Patient Presentation & History
A 48-year-old male presented to the orthopedic oncology clinic complaining of insidious onset of increasing left leg pain over the past 8 months, localized to the proximal tibia. The pain was described as a dull ache, intermittently sharp with activity, and progressively worsened, interfering with sleep and ambulation. He reported a palpable mass in the same region, which he had been aware of since childhood, but it had always been asymptomatic and stable in size. Over the last year, he noted a definite increase in the mass size, along with the onset of pain. He denied any specific trauma to the limb.
Past medical history was significant for well-controlled hypertension and dyslipidemia. There was no family history of hereditary multiple osteochondromas (HMO/HME). He was a non-smoker and consumed alcohol occasionally. His occupation involved prolonged standing, which exacerbated his symptoms. He reported no systemic symptoms such as fever, night sweats, or unintentional weight loss. Neurological symptoms such as paresthesia or weakness in the foot were initially absent but began to manifest as mild numbness in the superficial peroneal nerve distribution over the past 2 months.
The patient's initial awareness of a benign "lump" for decades, followed by a recent change in symptoms (pain, rapid growth, neurological compromise), is a critical red flag, strongly suggesting the possibility of malignant transformation of a previously quiescent osteochondroma to a secondary peripheral chondrosarcoma.
Clinical Examination
On general inspection, the patient walked with an antalgic gait, favoring the left lower extremity. A visible, firm, fixed mass was observed on the anteromedial aspect of the left proximal tibia, approximately 10 cm distal to the joint line. The skin overlying the mass appeared unremarkable, with no signs of erythema, ulceration, or increased vascularity. There was no obvious muscular atrophy of the thigh or calf.
Palpation revealed a bony hard, immobile, and tender mass measuring approximately 8 x 6 cm at its widest dimensions. The mass was firmly adherent to the underlying bone. Deep palpation elicited significant pain. There was no discernible warmth or fluctuance. Examination of the knee joint itself revealed no effusion.
Active and passive range of motion (ROM) of the left knee was limited by pain, with terminal extension restricted by 10 degrees and flexion to 110 degrees (normal 0-140 degrees). Passive ROM was painful at the extremes.
The left hip and ankle ROM were full and painless. Strength testing demonstrated 4/5 motor power in the tibialis anterior and extensor hallucis longus, consistent with mild weakness, likely due to pain and early peroneal nerve irritation. Sensation was diminished in the superficial peroneal nerve distribution (dorsum of the foot). Distal pulses (dorsalis pedis and posterior tibial) were present, strong, and symmetrical bilaterally, and capillary refill was prompt. Popliteal fossa examination was unremarkable.
The contralateral limb examination was entirely normal. No other masses were appreciated on a quick systemic survey.
Imaging & Diagnostics
Initial imaging commenced with plain radiographs of the left tibia and fibula, including the knee joint.
Plain Radiographs
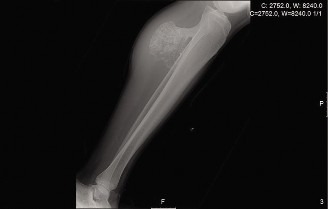
Antero-posterior and lateral views demonstrated a broad-based, pedunculated bony excrescence arising from the anteromedial metaphysis of the left proximal tibia. The lesion exhibited clear cortical and medullary continuity with the underlying parent bone, a hallmark feature of an osteochondroma. However, several concerning features were noted:
* Irregular, indistinct cortical margin of the lesion, particularly at its superior aspect.
* Areas of flocculent, irregular calcification within an apparent soft tissue cap overlying the bony stalk.
* Some indistinctness of the adjacent periosteal reaction.
* The overall dimensions of the lesion appeared larger than typical benign osteochondromas, and there was a subtle hint of an expanded, less defined cartilaginous cap.
Magnetic Resonance Imaging (MRI)
Due to the concerning clinical presentation and plain radiographic findings, a full staging MRI of the left tibia and knee with intravenous contrast was performed. This was crucial for assessing the cartilaginous cap thickness, marrow involvement, and soft tissue extent, including neurovascular relationships.
MRI findings were highly suggestive of malignant transformation:
* A large lesion (approximately 8 x 6 x 7 cm) was identified arising from the proximal tibial metaphysis.
* The cartilaginous cap measured approximately 3.5 cm in thickness, exceeding the critical threshold of 2 cm in an adult, which is highly indicative of malignant transformation to a peripheral chondrosarcoma. The cap itself demonstrated lobulated morphology with internal high T2 signal intensity and peripheral enhancement post-contrast, consistent with cartilage.
* There was evidence of mild marrow edema in the adjacent tibial metaphysis on STIR sequences, suggesting reactive changes or potential early invasion.
* The lesion was in close proximity to the superficial peroneal nerve, demonstrating early signs of compression. The popliteal vessels appeared displaced but patent, with no direct invasion.
* No skip lesions were identified within the tibia, and the knee joint appeared free of intra-articular extension.
Computed Tomography (CT)
A dedicated CT scan of the left proximal tibia was performed for better delineation of the bony architecture and cortical integrity. This confirmed the broad-based origin from the tibia, demonstrating the cortical and medullary continuity. It also provided superior detail regarding the calcification patterns within the cartilaginous cap, confirming the irregular, disorganized nature suggestive of chondrosarcoma. The CT helped in surgical planning by showing the precise bony attachment and its three-dimensional relationship to critical neurovascular structures.
Biopsy
Given the strong suspicion of malignant transformation, a computed tomography-guided core needle biopsy was performed. This was preferred over an open biopsy to minimize contamination of tissue planes and potential seeding, particularly important for achieving appropriate oncological margins. Biopsy material revealed atypical chondrocytes with increased cellularity, nuclear pleomorphism, and binucleation within a cartilaginous matrix, consistent with a low-grade (Grade 1) chondrosarcoma. The pathologist confirmed the diagnosis of a secondary peripheral chondrosarcoma arising from an osteochondroma.
Staging
Following the diagnosis, a full oncological workup was completed, including a chest CT to rule out pulmonary metastases and a bone scan (technetium-99m) to exclude other osseous lesions. Both were negative for metastatic disease. Based on the Enneking staging system, the tumor was classified as a Stage IA lesion (low-grade, intraosseous/intracompartmental, no metastases).
Differential Diagnosis
The presentation of a growing painful mass in an adult with a history of a pre-existing benign lesion necessitates a thorough differential diagnosis, particularly when considering malignant transformation.
| Condition | Key Clinical Features | Key Imaging Features | Management Approach |
|---|---|---|---|
| Secondary Chondrosarcoma | Insidious onset of pain in a known, previously asymptomatic osteochondroma; rapid increase in size; neurological symptoms (nerve compression). Often in middle-aged to older adults. | X-ray: Irregular surface, loss of cortical definition, increased radiolucency, irregular calcifications in soft tissue. MRI: Cartilaginous cap thickness > 2 cm (adults) with lobulated morphology, T2 hyperintensity, contrast enhancement, possible marrow edema/invasion. | Wide en bloc surgical resection with clear oncological margins (Enneking Stage I-II). Reconstruction as needed. Adjuvant therapy not typically effective for low-grade chondrosarcoma. Long-term surveillance for recurrence. |
| Symptomatic Osteochondroma | Pain due to mechanical irritation (e.g., bursa formation, tendon/nerve impingement, fracture of stalk, pseudoneurysm formation); stable size or slow benign growth. No rapid changes. | X-ray: Well-defined bony excrescence with clear cortical and medullary continuity with parent bone. MRI: Thin cartilaginous cap (< 2 cm in adults), often with overlying bursitis or evidence of mechanical irritation. No signs of aggressive features or marrow invasion. | Symptomatic excision if conservative measures fail. Curettage of the cap and removal of the stalk. Preservation of neurovascular structures. No need for wide oncological margins. |
| Osteosarcoma | Rapidly progressive pain, often severe, and swelling. May present with pathological fracture. Younger patient population (bimodal peak: adolescents/young adults and elderly). | X-ray: Aggressive periosteal reaction (sunburst, Codman's triangle), cortical destruction, osteoid matrix production, soft tissue mass. MRI: Extensive marrow involvement, large soft tissue component, heterogeneous enhancement. Skip lesions possible. | Neoadjuvant chemotherapy, followed by wide surgical resection and limb salvage or amputation. Adjuvant chemotherapy. |
| Myositis Ossificans | History of trauma (often minor or unremembered); rapidly growing, painful soft tissue mass that later becomes less painful and firm. Located within muscle. | X-ray: Zonal phenomenon - lucent center, peripheral calcification/ossification. Not attached to underlying bone in early stages. MRI: Varies with maturity; initially edematous, then peripheral ossification. No medullary continuity. | Observation; occasionally surgical excision after maturation (6-12 months) if causing functional impairment or pain. Biopsy can be misleading if performed too early or misinterpreted. |
Surgical Decision Making & Classification
The definitive diagnosis of a low-grade secondary peripheral chondrosarcoma based on biopsy, coupled with the patient's progressive pain and neurological symptoms, mandated operative intervention. Non-operative management was not an option due to the malignant nature of the lesion and its increasing morbidity.
The decision for surgical intervention was based on several critical factors:
1.
Malignant Diagnosis
: The biopsy confirmed chondrosarcoma, necessitating oncological resection.
2.
Symptomatic Progression
: The pain, mass enlargement, and early neurological compromise (superficial peroneal neuropathy) indicated an actively progressing lesion that required intervention to prevent further neurological deficit and improve quality of life.
3.
Risk of Further Progression
: Even low-grade chondrosarcomas have a potential for local recurrence and, less commonly, metastatic spread if not adequately resected.
The tumor was classified using the
Enneking Staging System for Musculoskeletal Sarcomas
:
*
Grade (G)
: G1 (Low-grade based on histology)
*
Site (T)
: T1 (Intracompartmental, within the osseous confines or adjacent soft tissues but not extending beyond the primary anatomic compartment). In this case, it was a peripheral lesion confined to the proximal tibial metaphysis and immediately adjacent soft tissue envelope without breaching the fascial planes into other compartments.
*
Metastasis (M)
: M0 (No regional or distant metastases).
Therefore, the tumor was classified as Stage IA (G1T1M0) . This staging dictates a wide surgical margin for local control, typically involving removal of the tumor en bloc with a cuff of normal surrounding tissue. For this low-grade lesion, a wide margin provides excellent local control and a high probability of cure. Marginal excision (intralesional or simple excision) carries a high risk of local recurrence and is not indicated for chondrosarcoma.
Due to the size and location of the lesion (proximal tibia), reconstruction considerations were paramount. While not intra-articular, the proximity to the knee joint and the weight-bearing nature of the tibia required careful planning to maintain structural integrity and optimize functional outcomes.
Surgical Technique / Intervention
The primary goal of surgery was an en bloc wide resection of the chondrosarcoma with adequate oncological margins, followed by reconstruction to ensure limb stability and function.
Pre-operative Planning:
Advanced imaging (CT and MRI) was used for precise templating. The bony attachment point, the extent of the cartilaginous cap, and its relationship to the patellar ligament, popliteal vessels, and peroneal nerve were meticulously mapped. A surgical safety margin of 2 cm of normal tissue around the tumor was aimed for.
Patient Positioning and Preparation:
The patient was positioned supine on the operating table. A high thigh tourniquet was applied but not inflated initially. The entire left lower limb, from the iliac crest to the toes, was prepared and draped in a sterile fashion to allow full manipulation of the limb and access to regional lymph nodes if needed (though not expected for a low-grade lesion). A bolster was placed under the ipsilateral hip to internally rotate the limb slightly, presenting the anteromedial aspect of the tibia.
Surgical Approach:
A straight longitudinal incision was made over the palpable mass, extending sufficiently proximal and distal to allow for adequate exposure and resection margins. The incision was centered over the mass, avoiding its contamination. The skin and subcutaneous tissues were incised. Full thickness skin flaps were elevated carefully, minimizing manipulation of the tumor to prevent capsular breach or tumor cell dissemination.
The superficial peroneal nerve was identified and protected proximally and distally, meticulously dissecting it away from the tumor capsule. The anterior compartment muscles were identified. The tumor, arising from the anteromedial proximal tibia, necessitated dissection between the tibialis anterior muscle and the medial gastrocnemius head. The sartorius and semitendinosus muscles were reflected as needed.
Resection:
The extent of the tumor was confirmed visually and by palpation. The resection plan involved removing the tumor en bloc with a cuff of surrounding normal muscle and periosteum. Osteotomies were performed with an oscillating saw, guided by pre-operative templating and intra-operative visual cues, ensuring at least a 2 cm margin of healthy bone proximally, distally, and posteriorly to the tumor attachment. The bone cut was through normal cortical and medullary bone.
The tumor was carefully detached from its muscular and fascial attachments, ensuring no direct contact with the tumor surface. The entire specimen was then delivered intact and sent for histopathological examination, with specific attention to the margins. Intraoperative frozen section analysis was not typically performed for low-grade chondrosarcoma margins, as interpretation can be challenging and the focus is on wide excision.
Reconstruction:
After wide resection, a significant segmental defect of the anteromedial proximal tibia was created. This particular defect, being extra-articular but involving a crucial load-bearing bone, required structural support.
In this case, a
vascularized fibular autograft
was not feasible due to the size of the defect. An
allograft reconstruction
was chosen, utilizing a structural cortical tibial segment from a bone bank. The allograft was contoured to match the resected tibial segment.
The allograft was secured to the host bone using a combination of bicortical screws and a locking plate construct. A long, pre-contoured locking plate (e.g., LCP Proximal Tibial Plate) was used, spanning the allograft and extending well into the healthy host bone both proximally and distally, ensuring rigid fixation. Lag screws were used to compress the allograft to the host bone at the osteotomy sites. Autogenous bone graft (harvested from the iliac crest or distal tibia) was packed at the host-allograft junctions to promote bony union.
Alternatively, for smaller defects or in cases where extensive muscle stripping is avoided, a simple defect without formal structural reconstruction might be considered, relying on the remaining tibia and soft tissue coverage. However, in this case with a low-grade tumor, adequate margins took precedence, creating a substantial defect requiring reconstruction.
Closure:
Hemostasis was meticulously achieved. The deep tissues were closed in layers using absorbable sutures. A subfascial drain was placed. The skin and subcutaneous tissues were closed with care to avoid tension. A sterile dressing was applied, and the limb was immobilized in a knee immobilizer or hinged knee brace set to restrict motion.
Post-Operative Protocol & Rehabilitation
The post-operative course focused on pain management, wound care, infection prophylaxis, and a structured rehabilitation program tailored to the reconstruction.
Immediate Post-Operative Period (Weeks 0-2):
- Pain Management : Multi-modal analgesia including opioids, NSAIDs, and regional nerve blocks (if applicable).
- Wound Care : Daily dressing changes, close monitoring for signs of infection (erythema, warmth, purulent discharge). Drain output monitored and removed when minimal.
- DVT Prophylaxis : Subcutaneous low-molecular-weight heparin or oral anticoagulants, along with mechanical prophylaxis (compression stockings, foot pumps).
- Immobilization : Knee immobilizer or hinged knee brace locked in full extension for stability of the allograft construct.
- Weight-Bearing : Strict non-weight-bearing (NWB) on the operated limb to protect the allograft-host junctions. Crutch or walker training initiated.
- Physiotherapy : Gentle active ankle ROM and isometric quadriceps/hamstring exercises to maintain muscle tone and prevent stiffness. Upper extremity strengthening to aid in mobility.
Early Rehabilitation (Weeks 2-6):
- Wound Healing : Staples/sutures removed at 2-3 weeks post-op. Scar management initiated.
- Weight-Bearing : Continued NWB. Some surgeons may allow touch-down weight-bearing (TDWB) if fixation is very robust.
- ROM : Gradual progression of knee ROM within the brace, guided by pain and surgeon's discretion. Typically, 0-30 degrees flexion initially, progressing to 0-60 degrees.
- Strengthening : Continued isometric exercises. Introduction of gentle active-assisted ROM exercises for the knee.
Intermediate Rehabilitation (Weeks 6-12):
- Radiographic Assessment : X-rays at 6 and 12 weeks to monitor allograft-host junction healing.
- Weight-Bearing : If radiographic evidence of healing is progressing, gradual progression to partial weight-bearing (PWB) in the brace, increasing by 25% increments over several weeks, eventually aiming for full weight-bearing (FWB) by 3-4 months post-op, assuming solid union.
- ROM : Continue to advance knee ROM, aiming for functional range (0-120 degrees) by the end of this phase.
- Strengthening : Progressive resistance exercises for quadriceps, hamstrings, and calf muscles. Balance and proprioceptive exercises initiated.
Late Rehabilitation & Return to Activity (Months 3-12+):
- Weight-Bearing : Full weight-bearing without the brace once radiographic union is solid.
- Functional Training : Focus on regaining full strength, endurance, and proprioception. Gait training to normalize walking pattern.
- Activity Modification : Avoid high-impact activities or contact sports until at least 12-18 months post-op, and only after complete radiographic union and full strength recovery.
- Long-term Surveillance : Regular clinical follow-up and imaging (X-rays, MRI) to monitor for local recurrence, allograft complications (non-union, fracture, infection), and potential metastases. For low-grade chondrosarcoma, recurrence risk is highest in the first 5 years but long-term surveillance is prudent.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
- Clinical Suspicion is Key : Any new or increasing pain, rapid growth, or neurological symptoms in a pre-existing osteochondroma, especially in an adult, mandates immediate investigation for malignant transformation. This is a common exam scenario.
- MRI for Cap Thickness : MRI is the gold standard for assessing cartilaginous cap thickness. A cap thickness > 2 cm in adults is the most reliable imaging feature suggestive of malignant transformation to a peripheral chondrosarcoma.
- Proper Biopsy : If malignancy is suspected, a well-planned, image-guided core needle biopsy (preferable) or open biopsy must be performed by or in consultation with an orthopedic oncologist to avoid tumor seeding and compromise of future definitive margins. The biopsy tract must be excisable en bloc with the definitive resection.
- Enneking Staging Dictates Margins : Understanding and applying the Enneking staging system is paramount for planning oncological resections. For a Stage IA chondrosarcoma, a wide surgical margin is curative and crucial for local control.
- Medullary Continuity : The hallmark of osteochondroma (and its transformed variant) is the continuity of the cortex and medulla of the lesion with the parent bone. This distinguishes it from other juxtacortical lesions.
- Neurovascular Proximity : Osteochondromas, particularly around the knee (distal femur, proximal tibia, proximal fibula), are notorious for causing neurovascular impingement (e.g., peroneal nerve palsy from proximal fibular lesions, popliteal artery pseudoaneurysm). Meticulous dissection and protection of these structures are vital.
- Reconstruction Considerations : For significant bone defects after wide resection, careful planning for reconstruction (allograft, endoprosthesis) is necessary to restore function and stability.
Pitfalls:
- Misinterpreting Benign Growth : Mistaking a rapidly growing or painful osteochondroma for a simple benign process without further investigation (MRI, biopsy).
- Inadequate Imaging : Relying solely on X-rays when malignancy is suspected. A high-quality MRI is essential for soft tissue and cartilaginous cap assessment.
- Suboptimal Biopsy : Performing a biopsy through an inappropriate approach or tract, contaminating unaffected tissue planes, which can complicate subsequent definitive resection and potentially necessitate a more morbid operation (e.g., amputation if the biopsy tract cannot be adequately resected with a wide margin).
- Insufficient Resection Margins : Performing a marginal or intralesional excision for a confirmed chondrosarcoma, which inevitably leads to high local recurrence rates. The adage "the first surgery is the best chance for cure" holds true.
- Ignoring Neurological Symptoms : Attributing neurological symptoms solely to pain or other causes without investigating potential nerve compression by the mass.
- Inadequate Rehabilitation : Not following a structured rehabilitation protocol can lead to stiffness, weakness, and poor functional outcomes, especially after complex reconstructions.
- Missing Systemic Staging : Failing to perform a full oncological staging (chest CT, bone scan) once a malignant diagnosis is made, potentially missing metastatic disease.
- Over-reliance on Frozen Section : For chondrosarcoma, intraoperative frozen section for margin assessment can be notoriously difficult due to the inherent cellularity variability of cartilage. Clinical judgment and pre-operative templating for wide margins are more reliable.
