Decode Upper Extremity Nerves: Drake RL ET Anatomy
Key Takeaway
Your ultimate guide to Decode Upper Extremity Nerves: Drake RL ET Anatomy starts here. The brachial plexus, a network of nerves (C5-T1) exiting between scalene muscles, comprises roots, trunks, divisions, cords, and branches (mnemonic: Real Texans drink cold beer). Key terminal nerves include musculocutaneous, axillary, radial, median, and ulnar. Drake rl et, this plexus is vital for upper limb motor and sensory function.
Introduction and Epidemiology
Upper extremity nerve injuries, encompassing both traumatic brachial plexus lesions and peripheral nerve entrapment syndromes, represent a complex domain of orthopedic and peripheral nerve surgery. The brachial plexus is a highly intricate neural network responsible for the motor and sensory innervation of the upper limb. Injuries to this structure, whether from high-energy trauma, iatrogenic causes, or obstetric complications, result in profound functional impairment.

Epidemiologically, adult traumatic brachial plexus injuries are predominantly observed in young males, typically secondary to high-speed motor vehicle collisions, particularly motorcycle accidents. The mechanism usually involves forced lateral flexion of the cervical spine with concomitant shoulder depression, leading to traction, rupture, or avulsion of the nerve roots. Obstetric brachial plexus palsies, such as Erb-Duchenne (C5-C6) and Klumpke (C8-T1) palsies, occur secondary to shoulder dystocia during difficult vaginal deliveries.

Understanding the precise anatomical hierarchy of the brachial plexus and distal peripheral nerves is paramount for accurate clinical localization, surgical decision-making, and execution of complex nerve reconstructions, including neurolysis, grafting, and targeted nerve transfers.
Pathophysiology of Nerve Injury
Peripheral nerve injuries are classically categorized by the Seddon (neurapraxia, axonotmesis, neurotmesis) and Sunderland classifications. In the context of the brachial plexus, a critical distinction must be made between preganglionic (avulsion) and postganglionic (rupture) lesions. Preganglionic injuries occur proximal to the dorsal root ganglion within the central nervous system. Because the central nervous system lacks regenerative capacity, these injuries are generally irreparable primarily and require nerve transfer strategies. Postganglionic injuries occur distal to the dorsal root ganglion and retain the potential for Wallerian degeneration and subsequent axonal regeneration, making them amenable to direct repair or nerve grafting if the gap is manageable.

Surgical Anatomy and Biomechanics
A rigorous understanding of upper extremity neuroanatomy is the foundation of peripheral nerve surgery. The brachial plexus follows a strict proximal-to-distal organizational hierarchy consisting of roots, trunks, divisions, cords, and terminal branches. A common mnemonic utilized in surgical training is "Real Texans drink cold beer."
Brachial Plexus Architecture
The brachial plexus is formed by the ventral primary rami of the fifth cervical through the first thoracic spinal nerves (C5-T1). Variations exist, including pre-fixed (significant C4 contribution) and post-fixed (significant T2 contribution) plexuses. The roots exit the cervical spine and pass between the anterior and middle scalene muscles, a critical anatomical landmark during supraclavicular exploration.

The five roots converge to form three trunks within the posterior triangle of the neck:
1. Upper Trunk: Formed by the union of C5 and C6 roots.
2. Middle Trunk: A direct continuation of the C7 root.
3. Lower Trunk: Formed by the union of C8 and T1 roots.
Each trunk subsequently bifurcates into anterior and posterior divisions posterior to the clavicle, reflecting the functional division of the limb into flexor (anterior) and extensor (posterior) compartments. There are no terminal branches arising directly from the divisions.

The six divisions recombine to form three cords, which are named based on their anatomical relationship to the axillary artery:
1. Lateral Cord: Formed by the anterior divisions of the upper and middle trunks (C5-C7).
2. Medial Cord: Formed by the anterior division of the lower trunk (C8-T1).
3. Posterior Cord: Formed by the posterior divisions of all three trunks (C5-T1).
Preclavicular and Minor Branches
Identifying preclavicular branches is essential for distinguishing preganglionic from postganglionic injuries. Preclavicular branches arise directly from the roots and trunks:
1. Dorsal Scapular Nerve (C5 root): Innervates the levator scapulae and rhomboid major/minor.
2. Long Thoracic Nerve (C5-C7 roots): Innervates the serratus anterior. Injury leads to medial scapular winging.
3. Suprascapular Nerve (Upper trunk, C5-C6): Innervates the supraspinatus and infraspinatus.
4. Nerve to the Subclavius (Upper trunk, C5-C6).

Minor branches arising from the cords provide specific regional innervation. All minor medial and lateral cord branches contain the words "medial" or "lateral" in their nomenclature, whereas posterior cord branches do not.
* Lateral Cord: Lateral pectoral nerve (C5-C7) to the pectoralis major.
* Medial Cord: Medial pectoral nerve (C8-T1), medial brachial cutaneous nerve, and medial antebrachial cutaneous nerve.
* Posterior Cord: Upper subscapular nerve (C5-C6) to the subscapularis; lower subscapular nerve (C5-C6) to the subscapularis and teres major; thoracodorsal nerve (C6-C8) to the latissimus dorsi.

Terminal Branches and Motor Innervation
The cords terminate into five major peripheral nerves:
1. Musculocutaneous Nerve (Lateral cord): Innervates the anterior compartment of the arm (coracobrachialis, biceps brachii, brachialis).
2. Axillary Nerve (Posterior cord): Innervates the deltoid and teres minor.
3. Radial Nerve (Posterior cord): Innervates the extensor compartments of the arm and forearm.
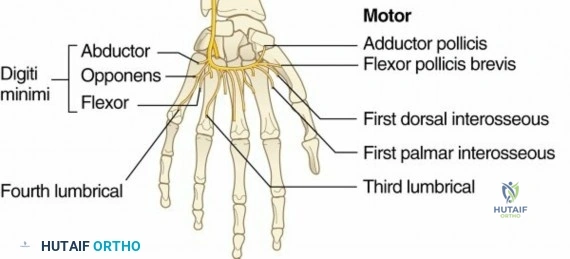
4. Median Nerve (Medial and Lateral cords): Innervates the majority of the flexor compartment of the forearm and the thenar eminence.
5. Ulnar Nerve (Medial cord): Innervates the intrinsic musculature of the hand and specific forearm flexors (flexor carpi ulnaris, medial half of flexor digitorum profundus).

Carpal Tunnel Borders and Distal Anatomy
Distally, the median nerve traverses the carpal tunnel, a rigid fibro-osseous canal. Understanding these borders is critical for endoscopic and open carpal tunnel release procedures.
* Roof: Transverse carpal ligament (flexor retinaculum).
* Floor: Proximal and distal rows of the carpal bones.
* Radial Border: Scaphoid tubercle and the crest of the trapezium.
* Ulnar Border: Pisiform and the hook of the hamate.

Indications and Contraindications
Surgical intervention for upper extremity nerve injuries requires meticulous patient selection. The timing of surgery is dictated by the mechanism of injury, the anatomical level of the lesion, and the presence of preganglionic versus postganglionic disruption.

Sharp, open transections warrant immediate exploration and primary neurorrhaphy. Conversely, closed traction injuries are typically observed for 3 to 6 months to monitor for spontaneous clinical or electromyographic recovery. Failure to demonstrate reinnervation within this window is a primary indication for surgical exploration and reconstruction.
Surgical Decision Making
| Indication Type | Specific Clinical Scenarios | Operative Strategy |
|---|---|---|
| Operative (Emergent/Urgent) | Open, sharp nerve transection; concomitant expanding hematoma/vascular injury; acute compartment syndrome. | Immediate exploration, primary end-to-end neurorrhaphy, or vascular repair. |
| Operative (Delayed 3-6 Months) | Closed traction injury with no clinical/EMG recovery; flail limb; severe neuropathic pain refractory to medical management. | Nerve exploration, neurolysis, autologous nerve grafting, or targeted nerve transfers. |
| Operative (Late >12 Months) | Chronic denervation with irreversible motor endplate loss; late presentation BPI. | Free functioning muscle transfer (FFMT), tendon transfers, arthrodesis. |
| Non-Operative | Neuropraxia; progressive clinical and EMG improvement; poor surgical candidate (severe comorbidities). | Physical therapy, orthoses, serial clinical and electrodiagnostic monitoring. |

Contraindications to primary nerve repair or grafting include prolonged denervation times (typically exceeding 12 to 18 months), as the motor endplates undergo irreversible fibrosis. In such cases, the surgical algorithm shifts toward palliative reconstructive options such as tendon transfers or free functioning muscle transfers (e.g., gracilis flap).
Pre Operative Planning and Patient Positioning
Comprehensive preoperative evaluation relies on a combination of serial clinical examinations, electrodiagnostic studies, and advanced neuroimaging.
Clinical Evaluation and Electromyography
Clinical examination must meticulously assess individual muscle groups to localize the lesion. Scapular winging is a critical localizing sign. Medial winging indicates long thoracic nerve (C5-C7) injury leading to serratus anterior dysfunction, characterized by superior elevation with the scapula translated medially. Lateral winging indicates spinal accessory nerve (Cranial Nerve XI) injury, often secondary to iatrogenic posterior triangle neck surgery, leading to trapezius dysfunction and lateral translation of the scapula.

Nerve conduction studies (NCS) and electromyography (EMG) are indispensable. The hallmark of a preganglionic avulsion injury is the presence of intact sensory nerve action potentials (SNAPs) in an anesthetic dermatome, coupled with absent compound muscle action potentials (CMAPs). This occurs because the dorsal root ganglion remains contiguous with the peripheral sensory axon, preventing Wallerian degeneration of the sensory nerve, despite central disconnection.

Imaging Modalities
Magnetic Resonance Imaging (MRI) and CT myelography are the gold standards for evaluating the cervical nerve roots. The presence of a pseudomeningocele (an extravasation of cerebrospinal fluid through a dural tear) is highly suggestive, though not pathognomonic, of a root avulsion. High-resolution MRI neurography can also visualize neuromas-in-continuity and assess the length of nerve gaps, aiding in preoperative graft calculation.

Positioning and Anesthesia
For comprehensive brachial plexus exploration, the patient is positioned supine with a bump placed under the ipsilateral scapula. The head is turned to the contralateral side to expose the posterior triangle of the neck. The entire upper extremity, hemithorax, and neck are prepped and draped free. If autologous nerve grafting is anticipated, the bilateral lower extremities must be prepped for sural nerve harvest. General anesthesia is utilized; however, long-acting paralytic agents must be strictly avoided to allow for intraoperative nerve stimulation and electrophysiological monitoring.

Detailed Surgical Approach and Technique
Surgical exploration of the brachial plexus is typically divided into supraclavicular and infraclavicular approaches, depending on the suspected level of injury. Often, an extensile combined approach is required, necessitating osteotomy of the clavicle for complete visualization of the retroclavicular divisions.
Supraclavicular Exploration
The supraclavicular approach targets the spinal nerves, roots, and trunks.
1. An incision is made along the posterior border of the sternocleidomastoid muscle, extending laterally parallel to the clavicle.
2. The platysma is divided, and the external jugular vein is either retracted or ligated.
3. The supraclavicular nerves are identified and preserved if possible.
4. The omohyoid muscle is identified and often divided to enhance exposure.
5. The transverse cervical and suprascapular vessels are identified and ligated as necessary.
6. The phrenic nerve is identified coursing from lateral to medial across the anterior surface of the anterior scalene muscle. It must be carefully mobilized and protected.
7. The anterior scalene muscle is divided, revealing the underlying roots (C5-T1) and trunks of the brachial plexus.
8. Intraoperative nerve stimulation is performed to assess for continuity and function.

Infraclavicular Exploration
The infraclavicular approach is utilized to expose the cords and terminal branches.
1. The incision continues from the clavicle distally along the deltopectoral groove.
2. The cephalic vein is identified and retracted laterally with the deltoid.
3. The pectoralis major is retracted medially. For extensive exposure, the pectoralis major tendon may be partially or completely released from its humeral insertion.
4. The pectoralis minor tendon is identified and tenotomized at the coracoid process.
5. The clavipectoral fascia is incised, exposing the axillary sheath.
6. The lateral, posterior, and medial cords are identified based on their relationship to the axillary artery. The musculocutaneous nerve is traced penetrating the coracobrachialis.

Nerve Transfer Strategies
In cases of preganglionic avulsion or delayed presentation where the proximal nerve stump is unavailable or the distance to the motor endplate is too great for a nerve graft, nerve transfers (neurotization) are employed. This involves taking an expendable functioning nerve (donor) and coapting it directly to the distal stump of an injured nerve (recipient) close to the target muscle.

Common nerve transfers include:
* Spinal Accessory Nerve to Suprascapular Nerve: Restores shoulder abduction and external rotation.
* Oberlin Transfer: Transfer of a redundant ulnar nerve fascicle (typically supplying the flexor carpi ulnaris) to the biceps motor branch of the musculocutaneous nerve to restore elbow flexion.
* Somsak Transfer: Transfer of a triceps motor branch (from the radial nerve) to the axillary nerve to restore deltoid function and shoulder abduction.
* Intercostal Nerves to Musculocutaneous Nerve: Utilized in global brachial plexus avulsions to restore elbow flexion.

Complications and Management
Surgical intervention in the brachial plexus is fraught with potential complications due to the dense anatomical concentration of critical neurovascular structures. Meticulous dissection and comprehensive anatomical knowledge are the primary deterrents to iatrogenic injury.

Intraoperative and Postoperative Complications
| Complication | Incidence / Risk Factors | Management and Salvage Strategy |
|---|---|---|
| Phrenic Nerve Injury | Highest risk during anterior scalene division. | Preoperative pulmonary function testing. Intraoperative identification. If injured, may require diaphragmatic plication if symptomatic. |
| Vascular Injury | Axillary or subclavian artery/vein laceration during scar dissection. | Immediate pressure. Proximal and distal control. Primary vascular repair or interposition vein grafting. |
| Chyle Leak | Left-sided supraclavicular dissection near the thoracic duct. | Intraoperative identification requires primary ligation. Postoperative leaks managed with medium-chain triglyceride diet or surgical re-exploration. |
| Neuroma Formation | Occurs at harvest sites (sural nerve) or failed coaptations. | Excision of neuroma and burying of the proximal nerve stump into deep muscle or bone. |
| Failure of Reinnervation | High risk in proximal injuries with long graft lengths or delayed surgery (>6-9 months). | Conversion to palliative procedures: Free functioning muscle transfer (FFMT), tendon transfers, or joint arthrodesis. |

Post Operative Rehabilitation Protocols
Postoperative rehabilitation is a protracted process, often spanning 18 to 24 months, mirroring the slow rate of axonal regeneration (approximately 1 mm per day). The rehabilitation protocol is phased to protect the surgical repair while preventing joint contractures and facilitating motor reeducation.
Phases of Motor Reeducation
- Immobilization Phase (Weeks 0-3): The limb is immobilized in a sling or custom orthosis to eliminate tension across nerve coaptations or graft sites.
- Passive Range of Motion Phase (Weeks 3-8): Gentle, protected passive range of motion is initiated to prevent capsular contractures and maintain joint suppleness. Extreme caution is maintained to avoid stretching the repaired neural structures.
- Active Range of Motion and Strengthening Phase (Months 2-12+): As reinnervation occurs, active-assisted and active range of motion exercises are introduced.
- Cortical Plasticity and Biofeedback: For patients undergoing nerve transfers, cortical remapping is essential. For example, following an Oberlin transfer, the patient must initially attempt to flex the wrist (activating the ulnar donor nerve) to achieve elbow flexion. Over time, neuroplasticity allows spontaneous elbow flexion without conscious wrist flexion. Electromyographic biofeedback is highly beneficial during this phase.

Summary of Key Literature and Guidelines
The surgical management of upper extremity nerve injuries has evolved significantly over the past several decades, transitioning from extensive, long-segment nerve grafting to targeted, distal nerve transfers.
Evidence Based Nerve Reconstruction
The foundational work by Millesi in the 1970s established the principles of tension-free interfascicular nerve grafting utilizing the sural nerve, fundamentally altering the approach to large nerve gaps. However, the outcomes for proximal root avulsions remained poor due to the prolonged time required for axons to reach distal targets.
The paradigm shifted with the introduction of targeted nerve transfers. Oberlin et al. (1994) revolutionized the treatment of upper trunk injuries by demonstrating reliable restoration of elbow flexion utilizing redundant fascicles of the ulnar nerve transferred directly to the biceps motor branch. This technique bypassed the zone of injury, significantly reduced the regeneration distance, and capitalized on the robust motor axon count of the ulnar nerve.
Subsequent advancements by Mackinnon and colleagues expanded the repertoire of nerve transfers, establishing reliable algorithms for restoring shoulder abduction (spinal accessory or triceps branch transfers) and hand function. Current guidelines emphasize early referral to specialized peripheral nerve centers, utilizing advanced high-resolution MRI neurography for preoperative planning, and prioritizing early nerve transfer (within 6 months of injury) to optimize motor endplate reinnervation and functional outcomes.
Clinical & Radiographic Imaging






















