Malignant Transformation of Osteochondroma: A Case of Secondary Peripheral Chondrosarcoma
Key Takeaway
Malignant transformation of an osteochondroma to a peripheral chondrosarcoma is suspected with new onset pain, rapid growth of a previously stable mass, or neurological symptoms. Diagnosis relies on MRI showing a cartilaginous cap exceeding 2 cm in adults, irregular calcifications, and signs of soft tissue invasion or marrow edema. Clinical history and imaging are crucial.
Patient Presentation and History
A 48-year-old male presented to the orthopedic oncology clinic complaining of insidious onset of increasing left leg pain over the past 8 months, localized to the proximal tibia. The pain was described as a dull ache, intermittently sharp with activity, and progressively worsened, interfering with sleep and ambulation. He reported a palpable mass in the same region, which he had been aware of since childhood, but it had always been asymptomatic and stable in size. Over the last year, he noted a definite increase in the mass size, along with the onset of pain. He denied any specific trauma to the limb.
Past medical history was significant for well-controlled hypertension and dyslipidemia. There was no family history of hereditary multiple osteochondromas (HMO/HME). He was a non-smoker and consumed alcohol occasionally. His occupation involved prolonged standing, which exacerbated his symptoms. He reported no systemic symptoms such as fever, night sweats, or unintentional weight loss. Neurological symptoms such as paresthesia or weakness in the foot were initially absent but began to manifest as mild numbness in the superficial peroneal nerve distribution over the past 2 months.
The patient's initial awareness of a benign "lump" for decades, followed by a recent change in symptoms (pain, rapid growth, neurological compromise), is a critical red flag, strongly suggesting the possibility of malignant transformation of a previously quiescent osteochondroma to a secondary peripheral chondrosarcoma. The risk of malignant transformation in a solitary osteochondroma is generally estimated at less than 1%, but the clinical presentation of new-onset pain and growth in a skeletally mature adult mandates an aggressive diagnostic workup to rule out secondary chondrosarcoma.
While this patient lacks a family history of Hereditary Multiple Exostoses (HME), it is imperative to recognize that mutations in the EXT1 and EXT2 tumor suppressor genes are frequently implicated in both sporadic and hereditary osteochondromas. The loss of heterozygosity or secondary mutations within the cartilaginous cap are the primary drivers of malignant degeneration into peripheral chondrosarcoma.
Clinical Examination Findings
On general inspection, the patient walked with an antalgic gait, favoring the left lower extremity. A visible, firm, fixed mass was observed on the anteromedial aspect of the left proximal tibia, approximately 10 cm distal to the joint line. The skin overlying the mass appeared unremarkable, with no signs of erythema, ulceration, or increased vascularity. There was no obvious muscular atrophy of the thigh or calf.
Palpation revealed a bony hard, immobile, and tender mass measuring approximately 8 x 6 cm at its widest dimensions. The mass was firmly adherent to the underlying bone. Deep palpation elicited significant pain. There was no discernible warmth or fluctuance. Examination of the knee joint itself revealed no effusion. The mass appeared to extend posteriorly, raising concerns for impending neurovascular compression.
Active and passive range of motion (ROM) of the left knee was limited by pain, with terminal extension restricted by 10 degrees and flexion to 110 degrees (normal 0-140 degrees). Passive ROM was painful at the extremes. The left hip and ankle ROM were full and painless.
Strength testing demonstrated 4/5 motor power in the tibialis anterior and extensor hallucis longus, consistent with mild weakness, likely due to pain and early peroneal nerve irritation. Sensation was diminished in the superficial peroneal nerve distribution over the dorsum of the foot, indicating compression or tethering of the common peroneal nerve or its branches as they course around the fibular neck in proximity to the expanding tumor mass. Distal pulses (dorsalis pedis and posterior tibial) were present, strong, and symmetrical bilaterally, and capillary refill was prompt. Popliteal fossa examination was unremarkable, though deep palpation was limited by the anterior mass.
The contralateral limb examination was entirely normal. No other masses were appreciated on a quick systemic survey, further supporting the diagnosis of a solitary lesion rather than an unrecognized polyostotic condition. Inguinal lymph node assessment was negative for palpable adenopathy, which is consistent with the typical biological behavior of chondrosarcomas, as they rarely metastasize via the lymphatic system.
Imaging and Diagnostics
Initial imaging commenced with plain radiographs of the left tibia and fibula, including the knee joint, followed by advanced cross-sectional imaging to accurately stage the local disease and assess the cartilaginous cap.
Plain Radiography Evaluation
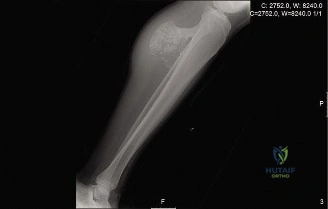
Antero-posterior and lateral views demonstrated a broad-based, pedunculated bony excrescence arising from the anteromedial metaphysis of the left proximal tibia. The lesion exhibited clear cortical and medullary continuity with the underlying parent bone, a hallmark feature of an osteochondroma.

However, several concerning features were noted:
* Irregular, indistinct cortical margin of the lesion, particularly at its superior aspect.
* Areas of flocculent, irregular calcification within an apparent soft tissue cap overlying the bony stalk, characteristic of "rings and arcs" or "popcorn" mineralization typical of a chondroid matrix.
* Some indistinctness of the adjacent periosteal reaction.
* The overall dimensions of the lesion appeared larger than typical benign variants, prompting further advanced imaging. The stalk itself appeared thickened, and the transition zone between the exostosis and the normal tibial diaphysis showed subtle areas of cortical remodeling.
Computed Tomography Analysis
A non-contrast Computed Tomography (CT) scan of the left lower extremity was obtained to better delineate the osseous architecture and matrix mineralization. CT is highly sensitive for detecting subtle cortical destruction and identifying the specific pattern of calcification within the tumor matrix. The CT confirmed the cortical and medullary continuity of the lesion with the proximal tibia. Crucially, it demonstrated focal areas of cortical breakthrough at the apex of the osteochondroma, a finding highly suspicious for malignant transformation. The cartilaginous cap exhibited dense, coalescing rings-and-arcs calcifications. CT also allowed for precise 3D templating to understand the relationship of the bony base to the tibial plateau and the proximal tibiofibular joint.
Magnetic Resonance Imaging Protocol
Magnetic Resonance Imaging (MRI) with and without intravenous gadolinium contrast is the gold standard for evaluating the unmineralized cartilaginous cap and assessing soft tissue extension. The MRI revealed a large, lobulated soft tissue mass arising from the surface of the osteochondroma.
The most critical finding was the thickness of the cartilaginous cap. On fluid-sensitive sequences (T2-weighted and STIR), the unmineralized cartilage cap demonstrated homogeneous hyperintensity and measured up to 3.2 cm in maximal thickness. In a skeletally mature adult, a cartilage cap thickness exceeding 1.5 cm to 2.0 cm is strongly indicative of malignant transformation to a secondary peripheral chondrosarcoma.
Post-contrast T1-weighted sequences demonstrated peripheral septal enhancement within the lobules of the cartilage cap, a classic feature of low-grade chondrosarcoma. The MRI also delineated the anatomical relationship between the tumor mass and the neurovascular bundle. The mass was noted to displace the anterior tibial artery and vein anteriorly and laterally, while the common peroneal nerve was draped over the lateral aspect of the expanding soft tissue component, explaining the patient's recent onset of neurological symptoms.
Image Guided Biopsy
Following the imaging workup, an image-guided core needle biopsy was performed. It is a critical oncological principle that the biopsy tract must be meticulously planned in consultation with the treating orthopedic oncologist to ensure it can be excised en bloc with the tumor during definitive surgery. The biopsy targeted the thickest, non-calcified portion of the cartilaginous cap, as a biopsy of the bony stalk will yield only benign bone and fail to diagnose the malignant transformation. Histopathological evaluation confirmed a Grade 1 (low-grade) secondary peripheral chondrosarcoma, characterized by increased cellularity, binucleated chondrocytes, and myxoid changes within the hyaline cartilage matrix, permeating the surrounding soft tissues.
Differential Diagnosis
The presentation of a growing, painful mass arising from the surface of a bone in an adult requires a focused differential diagnosis, primarily distinguishing between benign, active, and malignant cartilage-forming tumors, as well as surface-based osteogenic tumors.
| Diagnosis | Radiographic Features | Clinical Presentation | Histopathology |
|---|---|---|---|
| Secondary Peripheral Chondrosarcoma | Cortical/medullary continuity with parent bone. Thickened cartilage cap (>1.5 cm on MRI). Flocculent "rings and arcs" calcification. Cortical destruction at the stalk apex. | Skeletally mature adult. Insidious onset of new pain and growth in a previously stable, long-standing mass. Possible neurovascular compression. | Increased cellularity, binucleated cells, nuclear atypia, myxoid matrix changes. Invasion of the cartilaginous matrix into surrounding soft tissues or underlying bone stalk. |
| Benign Osteochondroma (with active bursitis) | Cortical/medullary continuity. Thin cartilage cap (<1.5 cm). Well-defined margins. Fluid collection over the exostosis on MRI (bursal formation). | Pain primarily due to mechanical irritation or bursitis, often activity-related. Mass size may fluctuate if a bursa is inflamed, but the bony component is stable. | Benign hyaline cartilage cap resembling normal growth plate. Regular arrangement of chondrocytes. No cytological atypia or invasive features. |
| Periosteal Chondrosarcoma | Surface lesion arising from the periosteum. Saucerization of the underlying cortex. Perpendicular periosteal reaction (hair-on-end). Chondroid matrix calcification. | Dull, aching pain. Palpable mass. Typically occurs in the metadiaphysis of long bones. | Lobules of malignant cartilage differentiating into bone at the periphery. Moderate cellularity and atypia. Does not arise from a pre-existing osteochondroma. |
| Parosteal Osteosarcoma | Broad-based surface mass. Dense, homogeneous, cloud-like ossification. "String sign" (radiolucent cleavage plane between tumor and underlying cortex). | Slow-growing, painless or mildly painful mass. Most common in the posterior distal femur. | Low-grade fibroblastic stroma with well-formed woven or lamellar bone trabeculae. Minimal cytological atypia. Lacks the extensive hyaline cartilage cap seen in chondrosarcoma. |
Surgical Decision Making and Classification
The management of secondary peripheral chondrosarcoma is dictated by the tumor's grade, anatomical location, and the functional demands of the patient.
Oncological Staging
Based on the Enneking Staging System for malignant bone tumors, this lesion is classified as a Stage IA tumor (Low Grade, Intracompartmental, No Metastases). However, due to the soft tissue extension of the cartilage cap beyond the anatomical compartment of the bone itself, it functions clinically as a Stage IB lesion (Low Grade, Extracompartmental).
Chondrosarcomas are notoriously resistant to both chemotherapy and radiation therapy due to their poor vascularity, slow rate of cell division, and the presence of the extracellular matrix which acts as a barrier to chemotherapeutic agents. Therefore, the mainstay of treatment is surgical resection.
Surgical Margins and Resection Strategy
The oncological goal is a wide en bloc resection. Intralesional procedures, such as curettage, are absolutely contraindicated for peripheral chondrosarcomas. Violating the tumor capsule or the cartilaginous cap leads to seeding of the surgical bed with malignant chondrocytes, resulting in a nearly 100% local recurrence rate. Recurrent chondrosarcomas often present with a higher histological grade and a significantly worse prognosis.
The surgical plan requires resecting the tumor with a continuous cuff of normal, healthy tissue in all dimensions. Given the location on the anteromedial proximal tibia, achieving a wide margin necessitates an extra-articular resection of the proximal tibia, including the biopsy tract, the overlying musculature (portions of the tibialis anterior and medial gastrocnemius), and potentially the proximal tibiofibular joint depending on lateral extension.
Reconstructive Options
Following a wide resection of the proximal tibia, the resultant massive bone and soft tissue defect requires complex reconstruction to restore limb length, stability, and extensor mechanism continuity. The primary options include:
- Osteoarticular Allograft: Provides biological reconstruction and allows for meniscal and ligamentous reattachment. However, it carries a high risk of non-union at the host-allograft junction, allograft fracture, and infection. It is generally reserved for younger patients with high biological healing potential.
- Arthrodesis: Utilizing a vascularized fibular graft or massive allograft with intramedullary nailing. Provides a durable, stable limb but sacrifices knee motion. Often considered a salvage procedure or for patients with high physical labor demands where an implant might fail.
- Endoprosthetic Reconstruction (Megaprosthesis): The most reliable method for immediate weight-bearing and early mobilization. Modern rotating-hinge proximal tibial replacements offer excellent medium-to-long-term survivorship. The critical challenge with proximal tibial megaprostheses is the reconstruction of the extensor mechanism, as the patellar tendon must be securely anchored to the prosthesis to prevent extensor lag or failure.
For this 48-year-old patient, a modular proximal tibial endoprosthetic reconstruction was selected to provide immediate stability, preserve joint motion, and allow for rapid rehabilitation.
Surgical Technique and Intervention
The surgical procedure is highly complex, requiring meticulous preoperative planning, advanced reconstructive skills, and a multidisciplinary approach involving orthopedic oncology and potentially plastic surgery for soft tissue coverage.
Patient Positioning and Anesthesia
The patient was placed in the supine position on a radiolucent operating table. General endotracheal anesthesia was administered. A Foley catheter was inserted. The left lower extremity was prepped and draped in a standard sterile fashion from the iliac crest to the toes, allowing for full manipulation of the limb.
A sterile tourniquet was applied to the proximal thigh. Crucially, the limb was exsanguinated by elevation only for 3 minutes prior to tourniquet inflation. The use of an Esmarch bandage is strictly contraindicated in oncological resections to prevent the mechanical embolization of tumor cells into the systemic circulation.
Surgical Approach and Exposure
An extensile anteromedial approach to the knee and proximal tibia was utilized. The incision incorporated the previous core needle biopsy tract with a 2 cm elliptical margin. The incision extended from the distal third of the thigh, over the medial parapatellar region, and down the anterior crest of the tibia, extending distal to the planned osteotomy site.
Full-thickness fasciocutaneous flaps were elevated. The deep dissection began with the identification and protection of the neurovascular structures. The common peroneal nerve was identified at the posterior border of the biceps femoris and traced distally as it wrapped around the fibular neck. Given the preoperative MRI findings of nerve tethering, careful neurolysis was performed, mobilizing the nerve away from the lateral aspect of the tumor capsule. The popliteal artery and vein were identified posteriorly and protected with vessel loops.
Tumor Resection
The boundaries of the tumor were identified without violating the pseudocapsule. The medial and lateral retinacula were incised. The patellar tendon was detached from its insertion on the tibial tubercle, taking a small wafer of bone if margins allowed, or sharply dissecting it if the tumor was in close proximity. The anterior cruciate ligament (ACL) and posterior cruciate ligament (PCL), as well as the medial and lateral collateral ligaments, were transected.
The level of the tibial osteotomy was determined based on preoperative MRI measurements, ensuring a minimum of a 3 cm marrow margin distal to the lowest extent of the tumor. The osteotomy was performed using an oscillating saw. The proximal tibiofibular joint was disarticulated. The posterior capsule of the knee was incised, and the entire proximal tibia, containing the tumor en bloc, was delivered from the surgical field.
The specimen was immediately sent to pathology for intraoperative frozen section analysis of the marrow margin and the surrounding soft tissue margins to confirm negative oncological margins before proceeding with reconstruction.
Endoprosthetic Reconstruction
Following confirmation of negative margins, the reconstruction phase commenced. The distal femur was prepared for the femoral component of the rotating-hinge knee system. Box cuts and chamfer cuts were made using standard instrumentation. The tibial diaphysis was reamed to accommodate the intramedullary stem of the tibial component.
A cemented, modular proximal tibial megaprosthesis was selected. The femoral and tibial stems were cemented using highly viscous polymethylmethacrylate (PMMA) bone cement mixed with antibiotics (tobramycin and vancomycin). The components were impacted into place, and excess cement was meticulously removed. The rotating hinge mechanism was assembled, linking the femoral and tibial components. Trial reductions confirmed excellent stability, restoration of limb length, and full passive range of motion from 0 to 120 degrees without impingement.
Extensor Mechanism Reconstruction and Soft Tissue Coverage
The most critical step in proximal tibial replacement is the reattachment of the patellar tendon to the prosthesis. Failure of this reconstruction leads to a catastrophic extensor lag.
A synthetic mesh tube (e.g., Trevira tube) was utilized. The host patellar tendon was sutured securely into the proximal end of the mesh using heavy, non-absorbable, krackow-style locking sutures. The distal end of the mesh was then secured to the designated attachment site on the proximal tibial prosthesis.
To enhance the biological healing of the extensor mechanism and provide robust soft tissue coverage over the metallic hinge mechanism, a medial gastrocnemius rotational flap was harvested. The medial head of the gastrocnemius was detached from its femoral origin, mobilized on its neurovascular pedicle (the medial sural artery), and rotated anteriorly. The muscle belly was sutured over the synthetic mesh and the proximal prosthesis, providing a highly vascularized bed. A split-thickness skin graft could be applied over the muscle if primary skin closure was not possible, though in this case, the fasciocutaneous flaps were advanced and closed primarily over closed suction drains.
Post Operative Protocol and Rehabilitation
The postoperative management following a massive endoprosthetic reconstruction requires a delicate balance between protecting the fragile extensor mechanism repair and preventing joint stiffness and deep vein thrombosis.
Immediate Postoperative Care
The patient was placed in a hinged knee brace locked in full extension immediately postoperatively. The limb was elevated to minimize edema. Intravenous prophylactic antibiotics were continued for 24 hours. Deep vein thrombosis (DVT) prophylaxis was initiated with low molecular weight heparin (LMWH) and mechanical sequential compression devices.
The closed suction drains were monitored closely and removed when output dropped below 30 cc per 24-hour period, typically on postoperative day 2 or 3. The viability of the skin flaps and the underlying medial gastrocnemius flap was assessed serially.
Physical Therapy Progression
- Weeks 0-6: The patient was allowed touch-down weight bearing (TDWB) with crutches or a walker. The knee brace remained locked in full extension during ambulation and sleep. Passive range of motion (PROM) was initiated strictly under the guidance of a physical therapist, limited to 0-30 degrees of flexion to protect the patellar tendon repair. Active knee extension was strictly prohibited.
- Weeks 6-12: Weight-bearing was progressively advanced to partial, then full weight-bearing as tolerated. The brace was unlocked to allow 0-60 degrees of flexion. Active-assisted range of motion was initiated. Isometric quadriceps strengthening began cautiously.
- Months 3-6: The brace was gradually weaned. Progressive resistance exercises for the quadriceps and hamstrings were introduced. The goal was to achieve a functional range of motion (0-100 degrees) and eliminate any extensor lag.
Oncological Surveillance
Despite the low-grade nature of the tumor, oncological surveillance is mandatory due to the risk of local recurrence and, less commonly, pulmonary metastasis. The surveillance protocol includes:
* Physical examination and plain radiographs of the reconstructed limb every 3 months for the first 2 years, every 6 months for years 3-5, and annually thereafter to assess for implant loosening, wear, or local bony recurrence.
* Non-contrast CT of the chest every 6 months for the first 2 years, then annually to screen for pulmonary metastases.
* MRI of the local surgical bed with metal-artifact reduction sequences (MARS) may be considered if there is clinical suspicion of soft tissue local recurrence, though the metallic artifact often limits interpretation.
Clinical Pearls and Pitfalls
Clinical Pearls:
* Cartilage Cap Thickness is Key: In the evaluation of an osteochondroma, MRI measurement of the unmineralized cartilage cap is the single most important imaging parameter. A cap >1.5 cm in an adult is highly suspicious for secondary chondrosarcoma.
* Biopsy the Cap, Not the Stalk: Core needle biopsies must target the non-calcified, soft tissue cartilaginous cap. Biopsying the calcified stalk will yield a false-negative result of benign bone.
* Extensor Mechanism Protection: The longevity and functional outcome of a proximal tibial replacement depend heavily on the extensor mechanism reconstruction. Utilizing a medial gastrocnemius flap significantly improves the biological environment and reduces the risk of skin necrosis and infection over the implant.
Clinical Pitfalls:
* Intralesional Excision: Misdiagnosing a secondary chondrosarcoma as a benign osteochondroma and performing a marginal or intralesional excision (e.g., "shaving" the bump) is a catastrophic error. This guarantees local recurrence, often with a higher histological grade, and severely complicates subsequent limb-salvage surgery.
* Esmarch Tourniquet Use: Utilizing an Esmarch bandage to exsanguinate a limb with a known or suspected malignancy can force tumor cells into the venous system. Always exsanguinate by elevation alone.
* Neglecting the Biopsy Tract: Failing to excise the biopsy tract en bloc with the tumor specimen risks local seeding of malignant cells in the superficial soft tissues. The biopsy tract must be tattooed or clearly documented and excised with a wide margin.
