Clinical Diagnosis and Surgical Evaluation of Peripheral Nerve Injuries
Key Takeaway
The accurate clinical diagnosis of peripheral nerve injuries requires a profound understanding of neuroanatomy, motor substitution patterns, and electrodiagnostic timelines. This guide details essential screening protocols, advanced electromyography (EMG) and nerve conduction velocity (NCV) interpretations, autonomic testing, and surgical exploration indications. Mastery of these diagnostic modalities is critical for orthopedic surgeons to differentiate neurapraxia from neurotmesis, optimize surgical timing, and maximize functional recovery in complex extremity trauma.
INTRODUCTION TO PERIPHERAL NERVE INJURIES
Immediately following a high-energy injury to an extremity, the recognition of a peripheral nerve injury presents a formidable clinical challenge. In the acute trauma setting, the patient's pain is often so severe that cooperation is inherently limited. While the preservation of life and limb remains the paramount objective according to Advanced Trauma Life Support (ATLS) protocols, the early identification of neurological deficits is critical for determining the trajectory of surgical management and long-term functional prognosis.
When clinically feasible, rapid and highly specific screening tests must be conducted to detect injuries to the major peripheral nerves. A precise, systematic evaluation requires an intimate knowledge of neuroanatomy, including the course of the nerve, the topographic origin of its motor branches, the specific muscles innervated, and common anatomical variations (e.g., Martin-Gruber or Riche-Cannieu anastomoses).
ACUTE CLINICAL SCREENING PROTOCOLS
In the emergency department or immediate postoperative setting, rapid screening procedures can effectively isolate major nerve deficits.
Upper Extremity Screening
- Ulnar Nerve: Loss of pain perception or two-point discrimination in the volar tip of the little finger is the hallmark of ulnar nerve compromise.
- Median Nerve: Loss of pain perception in the volar tip of the index finger strongly indicates median nerve injury.
- Radial Nerve: The inability to extend the thumb (the "hitchhiker’s sign") typically indicates radial nerve injury.
Lower Extremity Screening
- Sciatic or Tibial Nerve: Loss of pain perception on the plantar aspect (sole) of the foot usually indicates a high sciatic or isolated tibial nerve injury.
- Peroneal Nerve: The inability to actively extend the great toe (extensor hallucis longus) or dorsiflex the foot (tibialis anterior) indicates common peroneal or sciatic nerve injury.
Surgical Warning:
Concomitant injury to tendons or muscle bellies may render motor screening tests entirely invalid. For instance, a severed extensor pollicis longus (EPL) tendon will present as an inability to extend the thumb, mimicking a radial nerve palsy. Always correlate motor deficits with autonomous sensory zone testing.
COMPREHENSIVE MOTOR EVALUATION AND SUBSTITUTION PATTERNS
Evaluation of motor loss is the cornerstone of peripheral nerve assessment. However, this evaluation is only accurate if the examining surgeon can directly palpate or visualize the specific tendon or muscle belly under consideration. Relying solely on the gross analysis of joint movement frequently leads to catastrophic diagnostic errors due to "trick movements" or muscular substitution.
Common Deceptive Substitution Patterns
- Median Nerve (Opponens Pollicis): Opposition of the thumb to the little finger can often be accomplished by patients even when the median nerve supply to the opponens pollicis is completely severed. This is achieved through substitution by the deep head of the flexor pollicis brevis (often ulnar-innervated) or the adductor pollicis.
- Radial Nerve (Wrist Extension): The wrist can be partially extended even when all radial nerve-innervated extensors are paralyzed. This is accomplished by forceful, simple flexion of the fingers, utilizing the tenodesis effect to passively extend the carpus.
- Musculocutaneous Nerve (Elbow Flexion): The elbow can be forcefully flexed even when the musculocutaneous nerve is completely severed and the biceps brachii is paralyzed. This is achieved by substitution of the brachioradialis (innervated by the radial nerve).
To prevent these deceptions, the surgeon must physically palpate the opponens pollicis, the extensor tendons of the wrist, and the biceps tendon during active contraction. While some muscles cannot be easily palpated (e.g., lumbricals, short adductor of the thumb, and volar interossei), there are sufficient superficial muscles supplied by each nerve to allow for an accurate diagnosis.
The Highet Scale for Muscle Grading
A clinical assessment of muscle strength is mandatory for longitudinal tracking. The scale recommended by Highet (closely mirroring the Medical Research Council [MRC] grading system) is universally accepted:
* Grade 0: Total paralysis; no evidence of contractility.
* Grade 1: Muscle flicker or trace of contraction; no joint movement.
* Grade 2: Muscle contraction capable of producing joint movement with gravity eliminated.
* Grade 3: Muscle contraction capable of moving the joint against gravity.
* Grade 4: Muscle contraction against gravity and applied clinical resistance.
* Grade 5: Normal muscle contraction and strength compared with the contralateral side.
ADVANCED ELECTRODIAGNOSTIC STUDIES (EDX)
Electrophysiological studies have been utilized for decades to diagnose, map, and prognosticate a wide variety of neuromuscular disorders. Sequential studies following a peripheral nerve injury provide the orthopedic surgeon with an objective database regarding:
1. Documentation of the injury.
2. Precise anatomical location of the insult.
3. Severity of the injury (neurapraxia vs. axonotmesis/neurotmesis).
4. Recovery patterns and reinnervation timelines.
5. Long-term prognosis.
6. Objective data for impairment documentation (medicolegal and workers' compensation).
7. Underlying pathology.
8. Selection of optimal, fully innervated muscles for future tendon transfer procedures.
The most common electrodiagnostic modalities are Nerve Conduction Studies (NCS) and Electromyography (EMG). These tests are highly complementary, providing distinct information regarding nerve conductivity, myelin integrity, axonal continuity, and muscle recruitment capability.
Nerve Conduction Velocity (NCV)
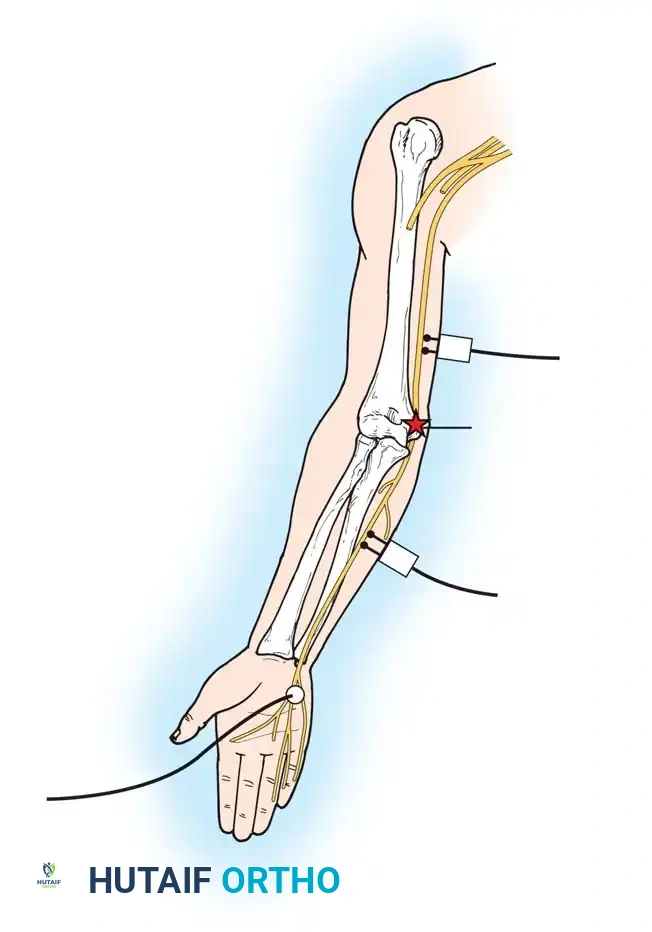
Stimulation of a peripheral nerve by a surface electrode readily evokes a response from the target muscle. This response can be visualized, palpated, and electronically measured. NCV is highly useful shortly after an injury to provide objective evidence of a conduction block; however, it is physiologically impossible to determine the ultimate severity of the insult immediately after the trauma.
The nerve is stimulated proximal to, distal to, and directly across the level of injury, with subsequent distal evoked potentials recorded using needle or surface pick-up electrodes.

Fig. 59-9 Neural injury pattern demonstrating proximal and distal stimulation sites for evaluating conduction blocks and Wallerian degeneration.
The Timeline of Wallerian Degeneration:
Immediately after a severe nerve injury (axonotmesis or neurotmesis), stimulation distal to the insult will still elicit a normal response because the distal axonal segment has not yet degraded. As Wallerian degeneration ensues—typically over 5 to 10 days—there is a progressive reduction in the amplitude and an alteration in the configuration of the evoked potential.
If the insult produced only a temporary physiological block (neurapraxia), conductivity distal to the lesion will remain completely normal even after 10 to 14 days. This finding carries a highly favorable prognosis, indicating that the axon remains intact and spontaneous recovery is expected. Standard NCV techniques include orthodromic motor and antidromic/orthodromic sensory studies. More proximal lesions (e.g., nerve root avulsions) require retrograde techniques such as F-wave studies or Somatosensory Evoked Potentials (SSEPs).
Electromyography (EMG)
Muscle activity observed with a needle pick-up electrode (monopolar, concentric, or single-fiber) placed directly into myotomes innervated by the injured nerve provides crucial diagnostic information. A basic monopolar needle samples approximately eight muscle fibers; by assessing multiple sites, a highly accurate representation of the muscle group is obtained.
The muscle is evaluated in two states: at rest (assessing insertional activity) and during volitional recruitment.
EMG Timeline and Pathological Findings:
* Immediate Post-Injury: Spontaneous rest potentials (denervation potentials) are not observed. Insertional activity remains normal. EMG at this stage is primarily useful for establishing a baseline or delineating pre-existing conditions in litigation cases.
* 10 to 14 Days: Abnormal spontaneous rest potentials known as positive sharp waves begin to appear in myotomes where axonal injury has occurred.
* 14 to 18 Days: Fibrillation potentials appear. These represent the spontaneous depolarization of single, denervated muscle fibers. If any voluntary recruitment is observable, the amplitude is typically reduced due to axonal loss.
* 3 Months: If regeneration is occurring, peripheral neural sprouting reaches the muscle. The EMG will show nascent motor unit potentials, often characterized by a polyphasic configuration.
* 2 to 6 Months: Larger, more stable motor unit potentials are established. Once reinnervation is complete, the motor unit potential configuration returns to a normal-appearing pattern.
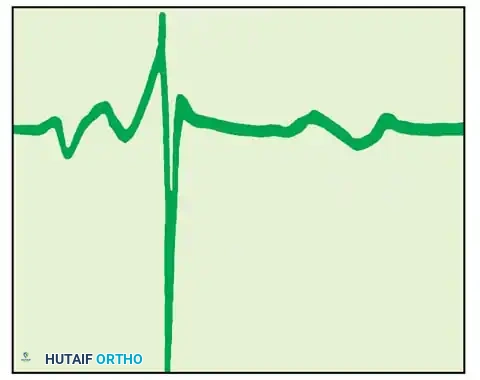
Fig. 59-10 & 59-11: Diagram of EMG tracings depicting normal insertion activity, followed by the appearance of positive sharp waves (10-14 days) and spontaneous denervation fibrillation potentials (14-18 days) consistent with severe axonal injury.
Clinical Pearl:
Denervation potentials (fibrillations and positive sharp waves) will last indefinitely until the muscle either becomes successfully reinnervated or undergoes irreversible fibrotic degeneration (typically 18-24 months post-injury).
PROVOCATIVE AND AUTONOMIC TESTING
The Hoffmann-Tinel Sign
The Tinel sign is elicited by gentle percussion (using a finger or a reflex hammer) along the anatomical course of an injured nerve, progressing in a distal-to-proximal direction. A positive sign is characterized by a transient tingling or "electric shock" sensation felt by the patient in the autonomous sensory distribution of the nerve, rather than at the site of percussion.
A positive Tinel sign is presumptive evidence that regenerating, unmyelinated axonal sprouts are progressing down the endoneurial tubes. As regeneration progresses and the proximal segments of the new axons become myelinated, the positive response fades proximally.
Prognostic Value of the Tinel Sign:
* Sunderland Type 1 (Neurapraxia): No advancing Tinel sign, as Wallerian degeneration does not occur.
* Sunderland Types 2 & 3 (Axonotmesis): A distally advancing Tinel sign is expected (progressing at approximately 1 mm/day or 1 inch/month).
* Sunderland Types 4 & 5 (Neurotmesis): No advancing Tinel sign will be present unless the nerve is surgically repaired, as scar tissue blocks axonal progression.
Note: A few regenerating sensory fibers can produce a positive Tinel sign. Its presence is encouraging but does not guarantee that motor fibers are successfully regenerating or that functional recovery will occur.
Autonomic Evaluation: Sweat and Skin Resistance Tests
Sympathetic nerve fibers are unmyelinated and highly resistant to mechanical stretch and trauma. The preservation of sweating within the autonomous zone of an injured nerve suggests that a complete anatomical transection has not occurred.
- Sweat Tests: Sweating can be observed directly using the +20 lens of an ophthalmoscope. Formal testing includes the iodine-starch test (quinizarin powder turns deep purple in the presence of sweat) or the Ninhydrin print test, which assesses sweat patterns on the digits.
- Skin Resistance Test: Utilizing a Richter dermometer, the surgeon can map autonomic interruption. Denervated skin (anhidrotic) exhibits significantly increased electrical resistance compared to adjacent, normally innervated skin.
SURGICAL EXPLORATION AND INTRAOPERATIVE ASSESSMENT
When clinical and electrodiagnostic evaluations indicate a severe nerve injury failing to spontaneously recover, surgical exploration is mandated.
Indications for Surgery
- Open injuries with sharp transections (immediate exploration and primary repair).
- Closed injuries demonstrating no clinical or EMG evidence of reinnervation by 3 to 6 months.
- An advancing Tinel sign that abruptly halts, indicating a neuroma-in-continuity blocking further regeneration.
Surgical Approach and Positioning
- Positioning: The patient must be positioned to allow full access to the nerve's anatomical course, both proximal and distal to the zone of injury. For upper extremity explorations (e.g., brachial plexus or major peripheral nerves), the arm is typically placed on a radiolucent hand table.
- Tourniquet Use: A pneumatic tourniquet is highly recommended for a bloodless dissection. However, the tourniquet must be deflated prior to intraoperative electrodiagnostic testing, as ischemia rapidly alters nerve conduction.
- Approach: Expansile incisions are utilized. The nerve must always be identified in pristine, unscarred tissue both proximally and distally before tracing it into the zone of injury or neuroma.
Intraoperative Nerve Action Potentials (NAPs)
For neuromas-in-continuity, visual inspection alone is insufficient to determine if functional axons are traversing the scar. Intraoperative NAP monitoring is the gold standard.
* Stimulating electrodes are placed proximal to the neuroma, and recording electrodes are placed distally.
* If a NAP is recorded across the lesion, it proves that viable axons are regenerating through the scar. The appropriate surgical action is external neurolysis to free the nerve from surrounding compression.
* If no NAP is recorded across the lesion by 3 to 6 months post-injury, the neuroma is functionally blocking regeneration. The appropriate surgical action is resection of the neuroma until healthy fascicles are visualized (the "pouting" of fascicles), followed by tension-free repair using autologous nerve grafting (e.g., sural nerve).
POSTOPERATIVE PROTOCOLS
Following peripheral nerve repair or grafting, meticulous postoperative care is essential to prevent disruption of the microvascular anastomosis and the delicate epineurial repair.
* Immobilization: The extremity is immobilized in a well-padded splint for 3 weeks in a position that minimizes tension on the repair site (e.g., slight joint flexion).
* Progressive Mobilization: At 3 weeks, the splint is transitioned to a dynamic orthosis. Supervised, progressive range of motion (ROM) is initiated to prevent joint contractures while avoiding excessive stretch on the regenerating nerve.
* Long-Term Monitoring: Serial clinical examinations (Tinel sign progression, MRC motor grading) and follow-up EMG studies are conducted at 3-month intervals to track the slow process of axonal regeneration and guide eventual rehabilitation or secondary reconstructive procedures (e.g., tendon transfers) if reinnervation fails.
You Might Also Like