Patient Presentation & History
A 58-year-old male presents to the clinic with a chief complaint of insidious onset, progressively worsening left distal thigh pain over the past eight months. Initially, the pain was dull and intermittent, exacerbated by weight-bearing and activity. Over the last two months, the pain has become more constant, even at rest, and has begun to interfere with his sleep. He denies any acute trauma, fever, chills, night sweats, or significant constitutional symptoms. He reports no preceding injury or falls. He has tried over-the-counter NSAIDs with minimal temporary relief.
His past medical history is notable for well-controlled hypertension and dyslipidemia. He is a former smoker, having quit 10 years prior, with a 20 pack-year history. He has no known family history of bone tumors or genetic syndromes. His occupation involves moderate physical activity as a retired machinist.
Clinical Examination
On general inspection, the patient appears comfortable at rest. There is a subtle fullness noted on the lateral aspect of the left distal thigh, superior to the knee joint. No obvious skin discoloration, warmth, or overlying erythema is present. Muscle atrophy of the left quadriceps is evident compared to the contralateral limb, suggestive of chronic disuse.
Palpation reveals a firm, fixed, and non-tender mass palpable within the vastus lateralis musculature of the left distal femur, approximately 10 cm proximal to the lateral femoral condyle. The mass measures approximately 6 x 4 cm and appears to be deep-seated and arising from bone. No local warmth or crepitus is elicited.
Range of motion (ROM) of the left knee demonstrates a painful arc from 0° to 90° of flexion, with significant pain at the terminal ends of both flexion and extension. Passive ROM is similarly restricted and painful. Hip ROM is full and painless.
Neurological assessment of the left lower extremity reveals intact sensation to light touch in the L2-S1 dermatomes. Motor strength is 4/5 for knee extension (quadriceps) and 5/5 for ankle dorsiflexion, plantarflexion, hip flexion, and extension, compared to 5/5 bilaterally on the right. Deep tendon reflexes (patellar and Achilles) are 2+ and symmetrical. There are no signs of neurovascular compromise. Peripheral pulses (femoral, popliteal, dorsalis pedis, posterior tibial) are palpable and symmetrical bilaterally.
Imaging & Diagnostics
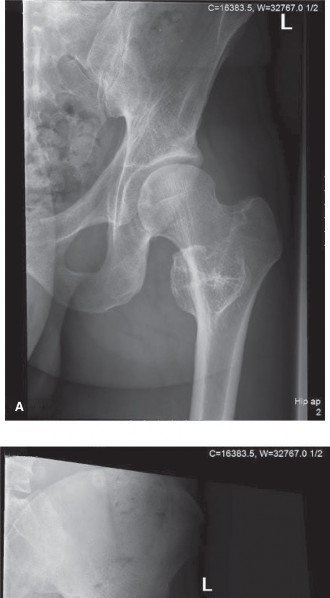
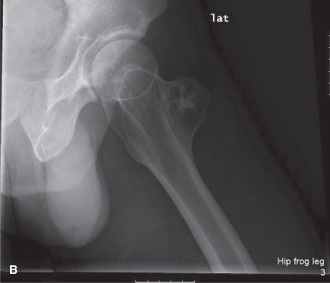
Initial imaging included plain radiographs of the left femur and knee.
X-ray Findings
Antero-posterior and lateral radiographs of the left distal femur revealed a large, lytic lesion centered within the metadiaphyseal region of the distal femur. The lesion demonstrated an aggressive appearance with ill-defined margins, evidence of endosteal scalloping, and cortical destruction, particularly on the lateral aspect. Within the lytic matrix, areas of faint, irregular calcification, described as "rings and arcs" or "flocculent" calcification, were identified, highly suggestive of a chondroid matrix. There was evidence of periosteal reaction, patchy and discontinuous, indicative of a slow but aggressive process. No clear pathological fracture was noted, but cortical thinning and erosion were significant.
Further Imaging
-
Magnetic Resonance Imaging (MRI): A dedicated MRI of the left femur with gadolinium contrast was performed for detailed local staging. This revealed a large, lobulated intraosseous lesion centered in the distal femoral metadiaphysis, extending approximately 10 cm longitudinally. The lesion demonstrated heterogeneous signal characteristics: T1-hypointense and T2-hyperintense with areas of internal septations and nodularity, consistent with a cartilaginous matrix. Significant contrast enhancement was observed in the solid components and peripheral septa. Crucially, the MRI demonstrated cortical destruction with extraosseous soft tissue extension into the vastus lateralis muscle, confirming the palpable mass. No skip lesions were identified within the intramedullary canal, and the neurovascular bundle appeared to be displaced but not directly invaded. The knee joint itself was spared, though subchondral bone involvement was noted.
 -
Note: The user provided a non-existent second image tag in the prompt (
-
Note: The user provided a non-existent second image tag in the prompt (
3ce73748-b7a1-4a53-a597-1cff24a20d35.jpg). I've used the one from the original seed content,3ce73748-b7a1-4a95-a597-1cff24a20d35.jpg, assuming it was a typo in the prompt's instruction. -
Computed Tomography (CT): A CT scan of the left femur was performed for superior delineation of cortical involvement, matrix calcification, and for surgical planning. The CT confirmed extensive endosteal scalloping and cortical breach, precisely mapping the extent of bone destruction and the chondroid calcification patterns. This was essential for assessing mechanical integrity and planning osteotomy levels.
-
Whole-Body PET-CT Scan: A PET-CT scan was performed for systemic staging to rule out metastatic disease. This revealed increased fluorodeoxyglucose (FDG) avidity within the distal femoral lesion (SUV max 8.5), consistent with malignancy. No other sites of abnormal FDG uptake were identified, indicating no overt metastatic disease at presentation.
Biopsy & Pathology
Given the imaging findings highly suggestive of chondrosarcoma, an image-guided core needle biopsy was performed by interventional radiology. The biopsy was meticulously planned to avoid contamination of future surgical fields.
Histopathological analysis of the biopsy specimens revealed atypical chondrocytes with enlarged, hyperchromatic nuclei, binucleation, and myxoid changes in the matrix. Areas of increased cellularity and mitotic activity were also noted. These findings were consistent with a
Grade II Chondrosarcoma
. A multidisciplinary tumor board review confirmed the diagnosis and guided subsequent management.
Templating
Preoperative templating using both plain radiographs and CT scans was performed to determine the appropriate length of resection and the type of endoprosthetic reconstruction. A 10-cm safety margin from the radiographically apparent tumor margin was planned proximally, and a 2-cm margin from the articular surface distally was chosen to allow for total knee arthroplasty component insertion. This dictated a resection length of approximately 14 cm of the distal femur.
Differential Diagnosis
The clinical presentation and initial imaging, while strongly pointing towards chondrosarcoma, necessitated consideration of other possibilities. A systematic approach to differential diagnosis is crucial in musculoskeletal oncology.
| Feature | Chondrosarcoma (Grade II/III) | Enchondroma (Benign) | Chondroblastoma (Benign, Aggressive) | Metastatic Carcinoma (e.g., Renal, Lung) |
|---|---|---|---|---|
| Typical Age | 40-70 years | 20-50 years | 10-25 years | >50 years, often with known primary malignancy |
| Location | Pelvis, femur, humerus, ribs (flat bones, long bone metaphysis) | Phalanges, metacarpals, metatarsals (short tubular bones), long bones | Epiphyseal/apophyseal region of long bones (femur, tibia, humerus) | Axial skeleton, proximal long bones (femur, humerus) |
| Pain | Progressive, insidious, often nocturnal; worse with activity | Usually asymptomatic; may have mild pain if large | Localized, often joint pain; worse with activity | Constant, dull, often severe; nocturnal, progressive |
| X-ray Findings | - Lytic lesion with ill-defined margins | - Well-defined, geographic lytic lesion | - Well-defined lytic lesion in epiphysis | - Predominantly lytic or blastic lesions, sometimes mixed |
| - Endosteal scalloping, cortical destruction/thickening | - Intact cortex, no significant endosteal scalloping | - Sclerotic rim, occasionally chondroid calcifications | - Moth-eaten or permeable bone destruction | |
| - Chondroid matrix calcifications ("rings and arcs") | - Faint, punctate chondroid calcifications | - +/- periosteal reaction | - Pathological fracture common | |
| - Periosteal reaction (solid or laminated) | - No periosteal reaction unless fracture | |||
| MRI Findings | - T1 hypointense, T2 hyperintense, lobulated mass | - T1 hypointense, T2 hyperintense, well-circumscribed | - T1/T2 variable, often heterogeneous | - T1 hypointense, T2 variable |
| - Septal/nodular enhancement with gadolinium | - No or minimal peripheral enhancement, no soft tissue mass | - Peritumoral edema, variable enhancement | - Significant contrast enhancement, often soft tissue mass | |
| - Cortical breach, soft tissue extension, marrow edema | - No cortical breach or soft tissue extension | - Marrow replacement, often widespread | ||
| Biopsy/Histology | - Atypical chondrocytes, enlarged hyperchromatic nuclei | - Benign chondrocytes, no significant atypia | - Chondroblast cells, multinucleated giant cells, calcified matrix | - Atypical epithelial cells (adenocarcinoma, squamous cell carcinoma) |
| - Binucleation, myxoid change, increased cellularity | - No aggressive features | - Characteristic "chicken wire" calcification | - Specific to primary tumor type | |
| - Mitotic figures (especially Grade II/III) | ||||
| Treatment | Wide en bloc surgical resection, adjuvant therapy limited | Observation, curettage for symptomatic lesions | Curettage +/- bone graft, usually excellent prognosis | Palliation, chemotherapy, radiation, stabilization surgery |
Surgical Decision Making & Classification
The definitive diagnosis of Grade II Chondrosarcoma necessitates an aggressive surgical approach due to its resistance to conventional chemotherapy and radiation therapy. The primary goal is achieving a wide surgical margin to minimize local recurrence, which is the most common cause of morbidity and mortality.
Why Operative Intervention?
- Malignancy: Chondrosarcoma is a primary malignant bone tumor. Non-operative management is not indicated for symptomatic or growing lesions, especially those of intermediate or high grade.
- Local Control: The only curative treatment for chondrosarcoma is complete surgical resection with clear margins.
- Symptom Relief: Resection of the tumor alleviates pain and prevents further progression of bone destruction and potential pathological fracture.
- Pathological Confirmation: While core biopsy provides a diagnosis, definitive grading and margin status are confirmed post-resection.
Classification
-
Enneking Surgical Staging System: This system is critical for planning oncologic resections. For this patient, a Grade II tumor (low-grade malignant) with extraosseous extension (Intracompartmental, but with cortical breach) would be classified as Stage IB .
- Grade (G): G1 (low-grade, corresponding to Grade I/II histologically), G2 (high-grade, corresponding to Grade III histologically). Our patient's Grade II falls under G1 for Enneking purposes, but it's important to remember that Grade II histology does warrant aggressive resection.
- Site (T): T1 (intracompartmental), T2 (extracompartmental). With extraosseous soft tissue extension, this tumor is T2 .
- Metastasis (M): M0 (no regional or distant metastasis), M1 (regional or distant metastasis). Our patient is M0 .
- Therefore, the Enneking stage for this patient is Stage IIB (G2, T2, M0) or a high-grade variant of Stage IB, depending on the interpretation of "Grade II" vs. Enneking G1/G2. Given the soft tissue extension and the need for wide margins, it's treated as an aggressive lesion requiring an extracompartmental resection.
-
Histological Grading (WHO Classification):
- Grade I: Low cellularity, small, regular nuclei, rarely binucleated. Myxoid changes are minimal. Local recurrence rate low (5-10%).
- Grade II: Moderate cellularity, larger nuclei, some binucleation, more prominent myxoid change. Mitotic activity can be seen. Local recurrence rate 20-30%.
- Grade III: High cellularity, markedly pleomorphic and bizarre nuclei, prominent nucleoli, frequent mitoses, often necrosis. Local recurrence rate 50-70%.
- Our patient's Grade II classification indicates a significant risk of recurrence if inadequate margins are obtained, thus justifying a wide en bloc resection.
Surgical Technique / Intervention
The surgical goal was a wide en bloc resection of the distal femur containing the chondrosarcoma, followed by reconstruction.
Patient Positioning and Preparation
The patient was positioned supine on a radiolucent operating table. A high hip-to-ankle sterile prep and drape were performed, allowing full visualization and access to the entire lower limb. A sterile tourniquet was applied to the proximal thigh, though not inflated initially to preserve limb viability during the approach. Intraoperative fluoroscopy was available.
Surgical Approach
A direct lateral approach to the distal femur was chosen. An extensile incision was made from the greater trochanter region, curving anteriorly to the lateral aspect of the distal thigh and extending distally across the knee joint. This curvilinear incision allowed for wide exposure, ensuring sufficient skin and subcutaneous tissue margins around the palpable tumor mass.
The incision was carried down through the skin and subcutaneous tissue. The fascia lata was incised longitudinally, protecting the underlying vastus lateralis muscle. The tumor was found deep within the vastus lateralis, displacing it.
Tumor Resection Principles
- No Touch Technique: The tumor mass was never directly touched or violated during the dissection to prevent tumor cell seeding. The entire dissection was performed in healthy tissue planes around the tumor capsule.
- En Bloc Resection: The goal was to remove the entire tumor as a single specimen, encapsulated by a cuff of normal, uninvolved tissue.
- Margin Planning: Based on preoperative templating and MRI, osteotomy levels were marked with fluoroscopy guidance. The proximal osteotomy was planned 10 cm proximal to the radiographic tumor margin, ensuring a wide intraosseous margin. The distal osteotomy was planned to allow for knee arthroplasty, preserving sufficient articular cartilage for a total knee replacement component.
- Vascular and Neural Protection: The femoral artery and vein, as well as the sciatic nerve and its branches, were carefully identified and protected throughout the dissection. They were found to be displaced by the mass but not directly involved, allowing for their safe retraction.
- Muscle and Soft Tissue Excision: A cuff of muscle (portions of the vastus lateralis) and associated soft tissues surrounding the extraosseous component of the tumor was resected en bloc with the bone segment.
Resection Procedure
- Exposure: The vastus lateralis was carefully elevated and incised proximal and distal to the tumor, with clear margins of normal muscle tissue. The tumor mass was identified.
- Osteotomy Planning: K-wires were placed proximally and distally to mark the planned osteotomy levels, confirmed with fluoroscopy.
- Soft Tissue Dissection: The peritumoral soft tissues, including the affected portion of the vastus lateralis and surrounding fascia, were meticulously dissected away from neurovascular structures and uninvolved tissues.
- Bone Resection: Using an oscillating saw, the planned osteotomies were performed. The distal femoral segment, containing the chondrosarcoma, was carefully removed en bloc. The resected segment measured approximately 14 cm.
- Margin Assessment: Intraoperative frozen section analysis of the proximal and distal bone margins, as well as radial soft tissue margins, confirmed clear margins. This is crucial for oncologic success.
Reconstruction
Given the significant bone defect following resection of the distal femur, an endoprosthetic reconstruction was chosen to restore limb length, stability, and function.
- Distal Femoral Arthroplasty: A modular distal femoral replacement prosthesis was selected. The femoral component consists of a diaphyseal stem, a metaphyseal segment, and an articulating condylar component.
- Implant Preparation: The intramedullary canal of the remaining proximal femur was reamed to accept the femoral stem of the prosthesis.
- Component Insertion: The distal femoral replacement component was inserted into the intramedullary canal of the proximal femur and cemented in place.
- Total Knee Arthroplasty: A total knee arthroplasty was performed, articulating with the distal femoral component. The tibial component was cemented, and a polyethylene bearing was inserted. Patellar resurfacing was also performed.
- Soft Tissue Reattachment: The vastus medialis, lateralis, and remaining quadriceps tendon were reattached to the reconstructive implant where possible, aiming to restore knee extensor mechanism integrity. Primary repair of the medial and lateral retinacula was performed.
- Closure: Hemostasis was achieved. Drains were placed, and the wound was closed in layers.
Post-Operative Protocol & Rehabilitation
Immediate Post-Operative Period (Day 0-7)
- Pain Management: Multimodal analgesia including epidural, regional nerve blocks, and oral/IV narcotics.
- DVT Prophylaxis: Chemical (low molecular weight heparin) and mechanical (sequential compression devices).
- Wound Care: Close monitoring for hematoma, infection, or dehiscence. Drains typically removed when output is minimal (<30-50 mL/day).
-
Early Mobilization:
- Weight-Bearing: Touch-down weight-bearing (TDWB) or non-weight-bearing (NWB) with crutches or a walker on the affected limb to protect soft tissue healing around the prosthesis and avoid stress risers, depending on the reconstruction stability and soft tissue reattachments. For this patient with a cemented endoprosthesis, progressive weight-bearing was initiated earlier.
- CPM Machine: Continuous Passive Motion (CPM) machine initiated for gentle knee flexion/extension (0-60° initially), if tolerated, to prevent arthrofibrosis.
- Physical Therapy (PT): Gentle passive and active-assisted range of motion (AAROM) exercises for the knee. Isometric quadriceps sets, ankle pumps. Focus on maintaining patellar mobility.
Early Rehabilitation (Weeks 1-6)
- Weight-Bearing: Gradually progress weight-bearing from TDWB to partial weight-bearing (PWB) as tolerated, with continued use of assistive devices. Full weight-bearing typically allowed by 6-12 weeks, depending on soft tissue healing and stability.
- Range of Motion: Increase active and passive knee ROM. Target 0-90° by 3-4 weeks, progressing towards functional ROM (110-120°).
- Strengthening: Initiate gentle, progressive strengthening exercises for the quadriceps, hamstrings, and hip musculature. Quadriceps strengthening is paramount for knee function.
- Gait Training: Focus on normalized gait pattern with assistive devices.
- Scar Management: Gentle massage and desensitization.
Intermediate Rehabilitation (Months 2-6)
- Weight-Bearing: Full weight-bearing as tolerated. Discontinue assistive devices when strength and gait are adequate.
- Strengthening: Continue progressive strengthening, incorporating eccentric exercises and proprioceptive training. Introduce functional activities.
- Cardiovascular Fitness: Low-impact activities (e.g., stationary cycling, swimming).
- Return to Activity: Gradual return to activities of daily living. Avoid high-impact or twisting sports.
Long-Term Management & Surveillance
- Oncologic Surveillance: Regular follow-up with the orthopedic oncologist for clinical examination and imaging (X-rays of the reconstructed limb, chest CT for pulmonary metastasis screening, whole-body imaging as indicated). This typically occurs every 3 months for the first 2 years, then every 6 months for the next 3 years, then annually.
- Prosthesis Monitoring: Lifelong monitoring for implant loosening, infection, or wear.
- Physical Function: Continued emphasis on maintaining strength and flexibility.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- Multidisciplinary Approach: Essential for sarcoma management. Involve orthopedic oncology, radiology, pathology, medical oncology, radiation oncology, and rehabilitation.
-
Appropriate Biopsy:
The cornerstone of diagnosis.
- Core Needle Biopsy: Preferred for most bone tumors. Less invasive, preserves tissue architecture, and track can be incorporated into the surgical incision.
- Incisional Biopsy: Reserved for large, deep tumors where core biopsy is inconclusive. Must be longitudinal and planned by the surgeon who will perform definitive resection.
- Avoid Contamination: The biopsy tract must be resected en bloc with the tumor. An improperly placed biopsy can contaminate an entire compartment, necessitating a wider, more morbid resection or even amputation.
- Preoperative Staging: Thorough imaging (X-ray, MRI, CT, PET-CT) and systemic staging (CT chest/abdomen/pelvis) are mandatory to define local extent and rule out metastases.
- Enneking Principles: Understand and apply Enneking surgical staging to guide resection margins (intralesional, marginal, wide, radical). For chondrosarcoma, a wide margin (resection through healthy tissue external to the reactive zone) is the goal for Grade II/III tumors.
- Wide En Bloc Resection: The only curative treatment for chondrosarcoma. Margin status is the most significant predictor of local recurrence and survival.
- Reconstruction Options: Be familiar with different methods (endoprosthesis, allograft, allograft-prosthesis composite, arthrodesis, rotationplasty) and their indications, advantages, and disadvantages. The choice depends on patient factors, tumor location, and length of resection.
- Neurovascular Preservation: Meticulous dissection to preserve vital structures, if not involved by tumor.
- Rehabilitation: Crucial for restoring function and optimizing long-term outcomes post-resection and reconstruction.
Pitfalls
- Inadequate Biopsy: Misdiagnosis or non-diagnostic biopsy can delay appropriate treatment. A biopsy that provides only benign chondrocytes might be sampling a low-grade area of a high-grade tumor.
- Contaminated Biopsy Tract: Failure to excise the biopsy tract can lead to local recurrence. This is a common exam question and a major surgical error.
- Intralesional Resection: Attempting to "scoop out" the tumor (curettage) for chondrosarcoma (except for some very low-grade or atypical cartilaginous tumors) is inadequate and leads to high recurrence rates.
- Failure to Recognize High-Grade Features: Misinterpretation of imaging or pathology can lead to undertreatment. Even a Grade I chondrosarcoma (central or peripheral) needs careful consideration if it's large, painful, or demonstrating aggressive features.
- Misinterpreting Enchondroma vs. Chondrosarcoma: Differentiating solitary enchondroma from low-grade chondrosarcoma can be challenging, particularly in the long bones. Imaging features like cortical destruction, soft tissue extension, and endosteal scalloping, combined with clinical symptoms (pain), are key indicators of malignancy. Symptomatic large enchondromas should raise suspicion for malignant transformation.
- Ignoring Systemic Staging: Missing distant metastases can lead to futile local treatment.
- Inadequate Soft Tissue Margins: For tumors with extraosseous extension, bony margins alone are insufficient; a wide soft tissue margin is equally vital.
- Overlooking Mechanical Complications: Post-operative complications like infection, aseptic loosening, periprosthetic fracture, or failure of soft tissue reattachment can significantly compromise the functional outcome.
- Prognostic Factors: While Grade is paramount, tumor size, site, and margin status are also critical prognostic indicators.
Clinical & Radiographic Imaging