Peripheral Nerve Injuries: Principles of Diagnosis, Microsurgical Repair, and Management of Complex Regional Pain Syndrome
Key Takeaway
Peripheral nerve injuries demand meticulous clinical evaluation and precise microsurgical reconstruction to restore motor and sensory function. This guide provides an evidence-based framework for orthopedic surgeons, detailing nerve anatomy, the Seddon and Sunderland classifications, advanced diagnostic modalities, and step-by-step surgical techniques for primary repair and nerve grafting. Furthermore, it addresses the complex management of postoperative sequelae, including Complex Regional Pain Syndrome (CRPS).
Introduction to Peripheral Nerve Surgery
The management of peripheral nerve injuries represents one of the most technically demanding and biologically complex frontiers in operative orthopaedics. Historically pioneered by luminaries such as Seddon, Sunderland, and Millesi, the evolution of peripheral nerve surgery has transitioned from macroscopic epineurial approximations to precise, topography-matched microsurgical reconstructions.
Successful outcomes in peripheral nerve repair are predicated not merely on technical surgical proficiency, but on a profound understanding of intraneural anatomy, the pathophysiology of Wallerian degeneration, and the biomechanics of nerve regeneration. This masterclass delineates the foundational principles of peripheral nerve injuries, encompassing clinical diagnosis, microsurgical repair techniques, and the management of debilitating sequelae such as Complex Regional Pain Syndrome (CRPS), formerly known as Reflex Sympathetic Dystrophy (RSD).
Anatomy and Physiology of the Peripheral Nerve
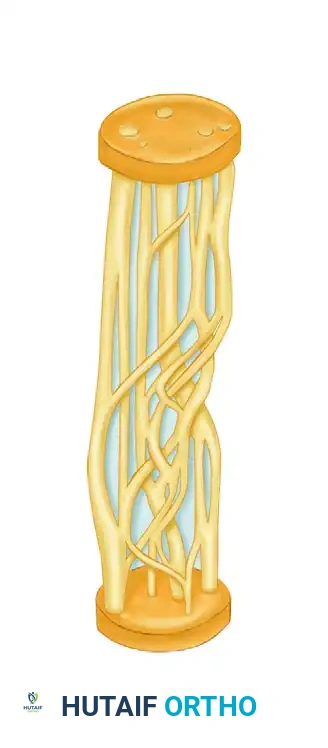
To execute a flawless neurorrhaphy, the operating surgeon must possess an intimate understanding of peripheral nerve microanatomy and the physiological barriers that protect axonal integrity.
Microscopic Architecture
A peripheral nerve is a highly organized, composite structure consisting of neural tissue and specialized connective tissue investments:
* Mesoneurium: The loose areolar tissue surrounding the nerve trunk, analogous to the paratenon. It provides the segmental extrinsic blood supply.
* Epineurium: Divided into the epifascicular epineurium (the outer sheath enveloping the entire nerve) and the interfascicular epineurium (the connective tissue packing between individual fascicles). It cushions the fascicles against compressive and tensile forces.
* Perineurium: A dense, lamellated sheath of specialized perineurial cells surrounding each individual fascicle.
* Endoneurium: The delicate connective tissue matrix within the fascicle, surrounding individual myelinated and unmyelinated axons.
Clinical Pearl: The perineurium is the primary mechanical load-bearing structure of the peripheral nerve and forms the critical blood-nerve barrier. Disruption of the perineurium leads to endoneurial edema, fibroblastic proliferation, and subsequent neuroma-in-continuity formation.
Pathophysiology of Nerve Injury
Following complete transection, the distal nerve segment undergoes Wallerian degeneration. Within 48 to 96 hours, axonal continuity is lost, and Schwann cells begin to phagocytose myelin debris. Concurrently, the proximal stump undergoes retrograde degeneration to the nearest Node of Ranvier, while the neuronal cell body undergoes chromatolysis—a metabolic shift from neurotransmitter production to structural protein synthesis required for axonal regeneration.
Regenerating axons sprout from the proximal stump, guided by neurotropic factors secreted by the distal Schwann cell tubes. Axonal regeneration occurs at an average rate of 1 mm per day (approximately 1 inch per month), though this varies based on patient age, injury proximity to the cell body, and the quality of the surgical repair.
Classification of Peripheral Nerve Injuries
Accurate classification is paramount for determining prognosis and surgical indications. The two most universally accepted systems are those of Seddon (1943) and Sunderland (1951).
Seddon’s Classification
- Neurapraxia: A focal conduction block secondary to localized demyelination (e.g., mild compression). Axonal continuity is preserved. Recovery is spontaneous and complete within days to weeks.
- Axonotmesis: Disruption of the axon and myelin sheath, but preservation of the endoneurial tubes, perineurium, and epineurium. Wallerian degeneration occurs distally. Recovery is typically spontaneous, guided by the intact endoneurial tubes.
- Neurotmesis: Complete transection of the nerve trunk. Spontaneous recovery is impossible without surgical intervention.
Sunderland’s Classification
Sunderland expanded Seddon’s framework into five distinct degrees of injury, correlating with the anatomical structures disrupted:
* First Degree: Equivalent to Neurapraxia.
* Second Degree: Equivalent to Axonotmesis (axon disrupted, endoneurium intact).
* Third Degree: Axon and endoneurium disrupted; perineurium intact. Intrafascicular scarring may impede regeneration, leading to variable recovery.
* Fourth Degree: Axon, endoneurium, and perineurium disrupted; epineurium intact. Results in a neuroma-in-continuity. Surgical excision and repair/grafting are required.
* Fifth Degree: Complete nerve transection (equivalent to Neurotmesis).
Clinical Diagnosis and Evaluation
The clinical evaluation of a peripheral nerve injury requires a meticulous, systematic approach. The diagnosis relies on a combination of motor testing, sensory mapping, and autonomic evaluation.
Sensibility Testing
Sensory recovery is evaluated using a hierarchy of modalities, reflecting the reinnervation of specific end-organs:
* Vibratory Stimuli (Dellon): High-frequency (256 Hz) and low-frequency (30 Hz) tuning forks are utilized to assess the early return of sensation, often preceding the return of moving touch.
* Two-Point Discrimination (Moberg): The gold standard for assessing functional tactile gnosis. Normal static two-point discrimination at the fingertip is 2 to 5 mm.
* Semmes-Weinstein Monofilaments: Provides a quantitative threshold of cutaneous pressure perception.
Autonomic Testing
Autonomic fibers (sympathetic) travel within peripheral nerves. Their disruption leads to a loss of sudomotor (sweating) and vasomotor function in the autonomous zone of the injured nerve.
* Ninhydrin Test (Aschan & Moberg): Detects the presence of amino acids in sweat. A denervated digit will not sweat, yielding a negative (colorless) ninhydrin print.
* Wrinkle Test (O'Riain / Leukens): Immersion of the hand in warm water (40°C) for 30 minutes normally causes skin wrinkling. Denervated skin remains smooth. This is particularly useful in pediatric patients or those unable to cooperate with subjective testing.
Provocative Signs
- Tinel’s Sign: Elicited by gently tapping along the course of the nerve. A tingling sensation radiating into the sensory distribution indicates the presence of regenerating axonal sprouts. The advancing Tinel's sign is a critical clinical marker of regeneration progress.
Surgical Management: Indications and Timing
The timing of surgical intervention is dictated by the mechanism of injury, the condition of the soft tissue envelope, and the type of nerve lesion.
Primary Repair (Acute)
Indicated for sharp, clean lacerations (e.g., glass or knife wounds) presenting within the first 72 hours. Primary repair prevents retraction of the nerve ends and capitalizes on the lack of established scar tissue.
Delayed Primary / Secondary Repair
Indicated for blunt trauma, avulsions, crush injuries, or gunshot wounds. In these scenarios, the extent of intraneural damage is not immediately apparent.
* Protocol: The nerve ends are initially tacked to adjacent fascia to prevent retraction. Definitive repair is delayed for 2 to 3 weeks. By this time, the zone of injury has demarcated, allowing the surgeon to accurately resect the fibrotic neuroma back to healthy, pouting fascicles.
Operative Technique: Microsurgical Neurorrhaphy
The overarching goal of nerve repair is the tension-free, topographically aligned coaptation of healthy nerve ends.
1. Patient Positioning and Preparation
- The patient is positioned supine. A pneumatic tourniquet is applied to provide a bloodless surgical field.
- High-resolution surgical loupes (minimum 3.5x to 4.5x) or an operating microscope are mandatory.
- Microsurgical instrumentation (jeweler's forceps, microscissors, Castroviejo needle holders) must be utilized to prevent iatrogenic crush injury to the delicate epineurium.
2. Surgical Approach and Debridement
- Exposure: The nerve is exposed via extensile incisions, identifying normal anatomy proximally and distally before tracing the nerve into the zone of injury.
- Preparation of Nerve Ends: The scarred nerve ends (neuroma proximally, glioma distally) must be resected sequentially using a fresh scalpel blade or specialized nerve-cutting forceps.
- Visual Endpoint: Resection continues until healthy, distinct fascicles are visualized, and the axoplasm "mushrooms" out of the perineurial tubes.
Surgical Warning: Failure to resect back to healthy, unscarred fascicles is the most common cause of failed nerve repair. Coapting fibrotic nerve ends guarantees a mechanical block to axonal regeneration.
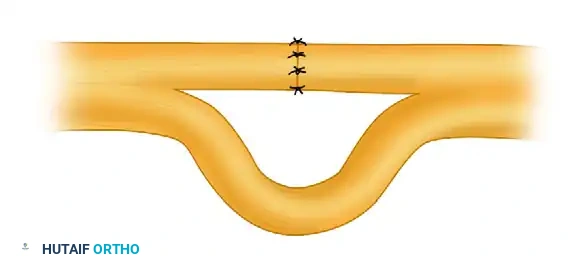
3. Techniques of Coaptation
- Epineurial Repair: The standard technique for most peripheral nerve lacerations. 8-0 or 9-0 nylon sutures are placed through the epifascicular epineurium. Alignment is guided by longitudinal epineurial blood vessels and fascicular topography.
- Group Fascicular Repair: Indicated when a nerve has distinct motor and sensory components that can be anatomically separated (e.g., the ulnar nerve at the wrist). Sutures (10-0 nylon) are placed through the interfascicular epineurium or perineurium to align specific fascicular groups.
4. The Tension-Free Principle and Nerve Grafting
Nerve regeneration is exquisitely sensitive to tension. Tension compromises the intraneural microcirculation, leading to ischemia and dense scar formation.
* The Gap Management Rule: If a primary repair cannot be achieved without tension (typically gaps > 1-2 cm depending on the joint position), an autologous nerve graft is mandatory.
* Donor Nerves: The sural nerve is the workhorse donor, providing up to 30-40 cm of graft material. The medial antebrachial cutaneous (MABC) nerve is an alternative for upper extremity defects.
* Cable Grafting: The donor nerve is cut into segments and interposed between the proximal and distal stumps. The grafts are reversed in orientation to prevent axonal escape through donor nerve branches.
5. Nerve Conduits
For small, non-critical sensory nerve gaps (typically < 3 cm in digital nerves), biological or synthetic nerve conduits (e.g., collagen, polyglycolic acid) may be utilized. These tubes provide a microenvironment that concentrates neurotropic factors and guides axonal sprouting while preventing fibrous tissue ingrowth.
Postoperative Protocol and Rehabilitation
- Immobilization: The extremity is immobilized in a well-padded orthosis for 3 weeks to protect the microvascular anastomosis and prevent tension on the repair site.
- Mobilization: At 3 weeks, progressive, protected range of motion is initiated.
- Sensory Reeducation: As reinnervation occurs, patients often experience hypersensitivity and altered tactile perception. A structured sensory reeducation program (as described by Dellon) is instituted, utilizing varied textures and stimuli to retrain the cerebral cortex to interpret the new afferent signals.
Complications: Complex Regional Pain Syndrome (CRPS)
Historically termed Reflex Sympathetic Dystrophy (RSD) or Causalgia, CRPS is a devastating, sympathetically maintained neuropathic pain syndrome that can complicate even the most technically perfect nerve repair.
Pathophysiology
CRPS is characterized by an aberrant inflammatory response, vasomotor dysfunction, and maladaptive neuroplasticity within the central nervous system. The classic "vicious cycle" involves peripheral nociceptive signals triggering abnormal sympathetic efferent activity, which in turn exacerbates peripheral pain and vascular abnormalities.
Clinical Diagnosis
The diagnosis is primarily clinical, based on the Budapest Criteria. Patients present with:
* Pain: Severe, burning, out of proportion to the inciting injury (allodynia and hyperalgesia).
* Autonomic Dysfunction: Altered skin temperature, abnormal sweating (hyperhidrosis or anhidrosis), and skin color changes (mottled, cyanotic, or erythematous).
* Trophic Changes: Abnormal nail and hair growth, glossy skin, and eventually, joint stiffness and osteopenia (Sudeck's atrophy).
Diagnostic Imaging
- Three-Phase Technetium-99m Bone Scan: As validated by Mackinnon and Kozin, this is a highly specific diagnostic adjunct. In the acute phase of CRPS, the scan demonstrates diffusely increased periarticular uptake in the delayed (third) phase.
Management Strategy
The treatment of CRPS requires an aggressive, multidisciplinary approach. Delay in treatment leads to irreversible joint contractures and chronic disability.
1. Physical Therapy: The cornerstone of treatment. Aggressive, active range of motion, stress loading (scrubbing and carrying exercises), and desensitization.
2. Pharmacotherapy: Gabapentinoids (pregabalin, gabapentin), tricyclic antidepressants (amitriptyline), and short courses of oral corticosteroids in the acute inflammatory phase.
3. Sympathetic Blockade: Stellate ganglion blocks (for the upper extremity) or lumbar sympathetic blocks (for the lower extremity) serve both diagnostic and therapeutic roles. They interrupt the aberrant sympathetic reflex arc, providing a window of pain relief to facilitate intensive physical therapy.
4. Intravenous Regional Blockade (Bier Block): Utilizing agents such as guanethidine or reserpine to deplete peripheral norepinephrine stores, though availability and use vary by region.
5. Neuromodulation: In refractory cases, spinal cord stimulation or peripheral nerve stimulation may be indicated.
Clinical Pearl: Surgical intervention on an extremity actively afflicted by CRPS is strictly contraindicated unless a discrete, surgically correctable trigger (e.g., a compressive neuroma or retained foreign body) is definitively identified. Surgery in the presence of active CRPS will almost universally exacerbate the syndrome.
Conclusion
The operative management of peripheral nerve injuries is a testament to the intersection of meticulous surgical technique and applied neurobiology. By adhering to the principles of tension-free microsurgical coaptation, respecting intraneural topography, and maintaining a high index of suspicion for complications such as CRPS, the orthopaedic surgeon can optimize functional recovery and restore quality of life to patients suffering from these profound injuries.
You Might Also Like