Pathological Subtrochanteric Femoral Fracture Secondary to Multiple Myeloma: Diagnostic Nuances & Bone Scintigraphy
Key Takeaway
Multiple myeloma presenting as a pathological femoral fracture often involves elderly patients with pre-existing vague symptoms. Diagnostic imaging shows comminuted lytic lesions and a widespread lytic pattern. Crucially, Tc-99m bone scans typically show 'cold' or equivocal lesions due to osteoclast predominance, a key differentiator from other metastatic processes.
Introduction and Epidemiology
Clinical Presentation of Pathological Subtrochanteric Fractures
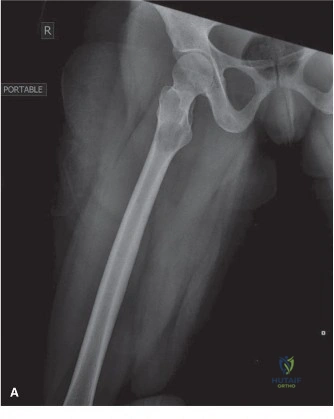
We present the case of Mr. J.A., an 82-year-old male, who presented to the emergency department following a mechanical fall from standing height. The patient reported immediate, severe pain in his left hip and thigh, rendering him unable to bear weight.
His past medical history is significant for well-controlled hypertension and type 2 diabetes mellitus. He reports a 6-month history of vague, intermittent lower back pain, which he attributed to "getting old," and had not sought medical attention for it. He denied any preceding trauma to the left hip or thigh. Prior to the fall, he noted increasing fatigue and mild unintentional weight loss of approximately 5 kg over the past year. He denied fever, chills, night sweats, or recent infections. There was no history of previous fractures or known osteoporosis. He is independent with all activities of daily living (ADLs) and resides at home with his wife. Social history includes a remote history of smoking, quit 20 years prior, and occasional alcohol consumption.
His current medications include lisinopril and metformin. No known drug allergies.
Upon arrival, Mr. J.A. was alert, oriented, and in obvious distress due to pain. Vital signs were stable. Inspection revealed the left lower extremity was externally rotated and shortened, characteristic of a proximal femoral fracture. There was no obvious gross deformity, skin tenting, or open wound. No ecchymosis was immediately visible. Direct palpation over the left greater trochanter and anterior hip joint elicited severe pain. No crepitus was appreciated due to patient guarding and pain. Distal pulses (dorsalis pedis and posterior tibial) were 2+ bilaterally, strong and symmetric. Skin temperature was normal. Active and passive range of motion of the left hip was severely limited and painful, particularly with any attempt at rotation or flexion. Neurological examination of the left lower extremity revealed intact sensation to light touch in the L2-S1 dermatomes. Motor strength was limited by pain, but no focal neurological deficits were identified. Capillary refill in the left toes was brisk.
Given the mechanism of injury (low-energy fall in an elderly patient) and the patient's reported preceding symptoms (back pain, fatigue, weight loss), a high index of suspicion for underlying pathological fracture was immediately raised, rather than a purely traumatic event.
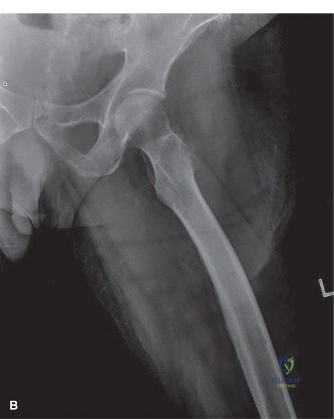
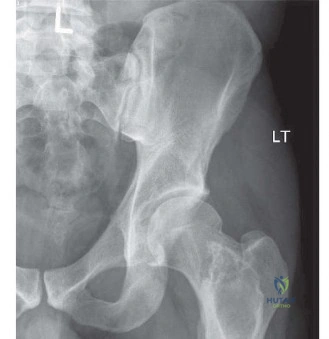
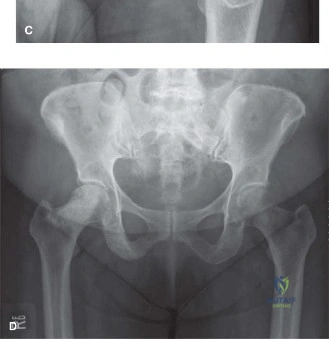
Initial plain radiographs of the left hip were obtained upon presentation. The anteroposterior (AP) and lateral views of the left hip demonstrated a comminuted, subtrochanteric fracture of the left femur. The fracture pattern involved significant cortical destruction and a large lytic lesion extending proximally into the intertrochanteric region and distally into the subtrochanteric area. The margins of the lytic lesion appeared ill-defined in several areas, raising strong suspicion for a neoplastic process. There was no evidence of periosteal reaction or overt sclerotic changes commonly associated with benign lesions or healing fractures.

Given the radiographic evidence of a pathological fracture, an expedited workup was initiated to identify the primary pathology and stage the disease. Computed Tomography (CT) Scan of the chest, abdomen, and pelvis was ordered to evaluate for primary malignancy or disseminated metastatic disease.
Pathophysiology of Multiple Myeloma Bone Disease
Multiple myeloma represents a malignant proliferation of plasma cells within the bone marrow, accounting for approximately ten percent of all hematologic malignancies. The skeletal manifestations of multiple myeloma are profound, driven by an uncoupling of normal bone remodeling. Myeloma cells secrete various cytokines, including Macrophage Inflammatory Protein 1 alpha and Receptor Activator of Nuclear factor Kappa B Ligand, which potently stimulate osteoclastogenesis. Simultaneously, osteoblast activity is actively suppressed through the secretion of inhibitors such as Dickkopf 1 and sclerostin.
This uncoupling results in aggressive osteolysis, presenting radiographically as punched-out lytic lesions, diffuse osteopenia, and ultimately, pathological fractures. The subtrochanteric region of the femur is particularly susceptible to these catastrophic failures due to its unique biomechanical environment, where immense physiological loads are concentrated across areas of focal cortical destruction.
Surgical Anatomy and Biomechanics
Osteology and Muscular Deforming Forces
The subtrochanteric region is anatomically defined as the proximal femoral segment extending from the lesser trochanter to approximately five centimeters distally into the proximal femoral diaphysis. This transition zone between the metaphyseal cancellous bone of the intertrochanteric region and the diaphyseal cortical bone is subjected to some of the highest mechanical stresses in the human skeleton.
Fractures in this region are notoriously difficult to reduce and maintain due to the powerful, asymmetric muscular forces acting upon the proximal and distal fragments. The proximal fragment is subjected to a triad of deforming forces. The abductors insert on the greater trochanter, driving the fragment into abduction. The short external rotators pull the fragment into external rotation. The iliopsoas, inserting on the lesser trochanter, flexes and externally rotates the proximal segment. Conversely, the distal fragment is drawn medially by the powerful adductor complex and proximally by the hamstrings and quadriceps, resulting in the classic flexed, abducted, and externally rotated proximal fragment with a shortened, adducted distal fragment.
Vascular Supply and Neurological Structures
The vascular supply to the subtrochanteric region is heavily reliant on the deep femoral artery and its perforating branches. The medial and lateral circumflex femoral arteries supply the proximal metaphysis and femoral head. During surgical exposure, particularly if an open reduction and internal fixation or tumor curettage is mandated, meticulous care must be taken to identify and ligate the perforating vessels to prevent significant hemorrhage, which is already a high risk in highly vascularized myeloma lesions. Neurologically, the sciatic nerve lies posterior to the surgical field and is generally protected during standard lateral approaches, provided retractors are placed carefully along the posterior border of the femur.
Biomechanical Considerations in Subtrochanteric Defects
Biomechanically, the subtrochanteric femur acts as a cantilever, subjected to high compressive forces along the medial cortex and immense tensile forces along the lateral cortex. Normal physiological loading during single-leg stance can generate forces across the subtrochanteric region exceeding three to four times body weight.
When a lytic lesion compromises the medial cortex, the bone loses its structural buttress against compression, leading to rapid varus collapse. Conversely, a lateral cortical defect acts as a stress riser in a tension zone, precipitating transverse or short oblique fractures. In the setting of multiple myeloma, where cortical thinning is often circumferential, the bone's torsional and bending rigidity is exponentially decreased. Surgical constructs must therefore bypass the zone of pathology entirely, spanning from the dense cancellous bone of the femoral head to the intact distal femoral diaphysis, effectively transferring load off the compromised segment.
Indications and Contraindications
The management of pathological subtrochanteric fractures requires a multidisciplinary approach involving orthopedic oncology, medical oncology, and radiation oncology. The primary goals of surgical intervention are immediate pain relief, restoration of mechanical stability, and facilitation of early mobilization to prevent the cascade of complications associated with prolonged bed rest in oncologic patients.
Operative Versus Non Operative Management
| Clinical Scenario | Management Strategy | Rationale and Considerations |
|---|---|---|
| Completed Pathological Fracture | Operative Fixation | Immediate stabilization required. Cephalomedullary nailing is the gold standard. Cement augmentation if severe bone loss. |
| Impending Fracture Mirels Score > 8 | Prophylactic Operative Fixation | High risk of spontaneous fracture. Prophylactic nailing reduces morbidity compared to fixing a completed fracture. |
| Impending Fracture Mirels Score < 7 | Non Operative Management | Lower fracture risk. Manage with systemic therapy, bisphosphonates, and localized radiotherapy. Close surveillance required. |
| Patient with Life Expectancy < 4 Weeks | Non Operative Palliation | Surgical morbidity outweighs benefits. Focus on aggressive pain management, positioning, and hospice care. |
| Severe Medical Comorbidities Unfit for Anesthesia | Non Operative Palliation | Optimization attempted; if unsuccessful, skeletal traction or palliative bracing may be utilized, though highly morbid. |
| Solitary Plasmacytoma Contained Lesion | Operative Fixation with Curative Intent | May require wide resection and endoprosthetic reconstruction if curative intent is established by oncology. |
Pre Operative Planning and Patient Positioning
Diagnostic Imaging and Bone Scintigraphy Nuances
The diagnostic workup for a suspected pathological fracture secondary to multiple myeloma must be comprehensive yet expedited. Following the initial plain radiographs and the CT scan of the chest, abdomen, and pelvis, specific attention must be directed toward oncologic staging and assessing the global skeletal burden.

A critical diagnostic nuance in multiple myeloma lies in the utility—or rather, the lack thereof—of standard Technetium-99m methylene diphosphonate bone scintigraphy. Standard bone scans rely on osteoblastic activity and regional blood flow for radiotracer uptake. Because multiple myeloma pathologically suppresses osteoblast function while upregulating osteoclasts, pure lytic myeloma lesions frequently present as "cold" or false-negative on standard bone scintigraphy.
Therefore, relying on a standard bone scan to stage skeletal involvement in myeloma is a fundamental clinical error. Instead, the gold standard imaging modalities for staging multiple myeloma bone disease include Whole Body Low Dose CT, Whole Body MRI, or 18F-FDG PET-CT. These modalities directly visualize marrow infiltration and lytic destruction regardless of osteoblastic response, providing an accurate map of impending structural failures throughout the axial and appendicular skeleton.
Systemic Optimization and Oncologic Staging
Preoperative laboratory evaluation must include a complete blood count, comprehensive metabolic panel (with specific attention to serum calcium and renal function), coagulation profile, and a type and crossmatch for potential massive transfusion. Multiple myeloma patients frequently present with the "CRAB" criteria: Hypercalcemia, Renal insufficiency, Anemia, and Bone lesions.
Hypercalcemia must be aggressively managed preoperatively with aggressive intravenous hydration, loop diuretics, and potentially bisphosphonates or calcitonin to prevent life-threatening cardiac arrhythmias. Renal optimization is critical, as myeloma cast nephropathy and contrast-induced nephropathy from staging scans can precipitate acute renal failure. Furthermore, a serum protein electrophoresis, urine protein electrophoresis, and serum free light chain assay should be drawn to confirm the diagnosis and establish baseline tumor markers.
Implant Selection and Biomechanical Planning
For subtrochanteric pathological fractures, standard locked intramedullary nails are biomechanically insufficient. The implant of choice is a reconstruction-type cephalomedullary nail. This device provides rigid fixation into the femoral head and neck, bypassing the compromised subtrochanteric region, and spans the entire length of the femur to protect against future distal lesions.
The surgeon must meticulously plan the nail diameter, length, and the neck-shaft angle based on the contralateral intact femur. In cases of massive osteolysis where cortical contact cannot be achieved, the surgeon must plan for open tumor curettage and polymethylmethacrylate cement augmentation to restore the compressive buttress of the medial cortex and enhance screw purchase in the proximal segment.
Operating Room Setup and Patient Positioning
Optimal patient positioning is paramount for successful closed reduction and intramedullary nailing. The patient is placed supine on a radiolucent fracture table.
The operative leg is secured in the traction boot, and the contralateral leg is placed in a hemilithotomy position or scissored posteriorly to allow unhindered access for the C-arm fluoroscope. The C-arm must be positioned to easily obtain true AP and lateral views of the femoral head, neck, subtrochanteric region, and the entire femoral shaft down to the distal condyles. Gentle longitudinal traction is applied. Due to the pathological nature of the bone, aggressive traction must be avoided to prevent iatrogenic neurovascular injury or propagation of the fracture. The torso is adducted away from the operative side to allow a straight-line trajectory for the rigid reamers and the intramedullary nail.
Detailed Surgical Approach and Technique
Choice of Surgical Approach
While prophylactic nailing of impending fractures can often be performed percutaneously, completed pathological subtrochanteric fractures with significant displacement or massive lytic defects frequently require a limited open approach. An open approach allows for direct fracture reduction, intralesional tumor curettage, and targeted cement augmentation. A lateral approach to the proximal femur is utilized, incising the fascia lata and elevating the vastus lateralis anteriorly off the intermuscular septum to expose the subtrochanteric region.
Step by Step Intramedullary Nailing Technique
- Entry Point and Guidewire Placement: A longitudinal incision is made proximal to the greater trochanter. The fascial layers are split in line with their fibers. Under fluoroscopic guidance, the entry point is established. For a trochanteric entry nail, the precise starting point is on the medial aspect of the greater trochanter, slightly anterior to the central axis on the lateral view. A guide pin is advanced into the proximal femur.
- Opening the Canal and Reduction: The proximal cortex is opened with a rigid entry reamer. A ball-tipped guidewire is then introduced. Navigating the guidewire across the subtrochanteric fracture site can be challenging due to the deforming forces. Finger reduction tools, percutaneous joy-sticks, or a limited open reduction via the lateral approach may be necessary to align the proximal and distal segments. The guidewire is advanced to the level of the physeal scar in the distal femur.
- Reaming the Medullary Canal: Sequential flexible reaming is performed. In pathological bone, reaming must be performed with caution to avoid eccentric reaming or cortical perforation. The canal is typically reamed 1.0 to 1.5 millimeters larger than the planned nail diameter to prevent excessive hoop stresses during nail insertion, which could shatter the fragile osteopenic cortex.

- Nail Insertion and Proximal Locking: The selected cephalomedullary nail is assembled on the insertion jig and advanced over the guidewire. Forceful impaction is strictly contraindicated. Once the nail is seated at the correct depth, the proximal reconstruction screws are placed into the femoral head and neck. The inferior screw should rest in the inferior half of the femoral neck on the AP view and centrally on the lateral view, maximizing the tip-apex distance biomechanics.
- Distal Locking: Distal locking is performed using a perfect circles freehand technique under fluoroscopy. A minimum of two distal interlocking screws should be utilized to provide adequate torsional and axial stability, particularly in the setting of extensive diaphyseal disease.
Tumor Curettage and Cement Augmentation
In cases like Mr. J.A.'s, where a large lytic defect compromises the structural integrity of the subtrochanteric region, hardware alone will inevitably fail due to cyclic fatigue. Through the limited lateral approach, a cortical window is created, or the existing fracture lines are utilized to access the intramedullary tumor.
Aggressive intralesional curettage is performed using ring curettes and high-speed burrs to remove the gross macroscopic myeloma tissue. The cavity is aggressively irrigated. Prior to final nail insertion, or through the cortical window after nail placement, low-viscosity polymethylmethacrylate bone cement is injected into the defect. The cement must interdigitate with the surrounding cancellous bone and encapsulate the intramedullary nail, effectively creating a composite structure that restores the compressive medial buttress and prevents varus collapse. Care must be taken to avoid cement extrusion into the soft tissues or the hip joint.
Complications and Management
The surgical management of pathological fractures in the setting of multiple myeloma is fraught with potential complications, exacerbated by the patient's systemic disease burden, immunocompromised state, and poor bone quality.
Intraoperative and Postoperative Complications
| Complication | Incidence | Prevention and Salvage Strategies |
|---|---|---|
| Intraoperative Hemorrhage | 10 - 20% | Preoperative embolization of highly vascular lesions. Meticulous surgical hemostasis. Availability of massive transfusion protocols. |
| Loss of Fixation Hardware Failure | 5 - 15% | Utilize cephalomedullary constructs spanning the entire femur. Employ PMMA cement augmentation for structural defects. Salvage requires revision to endoprosthesis. |
| Venous Thromboembolism VTE | 5 - 10% | Mechanical prophylaxis intraoperatively. Early postoperative chemical prophylaxis (LMWH or DOACs) tailored to platelet counts. Early mobilization. |
| Surgical Site Infection | 3 - 8% | Strict aseptic technique. Weight-based perioperative antibiotics. Minimize dead space. Salvage involves aggressive debridement, implant retention if stable, and targeted IV antibiotics. |
| Iatrogenic Fracture Propagation | 2 - 5% | Avoid forceful nail impaction. Over-ream the canal by 1.5mm. Use prophylactic cerclage wires if cortical integrity is questionable during reaming. |
| Hypercalcemic Crisis | 1 - 3% | Aggressive preoperative hydration. Monitor ionized calcium. Postoperative bisphosphonate or denosumab therapy in coordination with medical oncology. |
Post Operative Rehabilitation Protocols
Immediate Postoperative Care and Weight Bearing Status
The overarching goal of surgical intervention for pathological fractures is to allow immediate, unrestricted mobilization. Prolonged bed rest in a patient with multiple myeloma rapidly leads to deconditioning, deep vein thrombosis, pulmonary embolism, and hypostatic pneumonia.
Postoperatively, patients are encouraged to mobilize out of bed to a chair on postoperative day one. Weight-bearing status is dictated by the intraoperative stability of the construct. If a rigid cephalomedullary nail was placed and structural integrity was restored via cement augmentation, the patient is typically allowed weight-bearing as tolerated. If the fixation is tenuous due to massive unreconstructable bone loss, toe-touch or partial weight-bearing may be instituted, though this is less ideal in the elderly oncologic population. Physical therapy focuses on gait training, abductor strengthening, and restoring independence in activities of daily living.
Long Term Oncologic and Orthopedic Surveillance
Surgical fixation of a myeloma bone lesion is strictly a palliative or stabilizing procedure; it does not address the underlying systemic malignancy. Postoperatively, coordination with medical oncology is vital. The patient will require systemic therapy, which may include proteasome inhibitors (e.g., bortezomib), immunomodulatory drugs (e.g., lenalidomide), and corticosteroids.
Furthermore, bone-modifying agents such as intravenous zoledronic acid or subcutaneous denosumab are critical to inhibit further osteoclast-mediated bone destruction and reduce the risk of future skeletal-related events. Radiation oncology may be consulted for postoperative external beam radiotherapy to the operative bed to achieve local tumor control, typically initiated two to three weeks postoperatively to allow for initial soft tissue healing.
Orthopedic surveillance requires serial clinical examinations and radiographic evaluations at six weeks, three months, six months, and annually thereafter. The surgeon must monitor for signs of hardware loosening, loss of reduction, or disease progression at the margins of the fixation construct.
Summary of Key Literature and Guidelines
Mirels Classification and Prophylactic Fixation
The decision to operate on impending pathological fractures is heavily guided by the Mirels classification system, a widely validated scoring tool in orthopedic oncology. The Mirels score assigns points (1 to 3) based on four variables: the site of the lesion (upper extremity, lower extremity, peritrochanteric), the nature of the lesion (blastic, mixed, lytic), the size of the lesion (<1/3, 1/3-2/3, >2/3 of cortical diameter), and the presence of pain (mild, moderate, functional).
A cumulative score of 8 or higher indicates a high risk of spontaneous fracture and serves as a strong indication for prophylactic internal fixation. Prophylactic stabilization of an impending subtrochanteric fracture carries significantly lower perioperative morbidity, blood loss, and mortality compared to the surgical management of a completed fracture.
Current Evidence in Myeloma Fracture Management
Current academic literature emphasizes the necessity of full-length intramedullary devices for femoral lesions in multiple myeloma. Capanna's principles for the treatment of metastatic bone disease dictate that the entire involved bone should be protected. Short intramedullary nails or isolated plate constructs are associated with unacceptably high failure rates due to disease progression in the unprotected distal diaphyseal segments.
Furthermore, recent retrospective cohort studies have reinforced the survival benefit and improved functional outcomes associated with polymethylmethacrylate cement augmentation in massive subtrochanteric defects. Cement not only provides immediate structural support but also possesses a localized exothermic and cytotoxic effect that may aid in marginal tumor necrosis, though its primary utility remains biomechanical. The integration of advanced imaging modalities, multidisciplinary oncologic care, and biomechanically sound surgical reconstruction remains the cornerstone of managing complex pathological subtrochanteric fractures in the multiple myeloma population.
