Patient Presentation & History
A 38-year-old male presented with a 2-year history of insidious onset left knee pain, intermittent swelling, and mechanical symptoms including occasional catching and giving way. Initially, symptoms were mild and attributed to sports-related overuse; however, over the past 6 months, the pain has become more constant, the effusion more persistent, and functional limitations have increased, particularly with squatting and ascending stairs. He reports no specific inciting trauma. He denies fever, chills, night sweats, or unintentional weight loss. He has no significant past medical history, no history of inflammatory arthritides in himself or his family, and no bleeding diatheses. Previous aspirations revealed serosanguinous fluid which was negative for crystals and infection. Non-steroidal anti-inflammatory drugs (NSAIDs) provided minimal temporary relief.
Clinical Examination
Left Knee
- Inspection: A noticeable, palpable joint effusion was present, described as boggy. There was no obvious erythema or warmth. Quadriceps atrophy was mild.
- Palpation: Diffuse tenderness was elicited along the medial and lateral joint lines, as well as over the suprapatellar pouch. Significant synovial thickening was palpable, particularly in the suprapatellar region and medial gutter. No discrete masses were immediately obvious on superficial palpation.
- Range of Motion (ROM): Active ROM was limited from 5° of flexion contracture to 100° of flexion (normal: 0-140°), with pain at the extremes. Passive ROM mirrored active motion, with a firm end-feel. Crepitus was noted throughout the arc of motion.
- Ligamentous Stability: Medial and lateral collateral ligaments were stable to valgus and varus stress at 0° and 30° flexion. Anterior and posterior drawer tests were negative. Lachman test was negative. Pivot shift was negative.
- Meniscal Tests: McMurray's test elicited diffuse pain but no specific click. Apley's grind test was suggestive of meniscal irritation.
- Patellofemoral Joint: Mild patellofemoral crepitus, but no instability or significant apprehension.
- Neurovascular Assessment: Distal pulses were palpable and symmetric. Sensation was intact throughout the dermatomes of the lower extremity. Motor strength was 5/5 in all major muscle groups.
General
- No other joint effusions or signs of systemic disease.
- Cardiovascular, pulmonary, and abdominal examinations were unremarkable.
Imaging & Diagnostics
Plain Radiographs
- Left Knee AP/Lateral/Oblique/Patellofemoral Views: Initial radiographs demonstrated a moderate joint effusion. Joint space appeared preserved. No obvious osteophytes or significant degenerative changes were noted. Mild, diffuse osteopenia of the distal femur and proximal tibia was observed. No acute fractures or periosteal reactions were evident. Subtle erosions of the medial femoral condyle and proximal tibia, although not overtly destructive, raised suspicion.
Magnetic Resonance Imaging (MRI)
-
Left Knee MRI (T1, T2, PD, GRE sequences, with and without Gadolinium contrast):
The MRI was crucial for diagnosis.
- Findings: Demonstrated diffuse, hypertrophic synovitis involving all compartments of the knee joint, including the suprapatellar pouch, medial and lateral gutters, and intercondylar notch. Multiple nodular and villous synovial proliferations were clearly visible.
- Hemosiderin Deposition: Extensive areas of low signal intensity on T1- and T2-weighted sequences, with prominent "blooming artifact" on gradient-recalled echo (GRE) sequences, characteristic of hemosiderin deposition. This finding is pathognomonic for PVNS.
- Erosions: Articular cartilage appeared thinned in several areas, with subchondral bone erosions identified on the medial femoral condyle and posterior aspect of the tibial plateau. No significant bone marrow edema was noted in relation to the erosions.
- Effusion: A moderate to large joint effusion was present, often mixed with low signal intensity debris consistent with hemorrhage.
- Post-Contrast Imaging: Diffuse, avid enhancement of the proliferative synovium was observed.
- Menisci and cruciate ligaments appeared intact, although visualization was somewhat obscured by the extensive synovitis.
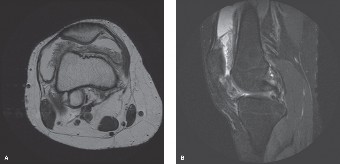
Figure 1: Representative MRI axial image (T2-weighted with fat saturation) demonstrating diffuse synovial proliferation with characteristic low signal intensity areas indicative of hemosiderin deposition (blooming artifact often more pronounced on GRE sequences). This appearance is highly suggestive of pigmented villonodular synovitis.
Synovial Fluid Analysis
- Aspiration: Grossly, the fluid was serosanguinous, dark brown, and viscous.
- Microscopic: WBC count was 2,500 cells/µL with a differential of 60% mononuclear cells and 40% polymorphonuclear cells. Glucose was 80 mg/dL (serum glucose 100 mg/dL). Protein was elevated. Gram stain and cultures were negative. No crystals were identified. The presence of hemosiderin-laden macrophages ("heart failure cells") was noted.
Biopsy
- Arthroscopic Synovial Biopsy: Histopathological examination revealed a characteristic pattern of synovial proliferation composed of villous and nodular architecture. Abundant hemosiderin deposition was identified within the stromal cells and macrophages. Multinucleated giant cells, foamy histiocytes, and a chronic inflammatory infiltrate were also present. Mitotic activity was low. These features confirmed the diagnosis of diffuse pigmented villonodular synovitis (PVNS).
Differential Diagnosis
The clinical presentation of chronic monoarticular effusion and pain can be broad. Differentiating PVNS from other conditions relies heavily on imaging, particularly MRI, and definitive histology.
| Feature | Pigmented Villonodular Synovitis (PVNS) | Synovial Chondromatosis (SC) | Rheumatoid Arthritis (RA) | Synovial Sarcoma (SS) |
|---|---|---|---|---|
| Pathology | Benign, locally aggressive proliferation of synovium with hemosiderin, giant cells, foamy macrophages. | Metaplasia of synovial tissue into cartilage, leading to formation of cartilaginous loose bodies. | Chronic systemic autoimmune inflammatory disease of synovial lining, pannus formation. | High-grade malignant tumor of uncertain differentiation, often biphasic (spindle cells + epithelial cells). |
| Typical Age | 20-50 years | 30-50 years | 30-60 years (but can affect any age) | 15-40 years (most common soft tissue sarcoma in young adults) |
| Clinical Pres. | Insidious pain, swelling, recurrent hemarthrosis, mechanical symptoms, limited ROM. Monoarticular. | Pain, swelling, mechanical symptoms (locking, catching), palpable loose bodies. Monoarticular. | Polyarticular, symmetrical joint involvement, morning stiffness, systemic symptoms (fatigue, fever). | Slowly enlarging, often painless mass. Deep-seated, may cause local pain/swelling if compressing structures. |
| X-ray Findings | Often normal early. Later: Effusion, diffuse osteopenia, periarticular erosions (distinct from osteophytes). | Multiple intra-articular calcifications/ossifications (loose bodies). Joint space preservation until late. | Periarticular osteopenia, marginal erosions, joint space narrowing, subluxation, soft tissue swelling. | Soft tissue mass, often with punctate or amorphous calcifications (30%). May cause pressure erosions on bone. |
| MRI Findings | Key: Diffuse or nodular synovial proliferation with low signal intensity on T1/T2/GRE ("blooming artifact") due to hemosiderin. Avid enhancement. Erosions. | Multiple iso/hypointense (T1) / hyperintense (T2) loose bodies. May show mineralization. Diffuse synovial hypertrophy less prominent than PVNS. | Diffuse synovial hypertrophy and enhancement. Marrow edema. Joint effusion. Characteristic bone erosions, often marginal. | Heterogeneous mass with variable signal intensity. May contain areas of necrosis, hemorrhage, or cystic degeneration. Variable enhancement. Often large at presentation. |
| Synovial Fluid | Serosanguinous/brown, hemosiderin-laden macrophages, moderate WBCs (2-10k). | Non-inflammatory, may contain small cartilage fragments. | Inflammatory, high WBCs (5-50k), often cloudy. Negative for crystals. | Non-specific if effusion present. Rarely specific findings. |
| Treatment | Complete surgical synovectomy (open or arthroscopic), adjuvant radiation for diffuse/recurrent disease. CSF1R inhibitors for unresectable. | Arthroscopic removal of loose bodies and synovectomy. | Medical management (DMARDs, biologics), physical therapy. Surgery for joint reconstruction/replacement. | Wide surgical excision with adequate margins, often combined with adjuvant radiotherapy and/or chemotherapy. |
| Recurrence Rate | High (20-50% for diffuse disease even after complete synovectomy). | Low after complete removal. | Chronic, fluctuating course. | High risk of local recurrence and metastasis (lung, lymph nodes). |
Surgical Decision Making & Classification
Given the confirmed diagnosis of diffuse PVNS of the knee, surgical intervention was imperative. PVNS is a locally aggressive tumor, and while benign, it has a high propensity for recurrence if not adequately excised. Non-operative management, such as aspiration, NSAIDs, or intra-articular steroids, provides only temporary symptomatic relief and does not address the underlying proliferative process, potentially leading to progressive joint destruction.
Classification of PVNS
- Localized (Nodular) PVNS: Characterized by a discrete, solitary nodule or a few nodules within the joint or tendon sheath. Often amenable to arthroscopic excision with lower recurrence rates.
- Diffuse PVNS: Widespread involvement of the entire synovial lining of the joint. This form carries a significantly higher risk of recurrence after surgical resection. The patient's MRI and biopsy findings were consistent with diffuse PVNS.
Surgical Rationale
For diffuse PVNS of the knee, the goal is complete synovectomy. The decision for an open versus arthroscopic approach, or a combined approach, depends on the extent of the disease, joint accessibility, and surgeon experience. Given the diffuse nature of the disease affecting multiple compartments including potential erosions, a thorough and complete synovectomy was deemed critical. While arthroscopy offers benefits of reduced morbidity, achieving a truly complete synovectomy in diffuse PVNS via arthroscopy alone, especially in posterior compartments and around cruciates, can be challenging.
Therefore, the decision was made for an arthroscopically-assisted open synovectomy to optimize complete excision, addressing both anterior and posterior compartments meticulously. This approach allows for preliminary arthroscopic assessment and debulking, followed by direct visualization and complete resection of the diseased synovium via an open approach where access is difficult.
Adjuvant Considerations
In cases of diffuse PVNS, especially after incomplete resections or in recurrent disease, adjuvant therapies are sometimes considered:
*
External Beam Radiotherapy (EBRT):
Can be used post-synovectomy to reduce recurrence, particularly in diffuse disease or when complete resection is difficult to achieve.
*
Intra-articular Radiotherapy (e.g., Yttrium-90):
Has been used in some centers, though efficacy and complications are debated.
*
Colony-Stimulating Factor 1 Receptor (CSF1R) Inhibitors (e.g., Pexidartinib):
A newer systemic targeted therapy approved for symptomatic tenosynovial giant cell tumor (which includes PVNS) that is associated with severe morbidity or functional limitations and is not amenable to improvement with surgery. This is typically reserved for unresectable or recurrent, problematic cases.
For this patient, given it was the primary surgical intervention, adjuvant therapy was not planned initially but would be considered in case of recurrence.
Surgical Technique / Intervention
The patient underwent an arthroscopically-assisted open synovectomy of the left knee.
Pre-operative Planning
- Review of MRI to map the extent of synovial involvement, especially posterior compartment and areas of erosion.
- Pre-operative templating for potential reconstructive options if joint destruction was more severe (not required in this case).
- Informed consent detailing potential for recurrence, stiffness, infection, neurovascular injury.
Patient Positioning and Preparation
- Patient positioned supine on the operating table.
- Pneumatic tourniquet applied to the proximal thigh.
- Standard sterile prep and drape of the left lower extremity.
- Leg holder used to allow full flexion and extension.
Arthroscopic Phase
- Portals: Standard anteromedial and anterolateral portals were established.
- Initial Assessment: Initial diagnostic arthroscopy confirmed diffuse, hypertrophic, pigmented synovial proliferation throughout the anterior compartment, suprapatellar pouch, and intercondylar notch. Multiple frond-like villi and nodular lesions were observed, many with dark brown/black hemosiderin staining.
- Debulking: A motorized shaver (4.5 mm aggressive blade) was utilized to debulk the accessible anterior and suprapatellar synovium. Meticulous care was taken to avoid iatrogenic cartilage damage. The arthroscope provided excellent illumination and magnification for visualization. This debulking reduced the overall volume and allowed for better visualization during the subsequent open phase.
- Intercondylar Notch Clearance: Careful debridement of synovium from the intercondylar notch, around the cruciate ligaments, was performed.
Open Synovectomy Phase (Anterior)
- Approach: Following arthroscopic debulking, a medial parapatellar arthrotomy was performed, extending from the vastus medialis obliquus origin distally to the medial aspect of the patellar tendon. The patella was everted laterally.
- Direct Visualization: This allowed for comprehensive direct visualization of the entire anterior compartment, including the medial and lateral gutters, suprapatellar pouch, and patellofemoral joint.
- Meticulous Synovectomy: Using a combination of scalpel, curved scissors, and electrocautery, a radical synovectomy was performed. All visibly diseased synovium, nodules, and villous proliferations were systematically excised. The synovium was dissected off the joint capsule, patella, and articular margins. Special attention was paid to the corners and recesses (e.g., medial plica area, fat pad margins) where lesions often persist.
- Erosion Management: Areas of subchondral erosion on the medial femoral condyle and tibial plateau were curetted thoroughly to remove any potentially embedded synovial tissue. The goal was to remove all macroscopic disease.
Open Synovectomy Phase (Posterior)
- Closure of Anterior Arthrotomy: The anterior arthrotomy was temporarily closed.
- Repositioning: The patient's leg was flexed, and the operating table adjusted to provide access to the posterior aspect of the knee. Alternatively, the patient can be flipped to prone, but this was avoided for efficiency.
-
Posteromedial and Posterolateral Approaches:
Separate small posteromedial and posterolateral incisions were made to access the posterior compartments.
- Posteromedial: Incision posterior to the medial collateral ligament, anterior to the semimembranosus. Care taken to protect the saphenous nerve and vein. The posterior capsule was incised to access the posteromedial compartment and remove synovium from the posterior aspect of the medial condyle and medial gastrocnemius head.
- Posterolateral: Incision posterior to the lateral collateral ligament, anterior to the biceps femoris tendon. Care taken to protect the common peroneal nerve. The posterior capsule was incised to access the posterolateral compartment, lateral condyle, popliteus tendon, and lateral gastrocnemius head.
- Posterior Synovectomy: A thorough synovectomy was performed in both posterior compartments. Particular attention was paid to the posterior capsule and around the posterior horns of the menisci and cruciates, where PVNS often extends.
Final Assessment and Closure
- Irrigation: The joint was copiously irrigated with saline to remove any remaining debris.
- Hemostasis: Meticulous hemostasis achieved with electrocautery to minimize post-operative bleeding, which can contribute to stiffness and recurrence.
- Capsular Closure: The posterior capsular incisions were closed. The medial parapatellar arthrotomy was repaired anatomically, often with absorbable sutures.
- Wound Closure: Subcutaneous layers and skin were closed in layers. A drain was typically inserted into the joint space and removed on post-operative day 1.
- Dressing: A sterile dressing and soft compression bandage were applied.
Post-Operative Protocol & Rehabilitation
The post-operative course aims to minimize stiffness, control pain, and prevent recurrence.
Immediate Post-Operative Period (Day 0-7)
- Pain Management: Multimodal analgesia including NSAIDs (if no contraindications), acetaminophen, and opioids as needed. Regional nerve blocks (e.g., adductor canal block) were utilized intra-operatively to aid immediate post-op pain control.
- Drain Management: Intra-articular drain typically removed within 24-48 hours once output significantly diminishes.
- Weight-Bearing: Full weight-bearing as tolerated with crutches for comfort and stability.
-
Range of Motion:
- Continuous Passive Motion (CPM) Machine: Initiated immediately post-op, typically for 4-6 hours daily, with gradual increase in range as tolerated (e.g., 0-90° initially, progressing to full). CPM helps prevent arthrofibrosis and adhesion formation, which are significant risks after extensive synovectomy.
- Active-Assisted and Passive ROM Exercises: Initiated by physical therapy, focusing on achieving full extension and progressing flexion.
- Cryotherapy: Application of ice packs or cryo-cuff to reduce swelling and pain.
- Wound Care: Daily dressing changes; monitor for signs of infection.
Early Rehabilitation (Weeks 1-6)
- Goals: Restore full range of motion, reduce swelling, initiate quadriceps and hamstring strengthening.
-
Physical Therapy:
Sessions 3-5 times per week.
- ROM: Continue aggressive ROM exercises to achieve and maintain full extension and progress flexion to functional levels (e.g., 120-130° by 6 weeks).
- Strengthening: Isometrics (quad sets, glute sets), straight leg raises, gentle closed-chain exercises (mini-squats, wall slides).
- Gait Training: Progress from crutches to single crutch, then cane, then independent ambulation.
- Edema Control: Compression bandages, elevation, cryotherapy.
- Activity Restrictions: Avoid high-impact activities, deep squats, or prolonged standing initially.
Intermediate Rehabilitation (Weeks 6-12)
- Goals: Restore strength, proprioception, and prepare for return to functional activities.
-
Physical Therapy:
- Progressive Strengthening: Open- and closed-chain exercises with increasing resistance (leg press, hamstring curls, calf raises). Incorporate balance and proprioception exercises (wobble board, single-leg stance).
- Cardiovascular Conditioning: Stationary cycling, elliptical, swimming.
- Functional Training: Agility drills, sport-specific movements if applicable.
- Activity: Gradual return to light recreational activities.
Long-Term Management & Monitoring
-
Follow-up:
Regular clinical follow-up for at least 5 years due to the high recurrence rate.
- Clinical assessment for pain, swelling, ROM.
- Periodic MRI scans (e.g., at 6 months, 1 year, then annually) to monitor for subtle signs of recurrence. The characteristic hemosiderin signal on MRI makes it an excellent surveillance tool.
- Patient Education: Crucial to educate the patient on the chronic nature of PVNS, the potential for recurrence, and the importance of adherence to follow-up.
- Recurrence: If recurrence is identified, re-evaluation for further surgical intervention or consideration of adjuvant therapies (e.g., radiotherapy, CSF1R inhibitors) is warranted.
Pearls & Pitfalls (Crucial for FRCS/Board Exams)
Pearls
- High Index of Suspicion: PVNS should be suspected in any young to middle-aged adult presenting with chronic, insidious, monoarticular joint pain and recurrent, usually serosanguinous or brown, effusions without clear trauma or systemic inflammatory signs. The knee is the most common joint involved, followed by the hip.
- MRI is Key: MRI is the diagnostic imaging modality of choice. The characteristic low signal intensity on T1- and T2-weighted images, and especially the "blooming artifact" on gradient-recalled echo (GRE) sequences, due to hemosiderin deposition, is highly specific for PVNS.
- Histological Confirmation: While MRI is highly suggestive, definitive diagnosis requires histopathological examination of synovial tissue, demonstrating villous or nodular synovial proliferation with hemosiderin-laden macrophages, multinucleated giant cells, and foamy histiocytes.
- Complete Synovectomy: The cornerstone of treatment for diffuse PVNS is radical, complete surgical synovectomy. Incomplete resection is the single biggest predictor of recurrence. This often necessitates a combined arthroscopic and open approach, particularly for the knee, to access all compartments (anterior, posterior, medial, lateral).
- Adjuvant Therapy Considerations: For diffuse or recurrent disease, especially in difficult-to-treat joints (e.g., hip) or if complete resection is challenging, adjuvant external beam radiotherapy can reduce recurrence rates. Newer systemic therapies like CSF1R inhibitors are emerging for unresectable or highly symptomatic recurrent cases.
- Arthrofibrosis Prevention: Early and aggressive post-operative range of motion (including CPM) is critical to prevent joint stiffness and arthrofibrosis, which are common complications after extensive synovectomy.
- Long-Term Follow-up: PVNS has a significant recurrence rate, even after seemingly complete synovectomy (20-50% for diffuse disease). Lifelong follow-up, often with periodic clinical examination and surveillance MRIs, is essential to detect recurrence early.
Pitfalls
- Misdiagnosis as Other Arthritides: Early PVNS can be misdiagnosed as meniscal pathology, osteoarthritis, or inflammatory arthritis due to non-specific symptoms and normal plain radiographs. Failure to pursue advanced imaging (MRI) is a critical pitfall.
- Inadequate Synovectomy: Relying solely on arthroscopy for diffuse PVNS can lead to incomplete resection, especially in posterior compartments or recesses. This significantly increases recurrence rates. An open or combined approach is often necessary for thorough debridement.
- Failure to Address Erosions: PVNS can cause subchondral bone erosions. These areas must be curetted to remove embedded synovial tissue, as leaving them can be a source of recurrence.
- Ignoring Adjuvant Options: In high-risk cases (e.g., diffuse, recurrent, difficult resection), failing to consider or discuss adjuvant radiotherapy can lead to preventable recurrences.
-
Complications of Synovectomy:
The procedure itself carries risks:
- Stiffness/Arthrofibrosis: High risk after extensive synovectomy, requiring aggressive post-op rehabilitation.
- Neurovascular Injury: Especially in the knee (peroneal nerve, popliteal vessels during posterior approach) and hip. Meticulous surgical technique is paramount.
- Infection: As with any joint surgery.
- Recurrent Hemarthrosis: Can occur post-operatively, leading to increased pain and stiffness. Careful hemostasis and drain placement are important.
- Overlooking Localized vs. Diffuse Forms: Treating localized PVNS with an unnecessarily extensive open approach or, conversely, attempting to treat diffuse PVNS with a limited arthroscopic approach are both pitfalls. Proper classification dictates the surgical strategy.
- Delayed Diagnosis: Prolonged delay in diagnosis can lead to extensive joint destruction, requiring more complex interventions, including arthroplasty, and increasing the risk of recurrence.
