Optimizing the Management of Difficult Metastatic Lesions
Introduction & Epidemiology
Metastatic bone disease represents a significant challenge in orthopedic oncology, impacting patient quality of life through pain, pathological fractures, functional impairment, and neurological deficits. With advancements in systemic cancer therapies leading to prolonged survival for patients with metastatic disease, the incidence of skeletal-related events (SREs) is increasing, necessitating a sophisticated, multidisciplinary approach to management. "Difficult metastatic lesions" are characterized by their location (e.g., spine, pelvis, periacetabular), extensive bone destruction, proximity to neurovascular structures, aggressive tumor biology, or the presence of established pathological fractures with significant displacement or comminution.
The primary goal of orthopedic intervention in metastatic disease is palliative: pain relief, preservation or restoration of function, and prevention or stabilization of pathological fractures. For patients with a longer life expectancy, robust reconstruction to facilitate early mobilization is paramount. The decision-making process is complex, integrating tumor biology, systemic disease burden, patient performance status, anticipated life expectancy, and specific lesion characteristics.
Epidemiologically, bone is the third most common site for metastasis, following the lung and liver. The most frequent primary tumors to metastasize to bone include breast (65-75%), prostate (60-70%), lung (30-40%), kidney (20-25%), and thyroid (10-15%) cancers, along with multiple myeloma. Melanoma and gastrointestinal malignancies also have a propensity for bone metastasis, though less commonly. The axial skeleton (spine, pelvis) is the most frequently affected site (60%), followed by the proximal femur (25%) and humerus (10%). Pathological fractures occur in 10-30% of patients with bone metastases, with the femur being the most common site. Predicting fracture risk is critical for prophylactic stabilization, with the Mirels score being a widely accepted tool.
Surgical Anatomy & Biomechanics
Successful management of difficult metastatic lesions hinges upon a thorough understanding of the affected region's surgical anatomy and biomechanical principles. Lesions often compromise critical load-bearing structures, altering normal stress distribution and predisposing to fracture.
Axial Skeleton
- Spine: Metastases most commonly involve the vertebral body, often sparing the posterior elements initially. However, progressive disease can lead to pedicle and lamina involvement, spinal canal compromise, and neurological deficit. The three-column model of Denis is crucial for assessing spinal stability. Biomechanical stability is significantly compromised by anterior column destruction. Surgical approaches must consider vital neurovascular structures, including the spinal cord, nerve roots, aorta, vena cava, and segmental vessels.
- Pelvis: The pelvis is a complex ring structure providing attachment for significant musculature and transferring weight from the axial to the appendicular skeleton. Metastases frequently affect the acetabulum, ilium, sacrum, and pubic rami. Periacetabular lesions, classified by Harrington or Enneking, are particularly challenging due to high mechanical stress, proximity to major neurovascular bundles (e.g., sciatic nerve, femoral vessels), and the intricate musculature of the hip. Extensive bone loss in this region can severely impair ambulation.
- Sacrum: Sacral metastases are often associated with intractable pain and can lead to sacral insufficiency fractures or neurological deficits (cauda equina syndrome). Surgical access is challenging due to the pelvis's posterior wall and the sacral nerve roots.
Appendicular Skeleton
-
Long Bones:
The femur (proximal, diaphyseal, distal), humerus (proximal, diaphyseal), and tibia are frequently affected.
- Proximal Femur: High weight-bearing stress, potential for hip joint involvement. Lesions often compromise the calcar femorale or subtrochanteric region.
- Diaphyseal Femur/Humerus: Cortical destruction reduces bending and torsional stiffness. Intramedullary nailing is often preferred for these sites.
- Periarticular Regions: Involvement near major joints (e.g., shoulder, knee) complicates reconstruction, often necessitating arthroplasty or custom endoprostheses.
-
Biomechanics of Fixation:
- Prophylactic Fixation: Indicated for impending fractures (Mirels score ≥ 7). Aims to restore normal stress distribution and prevent catastrophic failure.
- Fracture Fixation: Requires robust constructs to achieve immediate stability, allow early weight-bearing, and overcome the inherently poor healing potential of pathological bone. Cement augmentation (polymethylmethacrylate, PMMA) is frequently used to fill defects, provide immediate load-bearing support, and enhance screw purchase, particularly in osteoporotic or compromised bone. Tumor prostheses or custom implants may be required for large segmental resections or extensive joint involvement.
- Load Sharing vs. Load Bearing: In metastatic bone, fixation often needs to be load-bearing, as the compromised bone cannot reliably share load with the implant. This requires more robust implants and fixation principles than typical trauma cases.
Indications & Contraindications
The decision for operative versus non-operative management of difficult metastatic lesions requires a careful balance of patient factors, tumor characteristics, and systemic disease status. The primary goals of surgery are palliation of pain, restoration/preservation of function, prevention of impending pathological fractures, and stabilization of existing pathological fractures.
Indications for Operative Management:
- Impending Pathological Fracture: High Mirels score (≥ 7). This score considers lesion site, size, and type (lytic, blastic, mixed) to predict fracture risk. Early intervention prevents catastrophic events and facilitates easier reconstruction.
- Established Pathological Fracture: To restore structural integrity, alleviate pain, and enable mobility. This is often an urgent indication.
- Spinal Cord Compression: Progressive neurological deficit from vertebral body collapse or epidural tumor extension, requiring urgent decompression and stabilization.
- Intractable Pain: When conservative measures (radiation, chemotherapy, analgesics) fail to control severe, localized pain, particularly if mechanical in nature.
- Weight-Bearing Bone Lesions: Especially in the lower extremities, where stability is critical for ambulation.
- Failed Non-Operative Management: Progression of disease or symptoms despite systemic therapy and/or radiation.
- Long Life Expectancy (>3-6 months): For patients with good performance status and effective systemic therapies, more aggressive and durable reconstruction may be justified. Prognostic scores (Tokuhashi, Tomita) are helpful.
- Solitary Metastasis or Oligometastasis: In select cases, aggressive local treatment, including wide resection, may improve local control and potentially survival, particularly for renal cell carcinoma or thyroid carcinoma.
Contraindications for Operative Management:
- Extremely Short Life Expectancy (<1 month): Surgery may not provide sufficient palliative benefit to outweigh the risks and recovery time. Aggressive medical management and radiation therapy are typically favored.
- Poor Performance Status (ECOG 3-4): Patients too frail or debilitated to tolerate anesthesia and the surgical recovery period.
- Uncontrolled Systemic Disease or Rapid Progression: If systemic disease is rapidly advancing and expected to cause death before surgical recovery, or if surgery would delay critical systemic therapy.
- Unstable Medical Comorbidities: Uncontrolled cardiac, pulmonary, renal, or hematological conditions that significantly increase surgical risk.
- Extensive Disease Burden: Multifocal, widespread metastatic disease where local surgical intervention offers minimal global benefit.
- Lesions Amenable to Non-Operative Management: Stable lesions, non-weight-bearing sites, or those responding well to radiation and systemic therapy with low fracture risk.
Operative vs. Non-Operative Indications
| Feature | Operative Management | Non-Operative Management |
|---|---|---|
| Mirels Score | ≥ 7 (impending fracture) or existing pathological fracture. | < 7 (low fracture risk). |
| Pain | Intractable, mechanically-induced pain unresponsive to radiotherapy/analgesics. | Mild to moderate pain controlled by analgesics, radiotherapy, or systemic therapy. |
| Functional Deficit | Significant functional impairment due to structural instability or neurological compromise (e.g., inability to ambulate due to femoral lesion, neurological deficit from spinal lesion). | Minimal or no functional deficit; stable neurological status. |
| Location | Weight-bearing bones (femur, tibia, spine, periacetabular), lesions threatening major joints or vital structures. | Non-weight-bearing bones (e.g., ribs, sternum, scapula, distal extremities in select cases), lesions not threatening structural integrity or neurological function. |
| Life Expectancy | Generally >3-6 months (e.g., based on Tokuhashi/Tomita scores). Ability to recover from surgery and benefit from prolonged mobility. | Generally <3 months, or poor performance status where surgical recovery is unlikely or would significantly impact remaining quality of life. |
| Systemic Disease Control | Reasonable control of primary disease, or lesions requiring stabilization to allow continuation of systemic therapy. | Rapidly progressive systemic disease, or patient too unstable for surgery. |
| Lesion Type | Extensive lytic lesions, large cortical defects, significant bone loss, periacetabular destruction, spinal instability. | Small, stable lytic lesions, blastic lesions, lesions responding well to radiotherapy/chemotherapy, without impending fracture risk. |
| Primary Tumor | Often renal, thyroid, breast, prostate, lung primaries with reasonable prognosis. | Very aggressive primaries with very poor prognosis (e.g., some sarcomas, highly aggressive lung cancers), or widespread disseminated disease. |
Pre-Operative Planning & Patient Positioning
Meticulous pre-operative planning is the cornerstone of successful management for difficult metastatic lesions, especially given the typically compromised health status of these patients.
Multidisciplinary Team (MDT) Approach
Critical involvement of medical oncology, radiation oncology, palliative care, anesthesia, and rehabilitation services. This ensures coordinated care, optimal timing of surgical intervention relative to systemic therapies and radiation, and comprehensive post-operative support.
Imaging
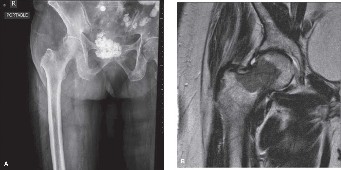
- Plain Radiographs: Initial assessment of lesion size, location, and bone destruction.
- Computed Tomography (CT): Essential for assessing cortical integrity, extent of medullary involvement, soft tissue mass, and proximity to neurovascular structures. For spinal lesions, CT provides detailed information on vertebral body collapse and pedicle involvement. For pelvic lesions, 3D reconstructions are invaluable for planning complex acetabular or sacral resections/reconstructions.
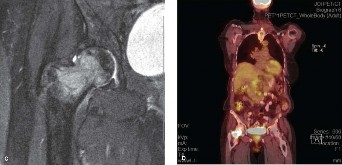
- Magnetic Resonance Imaging (MRI): Crucial for evaluating soft tissue extension, spinal cord compression, nerve root involvement, and intraosseous tumor extent. Helps delineate tumor margins, particularly for planning wide resections where appropriate.
- Positron Emission Tomography-CT (PET-CT): Useful for staging systemic disease, identifying additional lesions, and evaluating metabolic activity of the primary and metastatic sites.
-
Angiography/Embolization:
For highly vascular tumors (e.g., renal cell carcinoma, thyroid), pre-operative angiographic embolization (typically 24-72 hours pre-op) can significantly reduce intraoperative blood loss. This is especially important for large pelvic or sacral lesions.
Biopsy Considerations
- Diagnosis Confirmation: For newly diagnosed lesions without a known primary, or in cases of diagnostic uncertainty.
- Image-Guided Core Needle Biopsy: Preferred method for most lesions due to minimal invasiveness and risk of contamination. Must be performed by an experienced radiologist.
- Open Biopsy: Reserved for cases where needle biopsy is non-diagnostic or for superficial lesions where contamination risk is low.
- Biopsy Tract Planning: The biopsy tract must be planned such that it can be excised en bloc with the tumor during definitive surgery, or radiated effectively. Always perform the biopsy through a future surgical incision or in an area amenable to resection.
Patient Optimization
- Medical Workup: Comprehensive evaluation of cardiac, pulmonary, renal, and nutritional status. Optimization of comorbidities.
- Anemia Management: Pre-operative transfusions or erythropoietin administration.
- Coagulation Profile: Correction of any coagulopathies.
- Bone Health: Optimization of calcium and Vitamin D levels. Consideration of bisphosphonates or RANK ligand inhibitors (e.g., denosumab) to reduce SREs, though these need to be carefully timed around surgery to avoid osteonecrosis issues if extensive bone removal is planned.
- Pain Control: Aggressive pre-operative pain management to improve patient comfort and reduce opioid requirements.
Surgical Planning
- Approach Selection: Based on lesion location, extent, proximity to neurovascular structures, and need for wide margins vs. intralesional debulking.
- Implant Selection: Intramedullary nails, plates, endoprostheses, custom implants, cement augmentation. Considerations include achieving rigid fixation in compromised bone, bypassing the entire bone if necessary, and allowing for early mobilization.
- Contingency Planning: Anticipate potential complications like massive blood loss, nerve injury, or inability to achieve planned fixation. Ensure appropriate blood products, monitoring equipment (neuromonitoring), and additional implants are available.
- Radiation Sequencing: Coordinate with radiation oncology. Pre-operative radiation can downstage some tumors but also complicates wound healing. Post-operative radiation is common to improve local control after intralesional surgery.
Patient Positioning
- Standard Orthopedic Principles: Meticulous positioning to prevent pressure ulcers, nerve palsies, and compartment syndrome.
-
Site-Specific Considerations:
- Spine: Prone position on a radiolucent table (e.g., Jackson table) to allow for abdominal laxity, reducing epidural venous plexus engorgement and improving visualization. Neuromonitoring is often used.
- Femur/Acetabulum: Lateral decubitus for posterior approaches, supine for anterior approaches. Careful padding and access for fluoroscopy.
- Humerus: Beach chair or supine position, depending on the approach (deltopectoral vs. posterior).
- Intraoperative Imaging: C-arm or O-arm accessibility is crucial for confirming guide wire placement, reduction, and implant position.
Detailed Surgical Approach / Technique
The surgical approach to difficult metastatic lesions is highly individualized, focusing on stabilization, decompression, and pain relief. While specific approaches vary by anatomical site, general principles apply.
General Principles
- Preservation of Function: Aim to restore or maintain ambulation and upper limb use.
- Pain Palliation: Stable fixation often reduces mechanical pain.
- Local Disease Control: Achieved through intralesional debulking (curettage) or, in select cases, wide resection, often supplemented with PMMA cement, radiation, or cryoablation.
- Robust Fixation: Given poor bone quality and lack of healing potential, load-bearing constructs are required.
- Minimizing Morbidity: Procedures should be as minimally invasive as possible while achieving surgical goals, especially in systemically unwell patients.
Step-by-Step Surgical Technique (Illustrative for Long Bones and Spine)
I. Long Bone Stabilization (e.g., Femur)
A. Prophylactic Fixation of Impending Fracture:
1.
Incision & Exposure:
Standard extensile approach (e.g., lateral approach for femur). Careful dissection to minimize soft tissue stripping. Identify internervous planes (e.g., between vastus lateralis and rectus femoris for lateral femur).
2.
Lesion Evaluation:
Visually confirm the lesion's extent and cortical compromise.
3.
Intramedullary Nailing:
Preferred for diaphyseal and many metaphyseal lesions.
*
Entry Portal:
Appropriately sized based on nail design (e.g., piriformis fossa, greater trochanter tip).
*
Reaming:
Over-ream to create space for cement and nail.
*
Curettage & Cement Augmentation:
If the lesion is lytic, perform intralesional curettage to remove gross tumor burden. Irrigate thoroughly. Pack the defect with PMMA cement, ensuring good fill, particularly in cortical defects. The cement provides immediate structural support and an exothermic reaction that can cause localized tumor necrosis.
*
Nail Insertion:
Insert a robust, long intramedullary nail that bypasses the lesion by at least two cortical diameters (typically 5-10 cm) proximally and distally, engaging healthy bone. The nail should ideally extend from joint to joint or be adequately long to provide bypass fixation.
*
Locking Screws:
Place proximal and distal locking screws for rotational and angular stability.
4.
Plate Fixation (less common for primary long bone stabilization, but used for specific metaphyseal or very distal lesions):
*
Approach:
Standard exposure.
*
Curettage & Cement:
As above.
*
Plate Application:
Apply a long, strong locking plate, spanning multiple healthy cortices above and below the lesion. Screws are placed into healthy bone and, if possible, through the cement-augmented tumor site.
5.
Wound Closure:
Layered closure with meticulous hemostasis.
B. Fixation of Established Pathological Fracture:
1.
Reduction:
Gentle reduction of the fracture fragments, often challenging due to bone loss and comminution.
2.
Intramedullary Nailing:
*
Traction:
Apply traction to align the fracture.
*
Reduction Aids:
Use instruments like bone hooks, cerclage wires (temporarily), or reduction clamps.
*
Curettage & Cement:
Thorough curettage of the fracture site. PMMA cement is crucial to fill the defect and provide stability, especially in comminuted fractures. Cement is often injected before or after reaming/nailing, depending on fracture morphology.
*
Nail Insertion:
Insert a long nail bypassing the fracture by at least two healthy cortical diameters.
*
Locking Screws:
Secure the nail with proximal and distal locking screws.
3.
Endoprosthetic Replacement:
For extensive bone loss, highly comminuted fractures, or periarticular involvement (e.g., proximal femur, proximal humerus), segmental resection and reconstruction with a tumor prosthesis may be the most durable solution. This often provides the most immediate stability and allows for rapid weight-bearing.
*
Resection:
En bloc resection of the involved bone segment.
*
Prosthesis Insertion:
Implant a modular tumor prosthesis, restoring length and joint function. Cement may be used for stem fixation.
*
Reattachment:
Reattach surrounding muscles and tendons to the prosthesis where possible to restore function.
II. Spinal Stabilization (e.g., Thoracic or Lumbar Spine)
A. Posterior Approach for Decompression and Stabilization:
1.
Positioning:
Prone on a radiolucent table.
2.
Incision:
Midline posterior incision, exposing the posterior elements of the affected and adjacent vertebrae.
3.
Exposure:
Subperiosteal dissection of paraspinal muscles. Identify appropriate pedicles for instrumentation.
4.
Decompression (Laminectomy/Costotransversectomy):
*
Laminectomy:
If posterior compression is present.
*
Costotransversectomy/Transpedicular Approach:
For direct decompression of the spinal canal from epidural tumor extension or bone fragments. This involves resecting the transverse process and rib head/neck (thoracic) or facet joint (lumbar), accessing the lateral aspect of the vertebral body and epidural space.
5.
Instrumentation:
Pedicle screw fixation spanning at least two levels above and two levels below the affected segment.
*
Pedicle Screws:
Placed under fluoroscopic guidance.
*
Rods:
Contoured and secured to the pedicle screws.
6.
Vertebral Body Reconstruction (if significant anterior column loss):
*
Corpectomy:
Resection of the involved vertebral body/bodies.
*
Anterior Column Support:
Insertion of a expandable cage or allograft, often packed with PMMA cement, to restore anterior column height and stability.
7.
Cement Augmentation (Vertebroplasty/Kyphoplasty):
For vertebral body compression fractures without significant neurological compromise or instability requiring open fixation. Percutaneous injection of PMMA to stabilize the fracture and relieve pain. This can also be used adjunctively during open procedures to augment screw purchase.

8.
Wound Closure:
Meticulous layered closure.
III. Pelvic/Periacetabular Reconstruction
- Highly Complex: Often requires custom implants or cages (e.g., Harrington reconstruction, saddle prostheses) to bridge massive bone defects and restore hip function.
- Approaches: Combined anterior and posterior approaches may be necessary.
- Technique: Wide resection (if indicated and feasible), reconstruction with large volumes of cement, often reinforced with mesh or plates, followed by acetabular cup placement. Blood loss can be substantial, necessitating pre-operative embolization for hypervascular lesions.
Adjuvant Intraoperative Therapies
- PMMA Cement: Critical for filling defects, providing immediate stability, augmenting screw purchase, and generating local heat for tumor necrosis.
- Cryoablation/Radiofrequency Ablation: Can be used adjunctively for local tumor control, particularly in cases of intralesional curettage where residual microscopic disease may be present.
Complications & Management
Management of difficult metastatic lesions is inherently associated with a higher risk of complications due to the patient's compromised health, the aggressive nature of the disease, and the challenging surgical environment.
Common Complications and Salvage Strategies
| Complication | Incidence | Salvage Strategies & Management |
|---|---|---|
| Infection (SSI) | 5-15% (higher in immunocompromised) | Prevention: Strict aseptic technique, prophylactic antibiotics, optimization of patient health. Management: Debridement, irrigation, retention/removal of hardware (depending on timing and extent of infection), appropriate intravenous antibiotics based on culture results. May require staged procedures or implant exchange. |
| Hardware Failure | 10-25% (fatigue fracture, pullout) | Prevention: Use of robust, load-bearing implants; adequate bypass of lesion; cement augmentation; appropriate patient activity restrictions. Management: Revision surgery with stronger, longer construct; exchange to tumor prosthesis if original fixation inadequate; consider re-radiation or systemic therapy. |
| Local Recurrence | Varies widely (5-30% depending on primary and margin) | Prevention: Aggressive intralesional curettage with cement/adjuvant therapy, post-operative radiation. Management: Re-evaluation with imaging. If amenable, re-excision/debulking, re-radiation, cryoablation, or systemic therapy intensification. Prognosis often poor. |
| Vascular Injury | Rare (<1-5%) but limb/life-threatening | Prevention: Meticulous dissection, careful identification and protection of vessels, pre-operative angiography/embolization for hypervascular tumors. Management: Immediate vascular repair by a surgeon, potentially with bypass grafting. Significant blood loss management. |
| Neurological Deficit | <5% (limb paresis/paralysis, bowel/bladder dysfunction) | Prevention: Careful surgical technique, intraoperative neuromonitoring (SSEP, MEP), precise screw placement, cautious decompression. Management: Immediate post-operative assessment. If acute, consider urgent re-exploration for decompression or hematoma evacuation. Steroids for swelling. Rehabilitation. |
| Excessive Blood Loss | Common with hypervascular tumors (renal, thyroid) | Prevention: Pre-operative embolization, meticulous hemostasis, tourniquet use (if applicable), tranexamic acid, cell saver. Management: Massive transfusion protocol, correction of coagulopathy, reversal agents, careful monitoring of vital signs. |
| Deep Vein Thrombosis (DVT) / Pulmonary Embolism (PE) | 5-15% | Prevention: Pharmacological prophylaxis (LMWH), mechanical prophylaxis (SCDs), early mobilization. Management: Anticoagulation (heparin bridge to oral anticoagulation), IVC filter in select cases. |
| Fat Embolism Syndrome (FES) | Rare but serious (especially with IM nailing) | Prevention: Careful reaming, venting of medullary canal, avoidance of excessive intramedullary pressure. Management: Supportive care, oxygenation, ventilatory support. |
| Wound Dehiscence/Skin Necrosis | 5-10% | Prevention: Gentle tissue handling, meticulous closure, avoidance of tension, good nutritional status. Management: Local wound care, débridement, negative pressure wound therapy (NPWT). May require plastic surgery consultation for flap coverage. |
| Non-Union | Rare, as healing is not primary goal. More often hardware failure with non-progression. | Prevention: Robust, load-bearing fixation. Management: Re-evaluation of patient prognosis. If justified, revision surgery with further cement augmentation, stronger implants. Often, patient prognosis means this is less relevant than in typical trauma cases, focusing on pain and function with the existing (even failed) construct. |
| Systemic Complications | Varies widely (cardiac, renal, respiratory failure) | Prevention: Thorough pre-operative medical optimization, careful intraoperative fluid management, experienced anesthesia. Management: Intensive care unit (ICU) monitoring, specialist consultations, supportive care. |
Post-Operative Rehabilitation Protocols
Post-operative rehabilitation is crucial for achieving the palliative goals of surgery: pain relief, functional restoration, and improved quality of life. Protocols must be tailored to the individual patient, considering their underlying medical status, tumor biology, the extent of resection, and the stability of the surgical construct.
General Principles:
- Early Mobilization: The overarching goal is to mobilize the patient as quickly and safely as possible to prevent complications associated with immobility (e.g., DVT/PE, pneumonia, deconditioning).
- Pain Management: Aggressive, multimodal pain management is essential to facilitate participation in physical therapy. This includes opioids, NSAIDs (if not contraindicated), neuropathic agents, and regional blocks.
-
Weight-Bearing Status:
Dictated by the stability of the reconstruction and the bone quality.
- Protected Weight-Bearing: Often initially prescribed for long bone fixations, gradually progressing to full weight-bearing as tolerated, typically facilitated by cement augmentation and robust implants.
- Full Weight-Bearing as Tolerated (FWBAT): Achieved rapidly with endoprosthetic replacements due to inherent mechanical stability.
- Spinal Surgery: Patients are often mobilized with a brace, with progression to unbraced activity based on construct stability and surgeon preference.
- Activity Restrictions: Patients are typically advised to avoid high-impact activities, heavy lifting, or activities that place excessive torsional stress on the operated limb.
- Psychosocial Support: Addressing anxiety, depression, and fear of movement is critical. Involvement of palliative care and social work.
Site-Specific Considerations:
1. Lower Extremity (Femur, Tibia, Periacetabulum):
-
Day 0-3:
- Pain control, DVT prophylaxis.
- Bedside mobility exercises: ankle pumps, quad sets, gluteal sets.
- Initiate sitting balance.
- Initiate transfer training with assist to chair.
- Partial weight-bearing (PWB) or FWBAT, as dictated by construct, with assistive devices (walker).
-
Weeks 1-6:
- Progressive gait training with assistive devices.
- Increase weight-bearing as tolerated, if not FWBAT initially.
- Strengthening exercises for hip and knee musculature.
- Range of motion (ROM) exercises for affected joint.
- Stair training as appropriate.
-
Beyond 6 Weeks:
- Advance strengthening and functional training.
- Transition to cane or no assistive device.
- Focus on endurance and community ambulation.
- Continue monitoring for pain and hardware integrity.
2. Upper Extremity (Humerus):
-
Day 0-3:
- Pain control.
- Passive and active-assisted ROM for shoulder and elbow.
- Gentle pendulum exercises.
- Finger and wrist ROM.
-
Weeks 1-6:
- Progressive active ROM and gentle strengthening.
- Focus on activities of daily living (ADLs).
- Avoid lifting heavy objects or sudden movements.
-
Beyond 6 Weeks:
- Advance strengthening for shoulder girdle and arm.
- Focus on functional tasks, light carrying.
- Return to modified recreational activities if appropriate.
3. Spine:
-
Day 0-3:
- Pain control.
- Log-roll technique for position changes.
- Sitting balance, transfers to chair, initiation of ambulation with assistive device (walker).
- Often fitted with a thoracolumbar sacral orthosis (TLSO) or cervical collar if deemed unstable.
-
Weeks 1-6:
- Progressive ambulation with assistive devices.
- Core stabilization exercises (gentle).
- Maintain good posture, educate on body mechanics.
- Brace weaning considered based on spinal stability and fusion progress (if applicable) and surgeon preference.
-
Beyond 6 Weeks:
- Advanced core strengthening, gentle stretching.
- Gradual increase in activity level, avoiding twisting or heavy lifting.
- Continued emphasis on proper body mechanics.
Long-Term Follow-up:
- Regular clinical evaluation and imaging (plain radiographs) to monitor hardware integrity, local recurrence, and stability of the construct.
- Ongoing pain management and adjustment of systemic therapies.
- Coordination with the multidisciplinary oncology team.
Summary of Key Literature / Guidelines
The management of difficult metastatic lesions is guided by a growing body of evidence and consensus guidelines, primarily focusing on improving quality of life and functional outcomes.
1. Prognostic Scoring Systems:
- Tokuhashi Score (Revised): A widely used system to predict survival in patients undergoing surgery for spinal metastases. It considers the patient's general condition (ECOG), number of extraspinal bone metastases, number of vertebral metastases, metastasis to major internal organs, primary cancer type, and paralysis. Scores range from 0-15, correlating with predicted survival (e.g., <5 points: <3 months; 9-11 points: >10 months).
- Tomita Score: Another prognostic index for spinal metastases, focusing on primary tumor malignancy, visceral metastases, and bone metastases. It helps guide decisions on local surgery (intralesional vs. wide resection).
- Mirels Score: Essential for predicting the risk of impending pathological fractures in long bones. It considers the site, size, and type (lytic, blastic, mixed) of the lesion. A score of ≥ 7 typically indicates a high risk of fracture and justifies prophylactic surgical stabilization.
2. Surgical Indications and Techniques:
- National Comprehensive Cancer Network (NCCN) Guidelines: Provide comprehensive, evidence-based recommendations for managing bone metastases, including indications for surgery, radiation, and systemic therapy. They emphasize a multidisciplinary approach.
- European Spine Journal (ESJ) Consensus Guidelines: Offer practical guidance for the surgical management of spinal metastases, often recommending decompression and stabilization for neurological deficits, intractable pain, or instability.
- Cement Augmentation (PMMA): Numerous studies support the use of PMMA in orthopedic oncology, demonstrating its ability to provide immediate structural support, improve screw purchase in compromised bone, reduce pain, and generate local heat for tumor cell destruction. It is particularly valuable for stabilizing pathological fractures or impending fractures in osteolytic lesions.
- Intramedullary Nailing vs. Plating: Literature generally favors intramedullary nailing for diaphyseal long bone lesions due to its load-sharing capabilities and superior biomechanical properties in these locations, offering a more robust fixation for weight-bearing. Plates are often reserved for metaphyseal or very distal lesions.
- Endoprosthetic Reconstruction: Indicated for extensive bone loss, highly comminuted periarticular fractures, or when durable reconstruction for a longer life expectancy is desired. Studies demonstrate good functional outcomes and high rates of immediate weight-bearing, albeit with higher initial surgical morbidity.
3. Role of Adjuvant Therapies:
- Radiotherapy: Pre- or post-operative radiation is a cornerstone of metastatic bone disease management. Pre-operative radiation can downstage tumors but may impact wound healing. Post-operative radiation (especially after intralesional surgery) is highly effective for local control and pain relief.
- Systemic Therapies: Coordination with medical oncology for optimal timing of chemotherapy, targeted therapies, immunotherapy, and bone-modifying agents (bisphosphonates, denosumab) is crucial. These agents reduce SREs and can improve bone integrity.
- Pre-operative Embolization: For hypervascular tumors (renal cell carcinoma, thyroid carcinoma), pre-operative embolization has been consistently shown to significantly reduce intraoperative blood loss, improving surgical safety and outcomes.
4. Quality of Life and Functional Outcomes:
- Studies consistently demonstrate that appropriately timed and executed surgical interventions for difficult metastatic lesions can significantly improve patient quality of life by alleviating pain, restoring function, and enabling participation in activities of daily living, even in the context of advanced disease.
- Patient-reported outcome measures (PROMs) are increasingly used to assess the impact of interventions from the patient's perspective, emphasizing the palliative nature of these procedures.
The optimal management of difficult metastatic lesions demands a nuanced, individualized, and multidisciplinary approach, integrating prognostic factors, advanced imaging, robust surgical techniques, and comprehensive adjuvant therapies to maximize functional outcomes and improve quality of life for this challenging patient population.
Clinical & Radiographic Imaging