of differentiation (e.g., adipocytic, smooth muscle, Schwannian, osteoid, chondroid) was identifiable by routine H&E staining.
*
Immunohistochemistry (IHC):
IHC staining was performed. The tumor cells were positive for Vimentin, a non-specific mesenchymal marker. Crucially, they were negative for S100 (ruling out MPNST and melanoma), Desmin and smooth muscle actin (ruling out leiomyosarcoma), CD34 (ruling out solitary fibrous tumor), cytokeratins (ruling out carcinoma or synovial sarcoma), and MDM2/CDK4 (ruling out well/dedifferentiated liposarcoma). This extensive panel of negative markers, coupled with the highly pleomorphic and anaplastic morphology, led to the diagnosis of Undifferentiated Pleomorphic Sarcoma (UPS).
Differential Diagnosis
The clinical and initial imaging findings in this patient necessitated a broad differential diagnosis for a large, deep-seated soft tissue mass in the thigh of an adult. However, the rapidly growing, firm, and infiltrative nature, coupled with the MRI characteristics and patient demographics, strongly pointed towards a malignant etiology. Here, we compare Undifferentiated Pleomorphic Sarcoma with other high-grade sarcomas commonly encountered in this anatomical location.
| Feature | Undifferentiated Pleomorphic Sarcoma (UPS) | Dedifferentiated Liposarcoma (DDLPS) | Malignant Peripheral Nerve Sheath Tumor (MPNST) |
|---|---|---|---|
| Prevalence | Most common adult soft tissue sarcoma of intermediate to high grade. | Second most common adult soft tissue sarcoma, often retroperitoneal. | Rare, ~5-10% of soft tissue sarcomas. 50% associated with NF1. |
| Age Group | Typically 50-70s, rare in children. | Typically 50-70s, less common in younger adults. | Any age, often 20-50s, higher incidence in NF1 patients. |
| Common Location | Deep soft tissues of extremities (thigh), retroperitoneum, trunk. | Deep soft tissues (retroperitoneum, thigh, mediastinum), often large. | Proximal extremities, trunk, head and neck, often intimately associated with a major nerve. |
| Growth Rate | Often rapid, leading to presentation with large mass. | Variable; well-differentiated component slow, dedifferentiated component often rapid. | Variable, often rapid, frequently painful early due to nerve involvement. |
| Clinical Features | Large, deep-seated, firm, usually painless until mass effect. | Large, often painless. History of smaller, long-standing fatty lesion possible. | Often painful, neurological deficits common if nerve involved. History of NF1 or prior radiation. |
| Imaging (MRI) | Heterogeneous, ill-defined margins, large, may have necrotic/hemorrhagic areas. Intense, heterogeneous contrast enhancement. No specific diagnostic features. | Heterogeneous mass with distinct fatty components (well-differentiated) and solid non-lipomatous areas (dedifferentiated). | Ill-defined, infiltrative, T2 hyperintense. May follow nerve sheath. Can show "target sign" (central fibrosis, peripheral myxoid) in lower-grade forms. |
| Histopathology | Highly pleomorphic spindle and polygonal cells, high mitotic activity, marked nuclear atypia, abundant necrosis. Storiform pattern can be seen. No specific line of differentiation after extensive workup. | Admixture of well-differentiated liposarcoma (mature adipocytes, atypical stromal cells) and non-lipogenic high-grade sarcoma (often resembles UPS). | Spindle cell tumor, often with fascicular growth, nuclear palisading ("serpentine" nuclei), high cellularity, necrosis. Perineural invasion common. |
| Immunohistochem. | Vimentin positive. Negative for most lineage-specific markers (e.g., S100, desmin, cytokeratins, CD34, actin, MDM2, CDK4). Diagnosis of exclusion. | Vimentin positive. Dedifferentiated component: MDM2 and CDK4 positive (gene amplification via FISH). S100 negative. | S100 positive (variable, 50-70%), SOX10 positive. Negative for desmin, actin, cytokeratins. |
| Genetics | Complex karyotypes, frequent chromosomal gains/losses. No specific diagnostic translocation. | MDM2 and CDK4 amplification on 12q14-15, typically detected by FISH. | NF1 gene mutations (in NF1-associated cases). |
| Prognosis | High-grade, high risk of local recurrence and distant metastasis (lungs). | High risk of recurrence (local and distant) and metastasis for dedifferentiated component. | Aggressive, high risk of recurrence and metastasis, especially with NF1. Poorer prognosis than UPS. |
Other important considerations in the differential, especially before biopsy, would include:
*
Benign Lesions:
Atypical lipomatous tumor/well-differentiated liposarcoma (low risk of dedifferentiation, often smaller, purely fatty on imaging), intramuscular myxoma (hypocellular, myxoid stroma), hematoma (history of trauma, evolves over time), abscess (inflammatory signs, fever).
*
Other Malignancies:
Leiomyosarcoma (positive for smooth muscle markers), synovial sarcoma (often biphasic, specific translocations), extraskeletal osteosarcoma (matrix production).
The systematic diagnostic approach, including meticulous imaging and a well-executed core needle biopsy with comprehensive immunohistochemistry, was crucial in narrowing the diagnosis to Undifferentiated Pleomorphic Sarcoma.
Surgical Decision Making & Classification
The definitive diagnosis of a high-grade Undifferentiated Pleomorphic Sarcoma (UPS) in the posterior thigh, along with the absence of detectable distant metastases, rendered this patient a candidate for curative-intent surgery. Surgical decision-making was guided by the principles of oncologic sarcoma management and a multidisciplinary tumor board discussion involving orthopedic oncology, medical oncology, radiation oncology, diagnostic radiology, and pathology.
Staging:
Based on the imaging and biopsy findings, the tumor was classified according to:
*
AJCC 8th Edition Staging (Soft Tissue Sarcoma):
* T2b: Tumor size > 5 cm.
* N0: No regional lymph node metastasis.
* M0: No distant metastasis.
* G3: High-grade sarcoma.
* This placed the patient's tumor as Stage IIIB (T2b, N0, M0, G3).
*
Enneking Staging System (Surgical Staging System for Musculoskeletal Sarcomas):
* Grade: G2 (high-grade).
* Site: T2 (extracompartmental, as it was deep-seated and likely infiltrated beyond a single muscle compartment).
* Metastases: M0 (no distant metastases).
* This classified the tumor as G2T2M0.
Operative vs. Non-operative Decision:
Soft tissue sarcomas, particularly high-grade lesions like UPS, are primarily managed with surgical resection as the cornerstone of treatment. Non-operative management is typically reserved for patients with widespread metastatic disease, prohibitive comorbidities, or those for whom surgery would result in unacceptable functional deficits (e.g., amputation of a functionally critical limb where limb salvage is not oncologically safe). For this patient, with localized, high-grade disease, radical surgical excision with negative margins was deemed essential for optimal oncologic outcomes.
Role of Neoadjuvant/Adjuvant Therapy:
Given the tumor's large size (>5cm) and high-grade nature, the multidisciplinary tumor board recommended a strategy incorporating neoadjuvant radiation therapy.
*
Neoadjuvant Radiation Therapy:
The patient received 50 Gy in 25 fractions over 5 weeks to the tumor bed, with an emphasis on preserving a strip of skin and subcutaneous tissue to facilitate wound healing post-surgery. The rationale for neoadjuvant radiation included:
1.
Tumor Downsizing:
Potentially reducing tumor bulk, thereby facilitating a more complete resection with wider margins.
2.
Sterilization of Microscopic Disease:
Addressing microscopic tumor extensions not visible on imaging, potentially reducing local recurrence rates.
3.
Improved Functional Outcomes:
Often allowing for a less extensive resection compared to primary surgery, potentially reducing the need for extensive reconstruction.
4.
Better Tolerance:
Radiation is generally better tolerated pre-operatively than post-operatively, especially concerning wound healing.
*
Adjuvant Chemotherapy:
The role of adjuvant chemotherapy for localized UPS remains controversial, with varying protocols. For high-grade, deep-seated tumors larger than 5 cm, consideration for adjuvant chemotherapy with doxorubicin-based regimens is often made, especially in cases with positive margins or high risk features. In this patient's case, a decision on adjuvant chemotherapy would be made after final surgical pathology and a re-evaluation by the medical oncology team.
Limb Salvage vs. Amputation:
The primary goal was limb salvage while achieving oncologically adequate margins. The MRI demonstrated a clear enough plane from the sciatic nerve and popliteal vessels to suggest that a wide en bloc resection might be feasible without sacrificing the neurovascular bundle or requiring an amputation. The proximity to vital structures, however, necessitated careful planning and possibly reconstruction. The patient was extensively counselled regarding both limb salvage with potential functional deficits and the option of amputation, understanding the oncologic imperatives. He opted for limb salvage.
Surgical Technique / Intervention
The surgical intervention for this Undifferentiated Pleomorphic Sarcoma required meticulous planning and execution to ensure wide oncologic margins while maximizing functional preservation.
Pre-operative Planning:
Detailed surgical templating was performed using the pre- and post-radiation MRI scans. The biopsy tract, having been planned by the orthopedic oncologist, was clearly marked to ensure its complete excision. The anticipated extent of muscle resection (semimembranosus, semitendinosus, and portions of biceps femoris and adductor magnus) was outlined, as was the potential need for soft tissue reconstruction. The proximity of the sciatic nerve and popliteal vessels mandated careful neurovascular dissection.
Patient Positioning and Anesthesia:
The patient was placed in the prone position to allow optimal access to the posterior thigh. Ipsilateral hip and knee joints were appropriately padded and positioned to allow for controlled intraoperative manipulation. General anesthesia was administered, along with regional anesthesia (sciatic nerve block) for enhanced post-operative pain control. Prophylactic antibiotics and deep vein thrombosis (DVT) prophylaxis were administered.
Surgical Approach and Excision:
A longitudinal skin incision was made directly over the tumor and incorporating the biopsy tract. The incision extended approximately 20 cm, providing ample exposure.
*
Skin and Subcutaneous Tissue:
Full thickness excision of the skin and subcutaneous tissue overlying the tumor and the entire biopsy tract was performed elliptically.
*
Fascial Incision:
The deep fascia was incised widely, proximal and distal to the tumor, establishing a disease-free plane.
*
Neurovascular Dissection:
Meticulous dissection was initiated both proximally and distally to the mass. The sciatic nerve was identified proximally and distally to the tumor and carefully dissected free from the periphery of the mass. Similarly, the popliteal artery and vein were identified and protected. Their preservation required careful sharp and blunt dissection along the tumor capsule, ensuring clear margins. Intraoperative nerve stimulation was used to confirm nerve integrity.
*
Muscle Excision:
The tumor, along with a wide cuff of surrounding normal muscle tissue, was resected en bloc. This included the entire semimembranosus and semitendinosus muscles, which were directly involved or heavily irradiated, and significant portions of the adjacent biceps femoris and adductor magnus muscles, extending approximately 2-3 cm into healthy tissue where possible. The principle of achieving a true "wide" margin was paramount.
*
Specimen Orientation:
The resected specimen was meticulously oriented with sutures and sent for frozen section analysis of the peripheral margins. All margins were confirmed negative by frozen section. This ensures immediate feedback on oncologic clearance.
*
Hemostasis:
Meticulous hemostasis was achieved using bipolar cautery and ligatures.
*
Wound Irrigation:
The surgical bed was thoroughly irrigated.
Reconstruction:
Due to the large soft tissue defect created by the wide excision, particularly the significant muscle loss in the posterior compartment, reconstruction was essential to provide adequate soft tissue coverage, protect the neurovascular bundle, and optimize functional recovery.
*
Local Flap:
A large fascio-cutaneous flap was mobilized from the adjacent healthy posterior thigh tissue to cover the defect. This involved extensive undermining and careful elevation to ensure adequate vascularity.
*
Drainage:
Two large-bore suction drains were placed deep within the wound bed to prevent hematoma and seroma formation, crucial for wound healing and reducing infection risk.
*
Closure:
The deep fascia was approximated where possible without tension. The subcutaneous tissue and skin were closed in layers using absorbable sutures for the subcutaneous tissue and non-absorbable staples for the skin.
The entire procedure lasted approximately 4 hours, with estimated blood loss of 300 mL. The patient tolerated the procedure well.
Post-Operative Protocol & Rehabilitation
The post-operative course and rehabilitation plan for this patient were carefully orchestrated to optimize wound healing, functional recovery, and long-term oncologic surveillance.
Immediate Post-Operative Period (Days 0-7):
*
Pain Management:
Multimodal analgesia was initiated, including patient-controlled analgesia (PCA) with opioids, supplemented by NSAIDs (if no contraindications) and acetaminophen. The pre-operative sciatic nerve block significantly aided immediate post-operative pain control.
*
Wound Care:
Drains were monitored for output. Dressings were changed daily, and the wound site was inspected for signs of infection (erythema, swelling, discharge) or flap compromise. Drains were typically removed when output was consistently less than 30-50 mL over 24 hours (usually 3-5 days post-op).
*
Deep Vein Thrombosis (DVT) Prophylaxis:
Continued with subcutaneous low molecular weight heparin (LMWH) until full ambulation, along with pneumatic compression devices.
*
Mobilization:
Early, gentle ankle pump exercises and knee flexion/extension within a comfortable range of motion were encouraged while supine. Bedside chair transfers began on Post-operative Day 1 or 2, with partial weight-bearing (PWB) as tolerated using crutches or a walker, with strict instructions to avoid excessive tension on the surgical wound.
*
Pathology Review:
The final histopathology report, including detailed margin analysis, was reviewed by the multidisciplinary tumor board to confirm negative margins and determine the need for adjuvant therapy. In this case, wide negative margins were confirmed.
Early Rehabilitation Phase (Weeks 1-6):
*
Goals:
Protect wound healing, reduce edema, gradually increase range of motion, initiate gentle strengthening, and progress weight-bearing.
*
Physical Therapy (PT):
Inpatient PT began immediately. Exercises focused on:
*
Range of Motion:
Passive and active-assisted ROM exercises for the hip and knee, avoiding aggressive stretching of the posterior thigh musculature. Emphasis on knee extension to prevent flexion contracture and hip extension.
*
Strengthening:
Isometric quadriceps sets, gluteal sets, and gentle ankle strengthening. Gradually progressing to light resistance exercises for hip abductors, adductors, and quadriceps. Hamstring strengthening was significantly delayed due to their excision and to protect the reconstruction.
*
Gait Training:
Progression from PWB to full weight-bearing (FWB) as tolerated, using assistive devices, focusing on proper gait mechanics.
*
Edema Control:
Compression garments and elevation as needed.
*
Occupational Therapy (OT):
Assessment for activities of daily living (ADLs) and provision of adaptive equipment if necessary.
Intermediate Rehabilitation Phase (Weeks 6-12):
*
Goals:
Regain functional strength, improve endurance, normalize gait, and address specific deficits resulting from muscle resection.
*
PT Progression:
*
Strengthening:
Progressive resistive exercises for all major muscle groups, with continued caution for hamstring engagement. Introduction of proprioceptive and balance training.
*
Cardiovascular Conditioning:
Stationary cycling or elliptical trainer (low impact).
*
Functional Activities:
Stair climbing, uneven surfaces, progressing towards sport-specific movements if applicable.
*
Patient Education:
Emphasis on long-term implications of muscle loss, compensation strategies, and body mechanics.
Late Rehabilitation Phase (Month 3 onwards):
*
Goals:
Maximize functional return, address residual deficits, and safely integrate into desired activities.
*
PT Progression:
Advanced strengthening, power, and agility training. Gradual reintroduction of light hamstring strengthening under strict supervision, focusing on eccentric control.
*
Return to Activity:
Gradual return to work, recreational activities, and low-impact sports, guided by functional assessment and patient tolerance.
Oncologic Surveillance:
This is a critical component of post-operative care for sarcoma patients.
*
Clinical Examination:
Regular physical examinations, including palpation of the surgical site and regional lymph nodes (every 3 months for the first 2-3 years, then every 6 months for years 3-5, then annually).
*
Local Imaging:
MRI of the surgical site (every 6 months for the first 2 years, then annually for 3-5 years) to monitor for local recurrence.
*
Distant Staging:
CT chest (every 6 months for the first 2 years, then annually for 3-5 years) as the lung is the most common site for distant metastasis from UPS.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
*
"Any Mass > 5 cm is Sarcoma Until Proven Otherwise":
This clinical axiom is paramount. Any rapidly growing, firm, deep-seated soft tissue mass larger than 5 cm in an adult, especially in the thigh, mandates aggressive workup for sarcoma. Do not attribute it to benign etiologies without definitive proof.
*
MRI is the Gold Standard for Local Staging:
High-resolution MRI with and without contrast is indispensable. It provides critical information regarding tumor size, exact anatomical location, relationship to neurovascular structures, bone involvement, and compartmentalization, all vital for surgical planning.
*
Multidisciplinary Team Approach:
Sarcoma management is complex and requires a highly specialized team (orthopedic oncologist, medical oncologist, radiation oncologist, pathologist, radiologist). Case discussions in a dedicated sarcoma tumor board are mandatory for optimal patient care and treatment sequencing.
*
Biopsy is Key and Must Be Planned Meticulously:
*
Core Needle Biopsy (CNB) is Preferred:
Image-guided CNB (ultrasound or CT) by an experienced radiologist or surgeon is preferred over incisional or excisional biopsies. It provides sufficient tissue for diagnosis and ancillary studies (IHC, molecular genetics) while minimizing tumor spillage.
*
Biopsy Tract Excision:
The biopsy tract must always be planned such that it can be completely excised en bloc with the definitive tumor resection to avoid seeding and local recurrence.
*
Achieving Wide, Negative Margins is the Single Most Important Prognostic Factor:
The primary goal of surgery is an R0 resection (microscopically negative margins). This often necessitates sacrificing normal tissues (muscle, nerve segments, bone) to achieve oncologic clearance.
*
Neoadjuvant Radiation Therapy:
Consider for large (>5 cm), high-grade, deep-seated tumors. It can improve local control, facilitate limb-sparing surgery, and potentially reduce recurrence rates. Its timing (pre- vs. post-operative) has implications for wound healing.
*
Long-Term Follow-Up is Mandatory:
Sarcomas have a propensity for both local recurrence and distant metastasis (most commonly to the lungs) even years after initial treatment. A rigorous surveillance schedule with clinical exams and appropriate imaging is critical.
Pitfalls:
*
Inadequate Initial Biopsy:
Performing an unplanned, haphazard incisional or excisional biopsy by a surgeon inexperienced in sarcoma management. This can contaminate multiple tissue planes, compromise limb-sparing options, and necessitate a wider, more complex re-excision or even amputation. It is a major cause of local recurrence.
*
Misinterpreting Imaging:
Mistaking a high-grade sarcoma for a benign lesion (e.g., a complicated hematoma, atypical lipoma) on initial imaging, leading to delay in diagnosis or inappropriate treatment. Be wary of large, heterogeneous, deep-seated masses in adults.
*
Failure to Perform Systemic Staging:
Omitting a CT chest, abdomen, and pelvis in the initial workup of a suspected high-grade sarcoma. This can miss synchronous metastases, leading to an inappropriate treatment plan (e.g., performing a major limb-sparing surgery on a patient with widespread metastatic disease).
*
Compromising Margins for Function:
Attempting to preserve excessive normal tissue at the expense of clear oncologic margins. While limb salvage is a goal, it should never compromise oncologic safety. Positive margins are a strong predictor of local recurrence.
*
Inadequate Post-Operative Rehabilitation or Follow-up:
Neglecting the intensive rehabilitation required after extensive resections, or failing to maintain strict oncologic surveillance, can lead to poor functional outcomes or delayed detection of recurrence/metastasis.
*
Underestimating the Aggressiveness of UPS:
Undifferentiated Pleomorphic Sarcoma is a high-grade, aggressive tumor with a significant risk of both local recurrence and distant metastasis. Treating it less aggressively than warranted can have dire consequences for the patient.

