Internal Topography and Microsurgery of Peripheral Nerves
Key Takeaway
The internal topography of peripheral nerves dictates the success of microsurgical repair. Proximal nerve segments exhibit complex, interwoven fascicular networks, whereas distal segments maintain parallel, distinct fascicles over longer distances. Understanding this architecture, alongside intrinsic vascularity and Wallerian degeneration mechanics, is paramount for orthopedic surgeons performing accurate neurorrhaphy, minimizing neuroma formation, and optimizing functional recovery following peripheral nerve trauma.
INTERNAL TOPOGRAPHY OF PERIPHERAL NERVES
The precise internal topography, or fascicular arrangement, of major peripheral nerve trunks—specifically the radial, median, and ulnar nerves—was definitively described by Sunderland in 1945. He mapped a highly complex, dynamic network of branching and intermingling fascicles that constantly change their spatial orientation throughout the longitudinal course of the nerve.
Understanding this internal architecture is not merely an academic exercise; it is the foundational prerequisite for successful peripheral nerve microsurgery. Studies have consistently demonstrated that while the fascicular arrangement is highly complex and interwoven in the proximal aspect of a peripheral nerve, the topography simplifies distally. In the distal segments, fascicles can often be dissected over long distances before significant merging or crossover occurs. This anatomical characteristic is of paramount importance to the reconstructive microsurgeon when intraneural dissection, fascicular matching, and accurate neurorrhaphy are required.

FIGURE 62-6 Complexity of internal topography diminishes in more distal portions of nerve.
Clinical Pearl: When performing a repair of a proximal nerve transection, the surgeon must account for the plexiform nature of the fascicles. A resection of even a few millimeters can result in a completely different fascicular map on the proximal and distal stumps, making precise fascicular alignment highly challenging. Conversely, distal injuries allow for more reliable fascicular matching due to the parallel nature of the tracts.
Microscopic Anatomy of the Peripheral Nerve
To master the surgical management of nerve injuries, the surgeon must possess a granular understanding of the microscopic anatomy. The fundamental functional unit of the peripheral nerve is the nerve fiber, which consists of an axon, its myelin sheath (in myelinated nerves), and the enveloping Schwann cells.
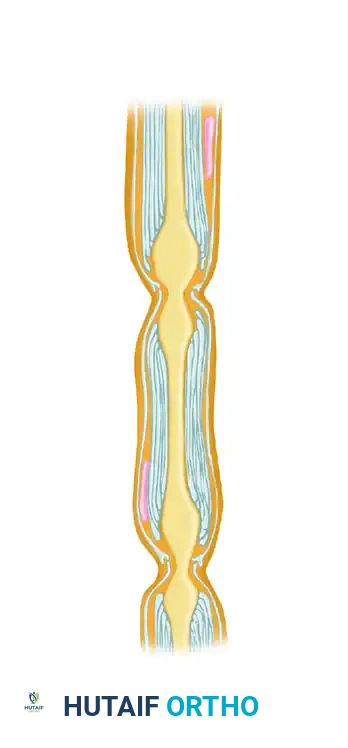
FIGURE 62-4 Basic anatomy of myelinated nerve fiber, demonstrating the Schwann cell nucleus, myelin sheath, basal lamina, and the nodal gap (Node of Ranvier).
The axon is a direct cytoplasmic extension of the nerve cell body located in the anterior horn of the spinal cord (motor) or the dorsal root ganglion (sensory). Axons are enveloped by Schwann cells, which produce the myelin sheath in myelinated fibers. This sheath is interrupted at regular intervals by the Nodes of Ranvier, which facilitate rapid saltatory conduction of action potentials.

Microscopic cross-section highlighting the myelin sheath and axonal core.

Spinal cord cross-section demonstrating the anterior and posterior roots converging to form the peripheral nerve.
Connective Tissue Architecture
The structural integrity and biomechanical properties of a peripheral nerve are dictated by its connective tissue layers, which also serve as the primary substrates for surgical repair.
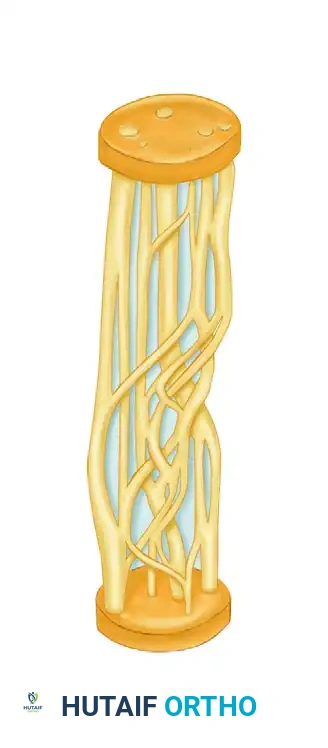
- Endoneurium: The innermost layer of loose connective tissue that surrounds individual nerve fibers (axons and their Schwann cells). It forms the "endoneurial tube," a critical structure for guiding regenerating axonal sprouts.
- Perineurium: A dense, metabolically active layer of connective tissue that bundles groups of nerve fibers into distinct fascicles (funiculi). The perineurium provides the primary tensile strength of the nerve and acts as a vital blood-nerve barrier, maintaining the intraneural microenvironment.
- Epineurium: The outermost layer, subdivided into the internal epineurium (which cushions the fascicles within the nerve trunk) and the external epineurium (which defines the outer boundary of the nerve). The epineurium is highly vascularized and absorbs compressive and longitudinal stress.

FIGURE 62-3 Microscopic anatomy of peripheral nerve, detailing the funiculus, endoneurium, perineurium, and epineurium.
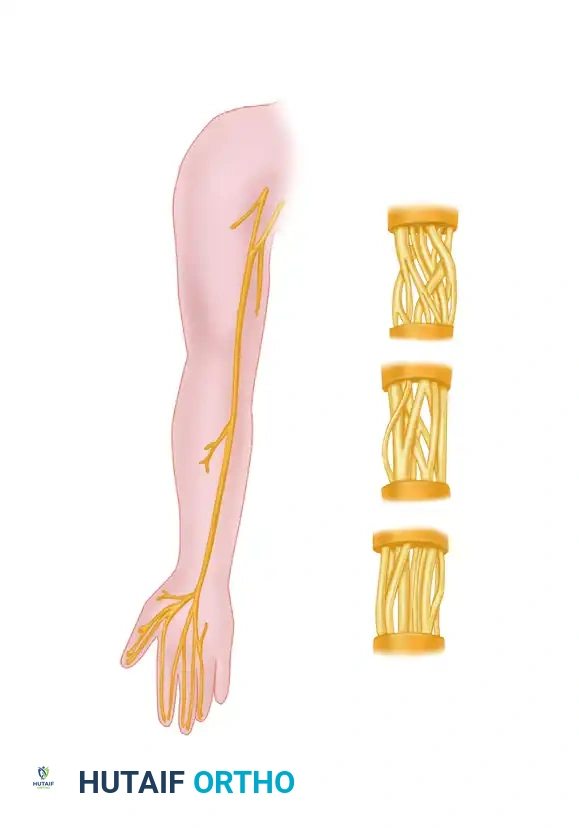
Internal topography of the Median nerve and Anterior interosseous nerve.
VASCULAR SUPPLY AND SURGICAL MOBILIZATION
The peripheral nerve enters the extremity through the mesoneurium, a specialized layer of loose connective tissue extending from the epineurium to the surrounding fascial beds. The vascularity of a peripheral nerve is highly redundant, comprising two distinct but interconnected systems:
- Extrinsic (Segmental) Blood Supply: Derived from regional vessels that enter the nerve via the mesoneurium at segmental intervals.
- Intrinsic (Longitudinal) Blood Supply: A rich, continuous plexus of microvessels running longitudinally within the epineurium, perineurium, and endoneurium.
Surgical Warning: The extensive nature of the intrinsic longitudinal blood supply allows the surgeon to mobilize peripheral nerves over variable lengths without causing complete devascularization. However, aggressive, circumferential stripping of the mesoneurium over extensive distances (typically >8-10 cm) can compromise this delicate intrinsic plexus, leading to ischemic fibrosis and failure of nerve regeneration.
PATHOPHYSIOLOGY OF NERVE INJURY AND REGENERATION
When a peripheral nerve is severed (neurotmesis) or severely crushed (axonotmesis), a predictable cascade of cellular events occurs both proximally in the cell body and distally in the isolated nerve segment.
Proximal Cellular Response: Chromatolysis
Following injury, the nerve cell body undergoes a profound metabolic shift to support regeneration, a process known as chromatolysis. The severity of chromatolysis is directly proportional to the proximity of the injury to the cell body; more proximal injuries elicit more pronounced changes.
Histologically, chromatolysis is characterized by swelling of the cytoplasm, eccentric displacement of the nucleus, and the dispersion of Nissl substance (rough endoplasmic reticulum). This reaction is typically evident by day 7 post-injury. The fate of the neuron—either cell death or the initiation of recovery—becomes apparent after 4 to 6 weeks. With successful recovery, cellular edema subsides, the nucleus migrates back toward the center of the cell, and Nissl substance reaccumulates to synthesize the proteins required for axonal elongation.
Distal Response: Wallerian Degeneration
Distal to the point of injury, the axon and its myelin sheath undergo Wallerian degeneration. Macrophages and Schwann cells clear the axonal and myelin debris, leaving behind an empty endoneurial tube filled with proliferating Schwann cells. This tube is biologically primed to accept regenerating axonal sprouts from the proximal stump.
Axonal Sprouting and Neuroma Formation
Axonal sprouting from the proximal stump may commence within the first 24 hours after injury. Initially, all axonal sprouts are unmyelinated, regardless of whether they originate from a myelinated or unmyelinated parent fiber. A single axonal stump may generate up to 100 individual sprouts.
- Intact Endoneurial Tubes: If the injury is a pure axonotmesis (Sunderland Second Degree) where the endoneurial tube and its contained Schwann cells remain uninterrupted, the sprouts pass readily along their former courses. Following regeneration, the surviving axons accurately reinnervate their previous end organs.
- Disrupted Endoneurial Tubes: If the injury is severe enough to transect the nerve (neurotmesis), the migrating axonal sprouts are barred from their designated endoneurial tubes by hemorrhage, edema, and subsequent scar tissue. These sprouts migrate aimlessly into the epineurial, perineurial, or adjacent extraneural regions, forming a disorganized, painful mass known as a stump neuroma or neuroma in continuity.
- Misdirection: As demonstrated by Cabaud et al., migrating axonal sprouts might enter empty endoneurial tubes of adjacent, unrelated funiculi. They may regenerate through these newly formed pathways only to terminate in myotomal or dermatomal areas entirely different from their original targets, resulting in synkinesis or poor functional recovery.
Clinical Pearl: Axons regenerate as if guided by specific neurotrophic and neurotropic substances contained within the distal nerve tissue. The primary goal of microsurgical neurorrhaphy is to coapt the proximal and distal stumps with minimal tension and maximal fascicular alignment, thereby providing the optimal mechanical and biochemical environment for these sprouts to find their correct distal pathways.
PRINCIPLES OF MICROSURGICAL NEURORRHAPHY
The surgical management of peripheral nerve injuries demands meticulous technique, a deep understanding of biomechanics, and strict adherence to microsurgical principles.
Indications for Surgery
- Primary Repair: Indicated for sharp, clean lacerations (e.g., glass or knife wounds) presenting acutely. Primary repair is ideally performed within the first 72 hours.
- Delayed Primary Repair: Indicated for blunt trauma, crush injuries, or avulsions where the zone of injury is initially indeterminate. The repair is delayed for 2 to 3 weeks to allow the zone of necrosis to demarcate, ensuring resection back to healthy fascicles.
- Secondary Reconstruction: Indicated for late presentations or failed primary repairs, often requiring nerve grafting or nerve transfers.
Patient Positioning and Preparation
- Positioning: The patient must be positioned to allow extensive proximal and distal extension of the surgical incision. For upper extremity nerves (median, ulnar, radial), the arm is typically placed on a radiolucent hand table.
- Tourniquet: A pneumatic tourniquet is essential for maintaining a bloodless field during the initial dissection. However, it should be deflated prior to final nerve coaptation to ensure absolute hemostasis, as postoperative hematoma is profoundly detrimental to nerve regeneration.
- Magnification: Operating loupes (minimum 3.5x to 4.5x) are required for the approach, while an operating microscope is mandatory for the intraneural dissection and suturing.
Surgical Approach: Step-by-Step
1. Exposure and Neurolysis
The incision must be extensile, avoiding crossing flexion creases at 90-degree angles to prevent scar contracture. The nerve is identified in healthy, unscarred tissue both proximally and distally before tracing it into the zone of injury.
* Carefully incise the overlying fascia.
* Mobilize the nerve gently using vessel loops, preserving the mesoneurium wherever possible to maintain the intrinsic blood supply.
2. Preparation of the Nerve Stumps
For a successful repair, the nerve ends must be resected back to healthy, viable tissue.
* Using a fresh diamond knife or a specialized nerve cutting guide, section the nerve stumps sequentially.
* Examine the cross-section under the microscope. Healthy fascicles will "pout" or protrude slightly from the surrounding epineurium. The presence of intraneural scarring or a "meaty" appearance dictates further resection until normal topography is visualized.
3. Fascicular Alignment (Topographic Matching)
Accurate rotational alignment is the most critical step in preventing axonal misdirection. The surgeon utilizes several anatomical landmarks to match the proximal and distal stumps:
* Surface Vessels: Matching the longitudinal epineurial blood vessels.
* Fascicular Map: Comparing the size, shape, and grouping of the fascicles (funiculi) on both stumps.
* Bands of Fontana: These transverse optical bands disappear under tension and can help orient the nerve.
4. Coaptation and Suturing
The repair must be entirely tension-free. If tension is present, a nerve graft (e.g., sural nerve) must be utilized.
* Epineurial Repair: The standard technique for most nerve trunks. Using 8-0 or 9-0 non-absorbable monofilament suture (e.g., Nylon), the external epineurium is approximated. The first two sutures are placed 180 degrees apart to act as traction sutures.
* Group Fascicular Repair: Indicated when distinct motor and sensory fascicular groups can be isolated (e.g., the ulnar nerve at the wrist). Sutures (10-0) are placed through the perineurium of matching fascicular groups, followed by a loose epineurial closure.
Pitfall: Over-tightening the sutures causes buckling of the fascicles, leading to axonal escape and neuroma formation. Sutures should merely approximate the nerve ends; they are not meant to bear mechanical load.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The postoperative management of a peripheral nerve repair is designed to protect the fragile coaptation site while preventing joint contractures.
Phase I: Immobilization (Weeks 0-3)
- The extremity is immobilized in a custom orthoplast splint. The adjacent joints are positioned to minimize tension on the repaired nerve (e.g., slight wrist and elbow flexion for a median nerve repair).
- Strict elevation is maintained to reduce edema.
- Passive range of motion (PROM) of uninvolved digits is encouraged to prevent stiffness.
Phase II: Protected Mobilization (Weeks 3-6)
- The splint is gradually modified to allow controlled, protected motion.
- A hinged brace may be utilized, increasing the permitted extension by 10 to 15 degrees per week.
- Nerve gliding exercises are initiated cautiously to prevent the nerve from adhering to the surrounding surgical bed.
Phase III: Strengthening and Sensory Re-education (Weeks 6+)
- Once full range of motion is achieved and the repair site is deemed mechanically stable, progressive strengthening begins.
- Sensory Re-education: As regenerating axons reach the distal targets (advancing at approximately 1 mm per day, or 1 inch per month), the patient will experience altered sensation (Tinel's sign). A formal sensory re-education program utilizing varying textures, temperatures, and vibrations is critical to help the cerebral cortex reinterpret the new, often imperfect, afferent signals.
- Monitoring: Clinical progress is monitored via advancing Tinel's sign and serial electromyography/nerve conduction studies (EMG/NCS), which typically show nascent motor unit potentials weeks before clinically detectable muscle contraction.
You Might Also Like
