Patient Presentation & History
The patient, a 72-year-old male, presented to the emergency department after a low-energy fall at home. He reported standing up from a chair, experiencing sudden, excruciating left hip pain, followed by an inability to bear weight and subsequent collapse. There was no direct high-impact trauma to the hip, which immediately raised suspicion for an underlying pathologic event.
His medical history included controlled hypertension (on ramipril), Type 2 Diabetes Mellitus (on metformin), and gout (managed with allopurinol). Of note, he reported chronic lower back pain, which had been progressively worsening over the past 6 months and was largely attributed to degenerative disc disease, self-managed with over-the-counter NSAIDs. He also reported diffuse bone aches, particularly in his ribs and sternum, dismissed as age-related. He denied any prior history of malignancy.
Social history revealed he lived alone and was previously independent in all activities of daily living (ADLs). He was a former smoker (quit 15 years prior) and an occasional alcohol consumer.
Review of systems elicited several significant findings:
*
Constitutional:
Profound fatigue and an unintentional weight loss of approximately 5 kg over the preceding 3-4 months.
*
Musculoskeletal:
Aside from the acute left hip pain, his chronic lower back pain had recently escalated in severity, and he described diffuse body aches, especially in the axial skeleton.
*
Hematological & Renal:
Denied easy bruising, bleeding, or changes in urinary habits.
Clinical Examination
Upon examination, the patient was alert and oriented but appeared acutely distressed due to pain. His mucous membranes exhibited pallor.
Vitals:
* Blood Pressure: 138/86 mmHg
* Heart Rate: 92 bpm
* Respiratory Rate: 18/min
* Oxygen Saturation: 96% on room air
* Temperature: 37.1°C
Inspection (Left Lower Extremity):
* The left lower limb demonstrated obvious shortening and external rotation, characteristic of a proximal femur fracture.
* No visible open wounds, significant ecchymosis, or gross swelling were noted around the hip.
* No other deformities of the knee or ankle were present.
Palpation (Left Lower Extremity):
* Exquisite tenderness was elicited over the left greater trochanter and the anterior hip joint line.
* Palpation of the lumbar spine and anterior chest wall (sternum and ribs) also revealed tenderness, correlating with his reported chronic pains.
* No palpable masses were identified locally.
* Distal pulses (dorsalis pedis and posterior tibial) were 2+ bilaterally, strong and symmetrical, confirming intact vascular supply.
Range of Motion (Left Hip):
* Active range of motion was severely painful and impossible to assess.
* Passive range of motion was markedly restricted and painful in all planes: flexion to approximately 10 degrees, abduction to 5 degrees, adduction at 0 degrees, and internal/external rotation were severely painful and limited, with significant guarding.
Neurological Assessment (Left Lower Extremity):
* Motor strength was 5/5 in ankle dorsiflexion, plantarflexion, great toe extension, knee extension, and knee flexion on the contralateral (right) limb. Assessment of left hip and knee motor function was precluded by pain.
* Sensory examination revealed intact light touch sensation in the L2-S1 dermatomes bilaterally.
* Deep tendon reflexes (patellar and Achilles) were 2+ bilaterally.
Systemic Examination:
*
Cardiovascular:
Regular rate and rhythm, normal S1S2, no audible murmurs.
*
Respiratory:
Lungs were clear to auscultation bilaterally.
*
Abdomen:
Soft, non-tender, non-distended, with no organomegaly.
*
Lymphatic:
No palpable lymphadenopathy.
Imaging & Diagnostics
Immediate plain radiographs of the left hip were obtained in the emergency department.
-
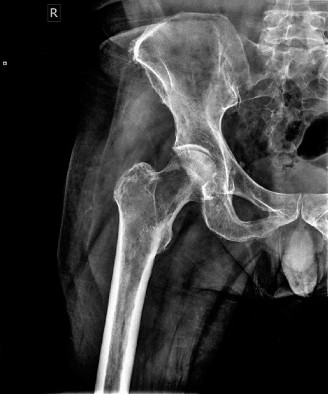
Initial X-ray Findings (Left Hip, AP & Lateral):
- These images revealed a severely comminuted subtrochanteric fracture of the left femur.
- Crucially, the bone quality appeared markedly osteopenic with widespread lytic lesions and irregular cortical destruction, particularly pronounced at the fracture site, extending into the intramedullary canal.
- A 'moth-eaten' pattern was distinctly visible in the cortex surrounding the fracture, highly indicative of a pathologic process.
-
 - This radiographic appearance strongly suggested an underlying pathologic fracture.
Prompted by these findings, a comprehensive diagnostic workup was initiated.
-
Laboratory Investigations:
- Full Blood Count (FBC): Hemoglobin: 9.8 g/dL (normocytic, normochromic anemia); WBC count: 7.2 x 10^9/L (normal); Platelet count: 210 x 10^9/L (normal).
- Renal Function Tests (RFTs): Creatinine: 1.8 mg/dL (significantly elevated from a baseline of 0.9 mg/dL documented 6 months prior); estimated Glomerular Filtration Rate (eGFR): 38 mL/min/1.73m^2 (indicating moderate to severe renal insufficiency).
- Electrolytes: Corrected Calcium: 12.5 mg/dL (pronounced hypercalcemia); Phosphate: 3.0 mg/dL (normal).
- Liver Function Tests (LFTs): Within normal limits.
- Inflammatory Markers: Erythrocyte Sedimentation Rate (ESR): 105 mm/hr (markedly elevated); C-Reactive Protein (CRP): 25 mg/L (moderately elevated).
- Serum Protein Electrophoresis (SPEP) with Immunofixation: Detected a distinct monoclonal IgG Kappa M-spike, confirming a monoclonal gammopathy.
- Urine Protein Electrophoresis (UPEP) with Immunofixation: Demonstrated Bence-Jones proteinuria (kappa light chains), indicative of free light chain excretion.
- Serum Free Light Chain Assay: Showed elevated kappa light chains and significantly suppressed lambda light chains, resulting in an abnormal kappa/lambda ratio.
- Beta-2 Microglobulin: Elevated.
- Lactate Dehydrogenase (LDH): Elevated.
- Alkaline Phosphatase: Slightly elevated at 130 U/L, a finding that can be inconsistent in myeloma.
- Parathyroid Hormone (PTH): Appropriately suppressed in response to hypercalcemia.
- Vitamin D: Levels were within the normal range.
-
Further Imaging:
- CT Scan of Left Femur with 3D Reconstructions: This confirmed the extensive lytic destruction associated with the subtrochanteric fracture, demonstrating significant cortical breach and intramedullary involvement. It provided critical detailed anatomical information for surgical planning, including the precise fracture morphology and assessment of residual bone stock.
-
 - Skeletal Survey (Plain X-rays of Skull, Entire Spine, Pelvis, Humeri): Revealed multiple, classic 'punched-out' lytic lesions without sclerotic reaction in the skull, several vertebral bodies (notably L2 and L4, correlating with his back pain), the left iliac wing, and the right proximal humerus.
- MRI of Entire Spine: Demonstrated diffuse bone marrow infiltration consistent with myeloma, particularly prominent in the lumbar and thoracic spine. It also identified pathological compression fractures in L2 and L4, confirming the etiology of his chronic back pain, and importantly, ruled out impending spinal cord compression.
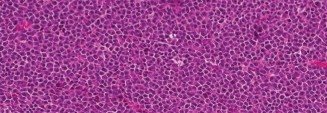
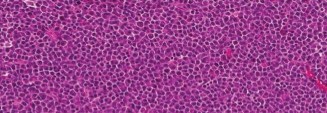
- Bone Marrow Biopsy and Aspirate (from posterior iliac crest): Pathology confirmed clonal plasma cells constituting over 60% of marrow cellularity, exhibiting an aberrant immunophenotype (CD138+, CD38+, CD45-). Fluorescence in situ hybridization (FISH) analysis identified high-risk cytogenetic abnormalities, including del(17p) and t(4;14).
- PET-CT (for staging): This whole-body scan demonstrated multiple metabolically active lesions throughout the axial and appendicular skeleton, consistent with widespread active multiple myeloma. The left subtrochanteric lesion showed very high avidity.
-
Diagnosis: Based on the presence of anemia, renal insufficiency, hypercalcemia, and widespread bone lesions (CRAB criteria), coupled with the findings from serum protein electrophoresis, free light chain assay, and confirmed bone marrow biopsy, the patient was diagnosed with Stage III Multiple Myeloma (International Staging System - ISS) with high-risk cytogenetics.
Differential Diagnosis
The presentation of a pathologic subtrochanteric fracture in an elderly patient with systemic symptoms necessitates a methodical approach to differential diagnosis, primarily considering other primary and secondary bone malignancies.
| Feature / Condition | Multiple Myeloma | Metastatic Carcinoma (e.g., Lung, Breast, Prostate, Renal) | Primary Bone Sarcoma (e.g., Osteosarcoma, Chondrosarcoma) |
|---|---|---|---|
| Demographics | Predominantly affects individuals over 60 years. Slight male predominance. | Varies widely based on primary tumor type (e.g., lung/prostate >50 years, breast any adult age). | Bimodal distribution: adolescents/young adults, and older adults (>60 years for secondary osteosarcoma or chondrosarcoma). |
| Clinical Presentation | Pathologic fracture (often multifocal), diffuse bone pain (axial skeleton common), fatigue, weight loss, recurrent infections, hypercalcemia symptoms. | Pathologic fracture (often solitary or few lesions), localized bone pain, symptoms related to primary cancer (e.g., cough, hematuria), weight loss. | Localized pain, soft tissue swelling, palpable mass. Pathologic fracture is less common, typically indicates an aggressive, high-grade lesion. Systemic symptoms are rare unless advanced. |
| X-ray Findings | Purely lytic, 'punched-out' lesions without sclerotic reaction. Diffuse osteopenia. Vertebral compression fractures. | Can be predominantly lytic (lung, renal, thyroid), blastic (prostate), or mixed (breast). Often show a sclerotic rim or mixed pattern. | Variable appearances. Osteosarcoma: mixed lytic/sclerotic, aggressive periosteal reaction (Codman's triangle, sunburst pattern). Chondrosarcoma: arc and ring calcifications, endosteal scalloping. |
| Lab Findings | CRAB criteria: Hypercalcemia, Renal failure, Anemia, Bone lesions. Monoclonal protein (M-spike on SPEP/UPEP), elevated Free Light Chains. High ESR. | Hypercalcemia (less common than myeloma, depends on tumor burden). Elevated specific tumor markers (e.g., PSA for prostate, CEA for colon). Elevated Alkaline Phosphatase common in osteoblastic or extensive lytic lesions. | Generally normal FBC/electrolytes. Alkaline Phosphatase may be elevated in osteosarcoma due to osteoblastic activity. No monoclonal protein. ESR may be elevated. |
| Pathology/Biopsy | Clonal plasma cells (>10% in bone marrow, or presence of plasmacytoma). Aberrant immunophenotype (CD138+, CD38+). | Carcinoma cells consistent with the primary tumor site (e.g., adenocarcinoma, squamous cell carcinoma). Positive immunohistochemical markers for primary. | Malignant cells forming osteoid (osteosarcoma), cartilage (chondrosarcoma), or other specific stromal elements. Often pleomorphic. |
| Imaging (CT/MRI/PET) | Diffuse marrow infiltration on MRI, widespread lytic lesions on CT, metabolically active lesions on PET. Usually multifocal. | Metastatic disease elsewhere (lungs, liver, brain) often identifiable. Primary tumor site usually discernible. | Localized aggressive tumor mass with cortical destruction and soft tissue extension. Skip lesions may be seen in osteosarcoma. Regional nodal involvement is possible. |
| Treatment Implications | Systemic chemotherapy (IMiDs, PIs, monoclonal antibodies), high-dose chemotherapy with autologous stem cell transplant. Local radiation for painful lesions. Orthopedic surgery for stabilization. | Systemic chemotherapy/hormonal therapy/targeted therapy. Local radiation. Orthopedic surgery for stabilization (often palliative). | Neo-adjuvant chemotherapy, wide surgical resection with limb salvage, adjuvant chemotherapy. Radiation for unresectable disease or positive margins. |
Surgical Decision Making & Classification
Given the patient's acute, comminuted, pathologic subtrochanteric fracture of the left femur, in the context of newly diagnosed, widespread multiple myeloma, operative intervention was deemed the most appropriate course of action.
Rationale for Operative Intervention:
- Pain Relief: Non-operative management of a severely unstable, pathologic fracture would result in intractable pain, significantly impairing the patient's quality of life.
- Early Mobilization and Functional Preservation: Essential for elderly patients, particularly those with malignancy, to prevent complications of immobility such as deep vein thrombosis (DVT), pulmonary embolism (PE), pneumonia, decubitus ulcers, and accelerated muscle atrophy. Early mobilization facilitates participation in systemic oncologic treatment.
- Local Tumor Control and Stability: Stabilization of the fracture prevents further collapse and progression of bone destruction at the fracture site. It also provides a mechanically sound construct to allow the application of adjuvant systemic and local oncologic therapies (e.g., radiation) without compromising fixation.
- Prevention of Impending Fractures: The presence of multiple other lytic lesions, even if not acutely fractured, indicated a high risk of future pathologic fractures. The chosen fixation strategy could incorporate prophylactic stabilization of adjacent compromised bone.
- Prognosis and Quality of Life: While multiple myeloma is currently incurable, maximizing functional independence and quality of life is a primary objective. Surgical stabilization directly contributes to achieving this goal.
Classification Systems Guiding Decision-Making:
-
Mirels Classification System:
Although primarily used for assessing the risk of
impending
pathologic fractures from metastatic bone disease, it is highly relevant here. This system scores lesions based on:
- Location: Weight-bearing long bones (femur, humerus) score higher.
- Size: Lesions involving >2/3 of the cortical circumference score higher.
- Type of Lesion: Lytic lesions score higher than blastic.
-
Pain:
Mechanical pain scores higher than non-mechanical pain.
The patient's subtrochanteric location, extensive lytic nature, and mechanical pain associated with the fracture (even pre-collapse) would yield a Mirels score of 9-12, strongly advocating for prophylactic stabilization if the fracture had not already occurred. With an overt fracture, Mirels' score reinforces the need for robust operative management.
- AO/OTA Fracture Classification: Descriptively, the fracture could be classified as 32-A3.3, denoting a comminuted subtrochanteric fracture. However, the pathologic etiology critically influences the surgical approach beyond standard trauma protocols.
- Enneking Staging System: Primarily for primary bone sarcomas, this system broadly categorizes tumor aggressiveness and extent. In the context of myeloma, which is a multifocal systemic disease, a localized lesion like this subtrochanteric fracture would fall under a high-grade (Stage III or similar) categorization for local disease, emphasizing the need for robust stabilization rather than attempts at local tumor ablation (which is not curative for myeloma).
Pre-Operative Planning:
- Oncologic Consultation: Immediate and close collaboration with the Hematology-Oncology team was paramount. This ensured prompt systemic management of the newly diagnosed myeloma (addressing hypercalcemia, renal failure, and anemia) and coordinated surgical timing within the overall treatment strategy. Bisphosphonates (e.g., zoledronic acid) or denosumab, critical for reducing skeletal-related events in myeloma, would be initiated post-operatively, or pre-operatively if time permitted.
- Medical Optimization: Aggressive intravenous hydration and calcitonin administration were initiated to manage the severe hypercalcemia. Anemia was corrected with packed red blood cell transfusions. Renal insufficiency was monitored closely.
-
Surgical Strategy:
- Implant Choice: Intramedullary nailing was the preferred method for stabilizing this subtrochanteric pathologic fracture. Its biomechanical advantages, including load-sharing properties and the ability to bypass the entire lytic lesion, were critical. A long intramedullary nail, extending well beyond the distal extent of the known lytic lesions, ideally spanning the entire diaphysis to the distal femur, was chosen to protect against future impending fractures distally.
- Biopsy: While the bone marrow biopsy and serum/urine studies had definitively diagnosed myeloma, an intra-operative open biopsy of the fracture site was still planned. This ensures local tissue confirmation of the lesion type and provides additional material for specific analyses (e.g., molecular diagnostics, culture if infection is suspected) relevant to guiding oncologic treatment.
Surgical Technique / Intervention
The primary objective was to achieve robust load-sharing internal fixation using an intramedullary nail, extending beyond the tumor proximally and distally, to enable immediate stability and facilitate early weight-bearing.
- Patient Positioning: The patient was positioned supine on a fracture table. The left leg was placed in traction, providing longitudinal alignment. Careful padding of all pressure points was performed. The contralateral limb was abducted and flexed to allow unrestricted fluoroscopic imaging access.
- Anesthesia: General endotracheal anesthesia was administered.
-
Surgical Approach:
- A standard lateral approach for antegrade femoral nailing was utilized. A longitudinal incision, approximately 5-7 cm in length, was made just proximal to the tip of the greater trochanter, extending proximally.
- The gluteus medius fibers were bluntly split to expose the piriformis fossa and the tip of the greater trochanter.
- Biopsy: Prior to reaming, an open biopsy of the abnormal, lytic bone at the fracture site was performed. Tissue samples were obtained using a curette and sent for histopathological analysis, flow cytometry, and microbiology. This confirmed the local presence of myeloma.
-
Reduction Techniques:
- Closed reduction was primarily attempted using traction and internal rotation via the fracture table.
- Fluoroscopic guidance was used to confirm satisfactory alignment in both AP and lateral views. Due to the significant comminution and inherently poor bone quality associated with myeloma, extreme caution and gentle manipulation were paramount to avoid iatrogenic fragmentation or propagation of the fracture.
- In cases where closed reduction is challenging, a limited open reduction with reduction clamps or cerclage wires may be considered. However, in this patient, acceptable alignment was achieved with traction.
-
Intramedullary Nailing:
- Entry Point: The entry portal was created at the tip of the greater trochanter, positioned slightly medial to the center in the AP view and slightly posterior in the lateral view, to ensure optimal nail trajectory and prevent varus malalignment.
- Guide Wire Insertion: A curved guide wire was carefully advanced across the comminuted fracture site and into the distal femoral canal. Navigating through the lytic bone required meticulous fluoroscopic guidance to prevent false passages or cortical breaches.
- Reaming: Progressive reaming was performed over the guide wire. The reamer size was typically chosen to be 1.5-2 mm larger than the intended nail diameter. Reaming through diseased bone was performed cautiously to minimize further cortical destruction.
- Nail Insertion: A long intramedullary nail, specifically a trochanteric-entry cephalomedullary nail (e.g., a Recon nail or similar design), was selected. The nail diameter was chosen to adequately fill the canal, maximizing purchase within the compromised bone. The nail length was critical: it extended well beyond the distal aspect of the lytic lesions, ideally into the distal metaphysis of the femur, to provide robust prophylactic stabilization of the entire diaphysis and prevent stress risers.
- Proximal Locking: Two lag screws were inserted into the femoral head and neck, providing crucial rotational and axial stability.
- Distal Locking: Static distal locking screws (typically two) were inserted using a targeting guide under fluoroscopic control to ensure length and rotational stability.
- Cement Augmentation: Given the severe lytic destruction and poor bone quality characteristic of multiple myeloma, polymethylmethacrylate (PMMA) cement augmentation was deemed highly beneficial. After nail insertion and proximal screw placement, low-viscosity PMMA cement was carefully injected through a small cannulated needle system into the femoral head around the lag screws under continuous fluoroscopic control. Further augmentation around the fracture site was considered, depending on the immediate stability achieved by the nail and the extent of cortical defects. This augmentation provides immediate mechanical stability and significantly enhances screw purchase in osteopenic bone.
-
- Wound Closure: Standard layered closure was performed, including irrigation, closure of subcutaneous tissues, and skin.
- Estimated Blood Loss: Approximately 300 mL.
- Fluoroscopy Time: Radiation exposure was minimized through careful planning and efficient execution.
Post-Operative Protocol & Rehabilitation
Post-operative management was meticulously planned to prioritize pain control, facilitate early mobilization, and seamlessly integrate with the systemic oncologic treatment.
-
Immediate Post-Operative Phase (Day 0-3):
- Pain Management: A multimodal analgesia regimen was initiated, including a continuous peripheral nerve block (e.g., fascia iliaca block), scheduled acetaminophen, cautious use of NSAIDs (titrated based on renal function), and opioid analgesics as needed.
- Deep Vein Thrombosis (DVT) Prophylaxis: Low Molecular Weight Heparin (LMWH) was commenced post-operatively and continued for 4-6 weeks, or as per institutional guidelines. Mechanical prophylaxis with sequential compression devices (SCDs) was also utilized.
- Early Mobilization: The patient was encouraged to sit out of bed on post-operative day 1 and commence toe-touch or partial weight-bearing (approximately 20-25 kg or 50-75 lbs) on the operated limb using a walker or crutches, as tolerated. The robust fixation provided by the long IMN with cement augmentation allowed for this aggressive early weight-bearing. Progression to full weight-bearing was guided by pain tolerance and radiographic stability.
- Wound Care: Daily dressing changes were performed, with close monitoring for signs of infection (erythema, swelling, discharge).
- Oncologic Management: Close coordination with the Hematology-Oncology team for initiation of systemic anti-myeloma therapy (e.g., proteasome inhibitors, immunomodulatory drugs, corticosteroids) once the patient was medically stable and beyond the immediate critical post-operative phase. Bisphosphonates or denosumab were commenced to reduce further skeletal-related events.
-
Rehabilitation Phase (Weeks 1-6):
- Physical Therapy: Gradual progression of weight-bearing was continued based on clinical symptoms and radiographic assessment. A comprehensive exercise program was implemented, including range of motion exercises for the hip, knee, and ankle, and progressive strengthening exercises for the core, hip abductors, quadriceps, and hamstrings. Gait training with appropriate assistive devices was a central component.
- Occupational Therapy: Assessment for adaptive equipment and home modifications was conducted to ensure safe return home and maximize independence in ADLs.
- Nutritional Support: Emphasis was placed on optimizing nutritional intake to support healing and combat cachexia, which is frequently associated with malignancy.
- Oncologic Follow-up: Regular follow-up appointments with the oncology team were scheduled for ongoing systemic treatment, monitoring of disease status (e.g., M-protein levels, FLCs), and management of treatment-related side effects.
-
Long-Term Follow-up (Months & Years):
- Orthopedic Follow-up: Serial radiographs (e.g., at 6 weeks, 3 months, 6 months, and 1 year post-op) were planned to assess fracture healing, implant integrity, and monitor for any progression of lytic lesions or signs of hardware failure or non-union.
- Oncologic Follow-up: Lifelong surveillance for myeloma progression, relapse, and long-term complications was scheduled with the oncology team.
- Adjuvant Radiation Therapy: Consideration for localized radiation therapy to the fracture site was discussed with the radiation oncologist, typically for persistent local pain, evidence of residual tumor burden, or high risk of local progression, usually after the surgical wound had completely healed and in conjunction with systemic therapy.
- Pain Clinic Referral: Referral to a chronic pain management clinic was considered if persistent or neuropathic pain developed.
- Bone Health Management: Continuation of bisphosphonates or denosumab, as directed by the oncology team, was crucial for ongoing prevention of skeletal-related events.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls:
- High Index of Suspicion: Always suspect a pathologic fracture in elderly patients presenting with low-energy trauma, atypical fracture patterns (e.g., extensive comminution in a seemingly trivial fall), or constitutional symptoms (fatigue, unexplained weight loss, generalized bone pain). The "fall" is often a symptom, not the cause.
- Recognize Myeloma Clues: The "CRAB" criteria (Hyper C alcemia, R enal failure, A nemia, B one lesions) are paramount. An elevated ESR, a normal or only slightly elevated alkaline phosphatase, and pure lytic, 'punched-out' lesions on X-ray should strongly raise suspicion for multiple myeloma.
-
Comprehensive Diagnostic Workup:
Beyond initial X-rays, a thorough workup for suspected pathologic fractures includes:
- Labs: FBC, RFTs, calcium, ESR, SPEP/UPEP with immunofixation, serum free light chain assay, beta-2 microglobulin.
- Imaging: Skeletal survey (for multifocal lesions), CT (for fracture morphology and cortical destruction), MRI (for marrow infiltration and spinal assessment), PET-CT (for metabolic activity and staging). A bone marrow biopsy is definitive.
- Multidisciplinary Team Approach: Optimal management of pathologic fractures from myeloma mandates close collaboration between Orthopedic Trauma, Hematology-Oncology, Radiation Oncology, Pathology, and Palliative Care specialists.
- Biopsy is Non-Negotiable: Always obtain a biopsy of the lesion to confirm the diagnosis, even if myeloma is strongly suspected systemically. An intraoperative biopsy through the surgical approach is efficient.
- Load-Sharing Fixation: For long bone pathologic fractures, intramedullary nails are superior to plates due to their biomechanical advantages (load sharing, reduced stress shielding), lower hardware failure rates, and ability to bypass extensive lesions.
- Complete Bypass of Lesion: The chosen implant must extend well beyond the lytic lesion both proximally and distally to neutralize stress risers and prevent subsequent periprosthetic fractures. For the femur, a long IMN spanning the entire diaphysis is frequently indicated.
- Prophylactic Stabilization: Utilize the Mirels score to identify high-risk impending pathologic fractures. Prophylactic fixation can prevent devastating fractures, reduce pain, and improve patient quality of life.
- Cement Augmentation (PMMA): In cases of severe osteopenia, extensive lytic destruction, or concerns for screw pullout (especially with cephalomedullary nails), PMMA cement augmentation significantly enhances immediate mechanical stability and screw purchase.
- Early Mobilization: Stable fixation allows for early weight-bearing and mobilization, which is critical for elderly patients with malignancy to prevent complications of immobility and preserve functional independence.
Pitfalls:
- Misdiagnosing as Traumatic Fracture: Failing to recognize the signs of a pathologic fracture can lead to an incomplete workup, delayed diagnosis of the underlying malignancy, and potentially suboptimal management.
- Inadequate Biopsy Sample: Obtaining insufficient or non-diagnostic biopsy material can lead to diagnostic delays and inappropriate treatment choices. Ensure adequate tissue collection.
- Underestimating Bone Quality: Myeloma-affected bone is severely compromised. Aggressive reaming, mal-tapping, or forceful reduction maneuvers can result in iatrogenic fractures, further fragmentation, or hardware failure.
- Insufficient Implant Length: Using an IMN that terminates too close to the lesion or fails to bypass other areas of cortical involvement creates stress risers and significantly increases the risk of subsequent fracture adjacent to the implant ends.
- Delaying Oncologic Treatment: Prolonged orthopedic recovery or delayed surgical clearance that significantly postpones systemic oncologic treatment can negatively impact the patient's overall prognosis. Meticulous pre-operative optimization and efficient surgical care are vital.
- Ignoring Systemic Complications: Failure to proactively manage hypercalcemia, renal insufficiency, or anemia pre- and post-operatively can lead to severe morbidity and mortality in these fragile patients.
- Inadequate Pain Control: Poorly managed pain significantly hinders rehabilitation efforts and patient compliance. A robust, multimodal analgesia strategy is crucial.
- Lack of Prophylaxis: Neglecting DVT prophylaxis in a patient with malignancy undergoing major orthopedic surgery carries a high risk of life-threatening thromboembolic events.
- Overlooking Other Lesions: Focusing solely on the fractured bone and missing other significant lytic lesions (e.g., in the spine, pelvis, or contralateral long bones) that might require prophylactic stabilization, radiation, or contribute to other symptoms.
- Suboptimal Implant Choice: Using a plate for a comminuted subtrochanteric pathologic fracture often results in higher failure rates compared to a properly executed IMN. Similarly, neglecting cement augmentation when indicated in poor bone quality is a significant pitfall.

