Diagnostic Imaging in Spinal Orthopaedics: A Clinical Guide
Key Takeaway
Diagnostic imaging of the spine requires a systematic, evidence-based approach. While plain radiography serves as the initial modality, its correlation with acute pain is often limited. Advanced modalities, including myelography and computed tomography (CT), provide critical anatomical detail for complex pathologies, revision surgeries, and cases involving spinal instrumentation. This guide details the indications, biomechanics, and step-by-step techniques for executing and interpreting these essential diagnostic studies in orthopaedic practice.
DIAGNOSTIC STUDIES IN SPINAL ORTHOPAEDICS
The evaluation of spinal pathology requires a rigorous, multimodal approach to diagnostic imaging. While the clinical examination remains the cornerstone of orthopaedic diagnosis, advanced imaging modalities are essential for confirming anatomical lesions, surgical planning, and navigating the complexities of the revision spine. This comprehensive guide delineates the indications, biomechanical considerations, and technical execution of radiography, myelography, and computed tomography (CT) in the context of modern operative orthopaedics.
RADIOGRAPHY: INDICATIONS AND CLINICAL UTILITY
The simplest, most cost-effective, and readily available diagnostic tests for lumbar or cervical pain are anteroposterior (AP) and lateral radiographs of the involved spinal region. These foundational radiographs frequently demonstrate a high incidence of abnormal anatomical findings; however, their immediate clinical utility in acute settings is highly nuanced.
Evidence-Based Utilization in Acute Low Back Pain
Extensive literature, including seminal studies by Rockey et al. and Liang and Komaroff, has demonstrated that routine spinal radiographs obtained during the initial clinical visit for acute low back pain rarely alter patient management and are generally not cost-effective.
Clinical Pearl: Plain radiographs are recommended only after a structured trial of initial conservative therapy fails, particularly in patients younger than 45 years of age without "red flag" symptoms (e.g., trauma, unexplained weight loss, neurological deficit, or history of malignancy).
Correlation Between Radiographic Findings and Symptomatology
A critical pitfall in spinal diagnostics is the over-interpretation of incidental radiographic anomalies. There is a statistically insignificant correlation between acute back pain and the radiographic presence of:
* Alterations in lumbar lordosis
* Transitional vertebrae (e.g., lumbarization of S1 or sacralization of L5)
* Mild to moderate disc space narrowing
* The disc vacuum sign (Knutsson's phenomenon)
* Claw-type osteophytes (traction spurs)
Furthermore, quantifying disc space narrowing is notoriously difficult and highly subjective, except in previously operated spines or instances of profound, end-stage degenerative disc disease.
In a landmark study of 321 patients, Frymoyer et al. established that the incidence of severe back and leg pain, lower extremity weakness, and radicular numbness only increased significantly when traction spurs or obvious, severe disc space narrowing (or both) were present. Notably, these positive radiographic findings demonstrated no correlation with occupational hazards such as heavy lifting, vehicular exposure, or the use of vibrating equipment.
While conditions such as spondylolysis, spondylolisthesis, and adult degenerative scoliosis show a stronger relationship with back pain, these structural abnormalities are also frequently observed in the radiographs of entirely asymptomatic individuals.
Specialized Radiographic Views
When standard AP and lateral views are insufficient, specialized projections can confirm or refute the initial clinical impression:
- Oblique Views: Highly efficacious for delineating the pars interarticularis. They are the gold standard plain film for identifying spondylolysis (the classic "collar on the Scotty dog") and evaluating spondylolisthesis. However, they offer limited diagnostic value for facet syndrome or hypertrophic osteoarthritis of the lumbar spine. Conversely, in the cervical spine, oblique views are exceptional for outlining hypertrophic osteophytic encroachment within the intervertebral foramina.
- Lateral Flexion and Extension Radiographs: Utilized to assess dynamic segmental instability. However, interpretation requires caution. Hayes et al. demonstrated a wide range of motion in asymptomatic patients, noting that translational movements of 2 to 3 mm are frequent and physiological. Consequently, there is minimal correlation between slight abnormal motion and true pathological instability, as standardized diagnostic thresholds remain controversial. Accurate interpretation relies heavily on patient cooperation, standardized positioning, and reproducible radiographic technique.
- The Ferguson View: A 20-degree caudocephalic AP radiograph. Wiltse et al. demonstrated its immense value in diagnosing the "far out syndrome." This condition involves the extraforaminal compression of the L5 nerve root, which becomes entrapped between a massively hypertrophied transverse process of the fifth lumbar vertebra and the ala of the sacrum.
- Angled Caudal Views: As noted by Abel, Smith, and Allen, these views, when localized to specific areas of clinical concern, can unmask subtle facet or laminar pathologies that are obscured on standard projections.
MYELOGRAPHY: MODERN INDICATIONS AND PHARMACOLOGY
Despite the ubiquity of Magnetic Resonance Imaging (MRI), myelography remains an indispensable diagnostic tool in the orthopaedic surgeon's armamentarium. Its primary value lies in its ability to dynamically evaluate the entire spinal axis for abnormalities and precisely define intraspinal lesions.
Primary Indications for Myelography
Myelography is generally reserved for complex clinical scenarios where MRI is contraindicated, artifact-heavy, or equivocal. Primary indications include:
1. Spinal Instrumentation: Patients with indwelling metallic hardware (e.g., pedicle screws, rods, cages) where MRI is rendered non-diagnostic due to severe susceptibility artifact.
2. Questionable or Conflicting Diagnoses: Scenarios where clinical radiculopathy conflicts with MRI findings.
3. The Revision Spine: Evaluating previously operated spines where differentiating between postoperative epidural fibrosis (scar tissue) and recurrent herniated nucleus pulposus (HNP) is challenging.
4. Severe Deformity: Patients with marked bony degenerative changes or severe scoliosis where MRI may underestimate the degree of foraminal or central canal stenosis.
Surgical Warning: Myelography is an invasive procedure. It should be bypassed if the clinical examination and non-invasive imaging (MRI/CT) are in complete diagnostic agreement.
The Superiority of Post-Myelography CT
The diagnostic yield of myelography is exponentially enhanced by the immediate addition of a post-myelography CT scan. This combination is the gold standard for evaluating complex spinal stenosis.

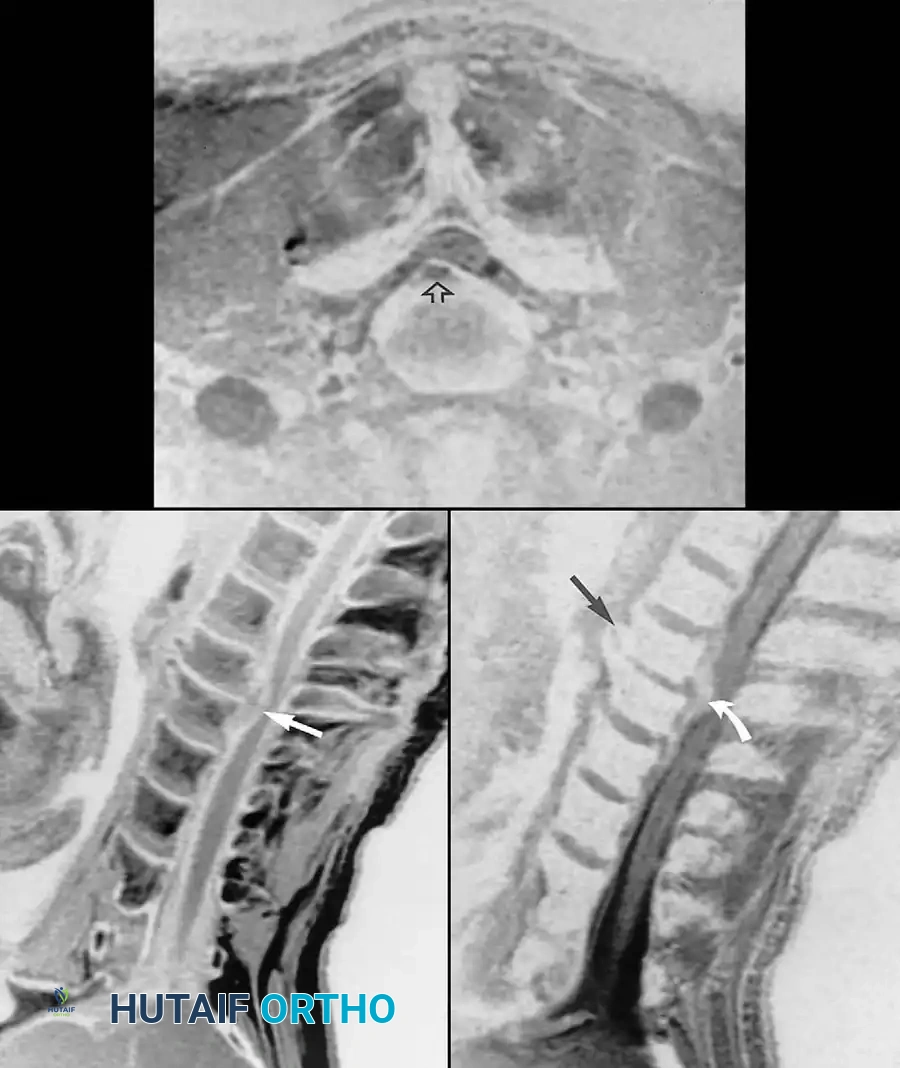
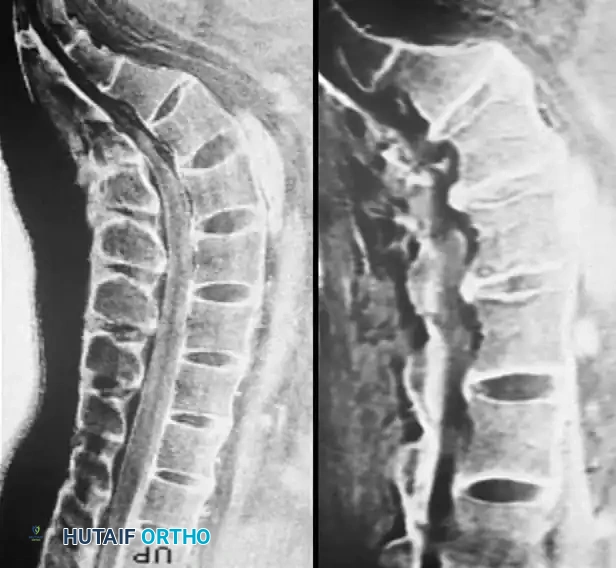
Fig. 39-6 A: MRI Sagittal view of a 45-year-old patient with right C7 radiculopathy. The MRI was inconclusive for a definitive disc herniation.
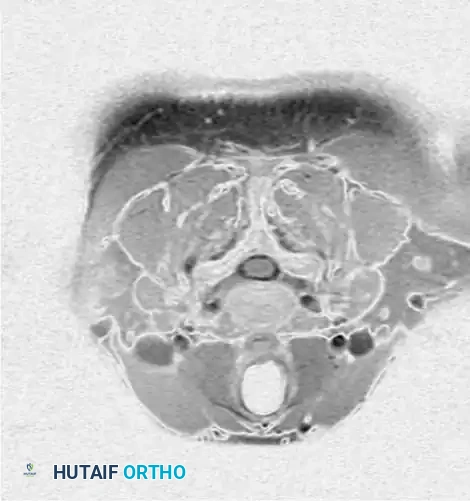
Fig. 39-6 B: MRI Axial view of the same patient, failing to clearly delineate the intraforaminal pathology.
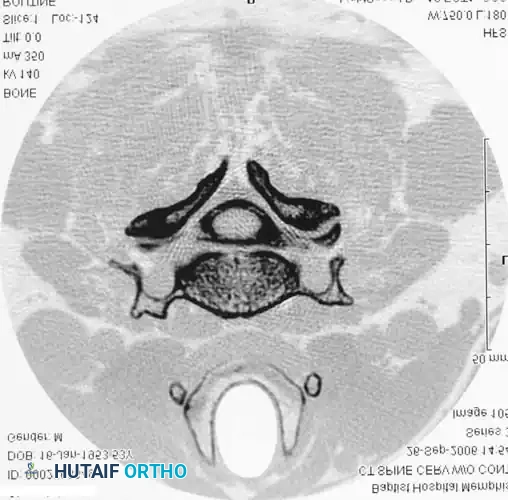
Fig. 39-6 D: Postmyelogram CT axial cut. The introduction of intrathecal contrast provides stark differentiation between the thecal sac, nerve roots, and surrounding osseous structures.
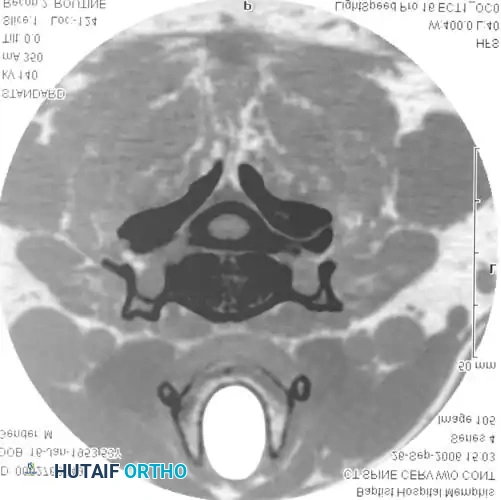
Fig. 39-6 E: Postmyelogram CT clearly revealing a right intraforaminal disc herniation compressing the exiting C7 nerve root, which was missed on the initial MRI.
Studies by Bell et al. found myelography to be more accurate than standard CT scanning for identifying HNP, and slightly more accurate for detecting spinal stenosis. While Szypryt et al. noted myelography is slightly less sensitive than MRI for general spinal abnormalities, its specificity in the presence of hardware is unmatched.
Contrast Agents and Complication Management
Historically, myelography utilized air, oil-based contrast (isophendylate), and early water-soluble agents (metrizamide).
Arachnoiditis: A devastating, severe complication historically associated with the combination of isophendylate (oil contrast) and blood in the cerebrospinal fluid (CSF). Arachnoiditis leads to progressive, intractable pain and, rarely, paralysis. Surgical neurolysis for arachnoiditis yields poor results, with symptoms typically returning within 6 to 12 months.
Today, non-ionic, water-soluble contrast media (e.g., iohexol [Omnipaque] and iopamidol [Isovue-M]) are the absolute standard. Their advantages include:
* Complete absorption by the body (eliminating the painful need to aspirate the contrast post-procedure).
* Enhanced anatomical definition.
* Zero proven association with arachnoiditis, even in the presence of a bloody tap.
Complications of Modern Agents:
While safer, water-soluble agents are meningeal irritants. Complications include headache (<20%), localized pain (8%), nausea (6%), and vomiting (3%). Severe complications like seizures, confusion, aseptic meningitis (0.01%), stroke, or paralysis are exceedingly rare.
Pharmacological Pitfall: The use of phenothiazine anti-nauseants and tricyclic antidepressants lowers the seizure threshold and is strictly contraindicated before and immediately after the administration of water-soluble intrathecal contrast.
Prevention of Complications:
1. Utilize the lowest effective dose of contrast.
2. Ensure aggressive pre- and post-procedural patient hydration.
3. Discontinue phenothiazines and tricyclics 48 hours prior.
4. Maintain the patient's head elevated at least 30 degrees post-procedure to prevent cranial migration of the contrast medium.
MYELOGRAPHY: OPERATIVE TECHNIQUE 39-1
Proper execution of a myelogram requires meticulous technique to ensure diagnostic quality and minimize post-dural puncture headaches (PDPH).
Step 1: Positioning and Preparation
- Place the patient prone on the fluoroscopic table. The use of a radiolucent abdominal pillow is optional but can help reduce lumbar lordosis, opening the posterior interlaminar spaces.
- Prepare and drape the lumbar spine in a standard sterile surgical fashion.
- Ensure adequate pre-procedural hydration to minimize post-myelographic complaints. Heavy sedation is rarely required and should be avoided to maintain patient cooperation.
Step 2: Level Selection and Local Anesthesia
- Determine the needle placement based on the suspected pathological level.
- CRITICAL: Placement of the needle cephalad to the L2-L3 interspace is highly dangerous due to the risk of direct mechanical trauma to the conus medullaris. Always aim for L3-L4 or L4-L5.
- Infiltrate the selected trajectory with a local anesthetic (e.g., 1% Lidocaine) down to the lamina.
Step 3: Needle Selection and Dural Puncture
- Use the smallest gauge needle practical. Smaller needles (22-gauge or 25-gauge) significantly reduce the incidence of PDPH.
- A Whitacre-type (pencil-point) needle is preferred over a cutting (Quincke) needle. The blunt tip and side-port opening separate rather than cut dural fibers, drastically reducing CSF leakage. Note: A 19-gauge introducer needle is usually required to bypass the skin and dense fascia when using a Whitacre needle.
- Advance the needle in the midline to minimize lateral nerve root irritation and inadvertent epidural injection.
- Keep the needle bevel parallel to the long axis of the body (sagittal plane) to spread the longitudinal dural fibers.
- Subarachnoid placement can be facilitated by tilting the patient head-up to increase hydrostatic intraspinal pressure, thereby dilating the thecal sac and minimizing the epidural space.
Step 4: CSF Collection and Test Injection
- Upon puncturing the dura and arachnoid mater, turn the bevel cephalad. A clear, continuous flow of CSF should be observed.
- If indicated, perform manometric studies.
- Collect a volume of CSF equal to the planned contrast injection volume for laboratory evaluation (cell count, differential, protein, glucose).
- Bloody Tap Protocol: If blood is present in the initial CSF return, wait to see if it clears. If it does not clear rapidly, abort the procedure. Reattempt in several days if the patient is asymptomatic and well-hydrated. If CSF flow is minimal/absent despite confirmed placement, maintain a high index of suspicion for a neoplastic block.
- Inject a small test dose of contrast under live fluoroscopy to confirm subarachnoid placement. If a mixed subdural-subarachnoid pattern is seen, adjust the needle depth.
Step 5: Contrast Administration and Imaging
- Once flow is confirmed, slowly inject the contrast material (e.g., 10 to 15 mL of iohexol at a concentration of 170 to 190 mg/mL for an adult lumbar myelogram). Higher concentrations are needed for cervical studies.
- Aspirate occasionally during injection to ensure the needle tip remains subarachnoid.
- Remove the needle.
- Tilt the fluoroscopy table to allow the contrast to flow caudally for optimal visualization of the distal sac and lumbar roots. Obtain spot films in AP, lateral, and bilateral oblique projections.
- A complete lumbar examination must include evaluation up to the T7 level, as lower thoracic lesions can clinically mimic lumbar disc disease.
- For cervical evaluation, tilt the table head-down (Trendelenburg) to allow cranial flow. Crucially, extend the patient's neck and head maximally to physically block the contrast from migrating into the intracranial vault, which can precipitate seizures.
COMPUTED TOMOGRAPHY (CT) IN SPINAL PATHOLOGY
The advent of Computed Tomography (CT) revolutionized the diagnosis of spinal osteoarticular disease. Modern multi-detector CT technology, coupled with advanced computer software, allows for the reformatting of standard axial cuts into sagittal, coronal, and 3D volumetric images with sub-millimeter accuracy.
Axial CT of the cervical spine without contrast, demonstrating the osseous boundaries of the central canal and neuroforamina.
Biomechanics and CT Protocols
Software capabilities now allow surgeons to evaluate the exact Hounsfield Unit (HU) density of a selected vertebra, comparing it against normative population data to provide a numerically reproducible estimate of osteopenia—a critical factor when planning pedicle screw fixation.
When ordering a CT, the surgeon must specify the exact clinical parameters to ensure the region of interest is captured. The standard routine for suspected lumbar disc herniation involves serial axial cuts through the lowest three lumbar intervertebral discs (L3-L4, L4-L5, L5-S1).
Technical Pitfall: If the CT equipment utilizes a tilting gantry, the technician will attempt to align the axis of the cuts parallel to the disc space. However, due to the steep sacral slope, the gantry frequently cannot tilt enough to achieve a perfectly parallel beam through the L5-S1 disc space. This oblique slice angle can artificially distort the appearance of the neuroforamen and must be accounted for during surgical planning.
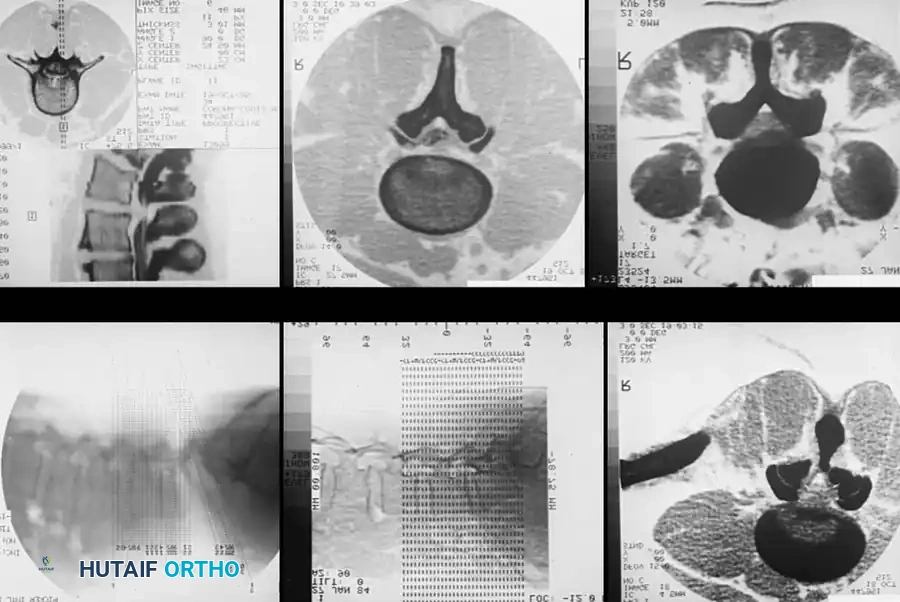
Multi-panel CT imaging demonstrating various reformatted views, essential for comprehensive 3D spatial understanding of spinal pathology prior to surgical intervention.
Classification of Neural Deformation
To standardize the reporting of spinal stenosis and disc herniations on advanced imaging, Beattie et al. developed a highly reproducible classification system for both spinal nerve and thecal sac deformation. This grading scale is essential for academic communication and surgical decision-making.
Table 39-2: Classification for Spinal Nerve and Thecal Sac Deformation
Spinal Nerve Deformation (in Lateral Recess or Intervertebral Foramen)
* Grade 0 (Absent): No visible disc material contacting or deforming the nerve root.
* Grade I (Minimal): Contact with disc material deforming the nerve, but displacement is < 2 mm.
* Grade II (Moderate): Contact with disc material displacing the nerve ≥ 2 mm; the nerve is still clearly visible and not obscured by disc material.
* Grade III (Severe): Contact with disc material completely obscuring the nerve root.
Thecal Sac Deformation (in Vertebral Canal)
* Grade 0 (Absent): No visible disc material contacting or deforming the thecal sac.
* Grade I (Minimal): Disc material is in direct contact with the thecal sac without significant compression.
* Grade II (Moderate): Disc material deforming the thecal sac; however, the anteroposterior (AP) distance of the thecal sac remains ≥ 7 mm.
* Grade III (Severe): Disc material severely deforming the thecal sac; the anteroposterior (AP) distance of the thecal sac is reduced to < 7 mm.
(Adapted from Beattie PF, Meyers SP, Stratford P, et al: Associations between patient report of symptoms and anatomical impairment visible on lumbar magnetic resonance imaging, Spine 25:819, 2000.)
Conclusion
Mastery of spinal diagnostic studies requires more than the ability to read a radiology report; it demands a profound understanding of the biomechanical, pharmacological, and technical principles underlying each modality. By judiciously applying plain radiography, executing flawless myelography when indicated, and leveraging the advanced reformative capabilities of modern CT, the orthopaedic surgeon can navigate the most complex spinal pathologies with precision and confidence.
You Might Also Like