Operative Management of Dupuytren Disease: A Comprehensive Surgical Guide
Key Takeaway
The treatment of Dupuytren disease ranges from nonoperative modalities, such as clostridial collagenase injections and percutaneous needle aponeurotomy, to definitive surgical interventions like selective fasciectomy. Surgical timing is critical; interventions are most successful during the residual phase when cords are mature. Indications typically include metacarpophalangeal contractures exceeding 30 degrees or any proximal interphalangeal joint contracture. This guide details the biomechanics, surgical approaches, and step-by-step techniques required for optimal patient outcomes.
Introduction to Dupuytren Disease Management
The treatment of Dupuytren disease requires a nuanced understanding of palmar fascial anatomy, myofibroblast biology, and the natural history of the disease. Various nonoperative and operative treatment regimens continue to evolve, focusing heavily on the fundamental histopathology of the condition. The primary goal of any intervention is the restoration of hand function through the release or excision of contractile cords, while minimizing complications such as neurovascular injury, hematoma, and postoperative stiffness.
Recent histopathological studies have demonstrated that nodule-derived fibroblast contractile properties are significantly greater than those of cord-derived fibroblasts. Bisson et al. highlighted that both of these pathological fibroblast lines generate substantially more force than normal carpal tunnel ligament fibroblasts. This cellular hyperactivity underscores the progressive nature of the contractures and dictates the timing and modality of treatment.
Nonoperative and Minimally Invasive Modalities
While definitive surgical fasciectomy remains the gold standard for advanced disease, nonoperative and minimally invasive techniques have gained significant traction for specific patient populations, particularly the elderly or those with medical comorbidities precluding extensive surgery.
Corticosteroid Injections
Early intervention in the nodular phase may involve intralesional corticosteroid injections. Ketchum and Donahue demonstrated that after an average of 3.2 injections of triamcinolone acetonide per nodule, 97% of 75 treated hands exhibited softening or flattening of the nodules.
Clinical Pearl: Although complete resolution of Dupuytren disease via corticosteroid injection is exceedingly rare, it is highly effective at halting progression. In clinical studies, only 50% of patients experienced nodule reactivation within three years post-injection.
Clostridial Collagenase Histolyticum (CCH) Injections
Clinical trials evaluating clostridial collagenase injections have demonstrated prompt and impressive release of both metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joint contractures. Badalamente et al. established the safety and efficacy of this enzymatic degradation. In a randomized placebo-controlled study, Hurst et al. reported contracture reduction to 0 to 5 degrees in 77% of MCP joints and 40% of PIP joints across 204 joints receiving collagenase.
Despite these excellent short-term results, recurrence remains a significant concern. Transient side effects are common and include localized swelling, pain, bruising, pruritus, regional lymph node enlargement, and tenderness. Permanent adverse effects are rare but can be catastrophic, notably including flexor tendon ruptures.
Surgical Warning: Collagenase injections are contraindicated in patients on systemic anticoagulation, those with ipsilateral lymphedema, a history of axillary lymph node surgery, or indwelling orthopedic implants in the affected digit. The best results are obtained in patients with a well-defined, palpable cord located safely away from the flexor tendon sheath.
Percutaneous Needle Aponeurotomy (PNA)
Percutaneous needle aponeurotomy has experienced a resurgence in interest since its initial description by Lermusiaux and Debeyre in 1980.
Technique:
A 25-gauge needle mounted on a 10-mL syringe is utilized to mechanically divide the contractile cords. The area is first infiltrated with 1.0 mL or less of 1% lidocaine with 1:100,000 epinephrine. Multiple areas of the contractile cord are sectioned in the palm and fingers using the bevel of the needle.
Outcomes and Limitations:
PNA provides satisfactory short-term results. In a cohort of 74 releases followed for 33 months, there was an 88% reduction in MCP joint contractures and a 46% reduction in PIP joint contractures. However, the recurrence rate is exceptionally high—reported at 65% (defined as a loss of 30 degrees or more from the immediate postoperative correction).
Pitfall: Special care must be taken to avoid sensory nerve injury, particularly in the distal palm where the neurovascular bundles are displaced superficially by spiral cords. Digital nerve stretch injury and direct needle trauma remain significant concerns. PNA is best reserved for elderly patients, those desiring minimally invasive options, and those who accept that repeat procedures will likely be necessary.
Principles of Surgical Intervention
Surgical treatment is technically easier when joint contractures are less severe; however, operating too early in the disease process is a common pitfall. Minor contractures are typically encountered early in the disease progression (the proliferative phase). During this stage, the cellular process is highly active, planes between normal and abnormal tissue are ill-defined, and surgical trauma can paradoxically accelerate the disease process, leading to aggressive recurrence or flare reactions.
Indications and Timing
Ideally, patients should be operated on during the residual phase, when the diseased tissues have matured into dense, well-defined cords, and the tendency for surgical trauma to incite a flare is minimized.
Standard indications for surgical intervention include:
* MCP Joint Contracture: 30 degrees or more.
* PIP Joint Contracture: 15 degrees or more (PIP contractures are notoriously difficult to correct and warrant earlier intervention than MCP contractures).
Indications and timing must also account for the patient's functional disability, the presence of degenerative joint disease, and predisposing factors for poor outcomes (Dupuytren diathesis), rather than relying solely on absolute goniometric measurements.
Pathoanatomy: The Spiral Cord
Understanding the pathoanatomy of the spiral cord is paramount to safe surgical dissection. The spiral cord is responsible for PIP joint contracture and is formed by the diseased progression of four normal anatomical structures: the pretendinous band, the spiral band, the lateral digital sheet, and Grayson's ligament.
As the spiral cord contracts, it draws the neurovascular bundle proximally, centrally, and superficially, placing the digital nerve at extreme risk during surgical exposure.
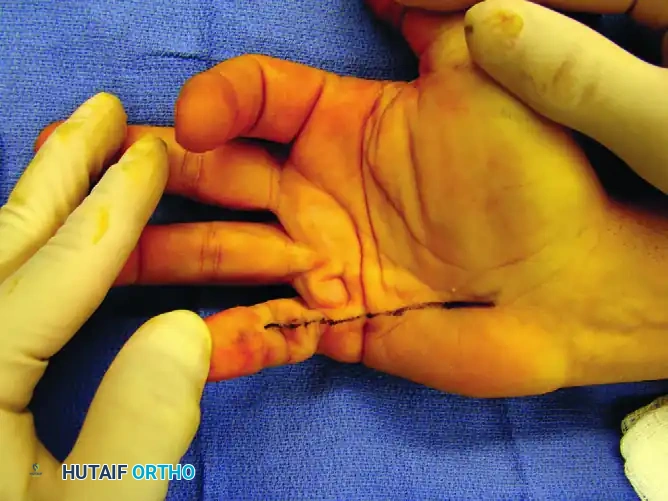
FIGURE 75-5 A: Clinical presentation of a severe spiral cord contracture involving the little finger.
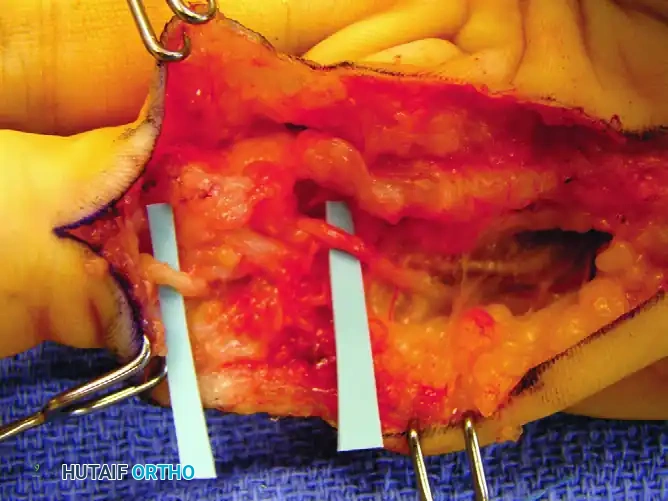
FIGURE 75-5 B: Intraoperative exposure revealing the dense nodule and the spiral cord displacing the neurovascular structures.
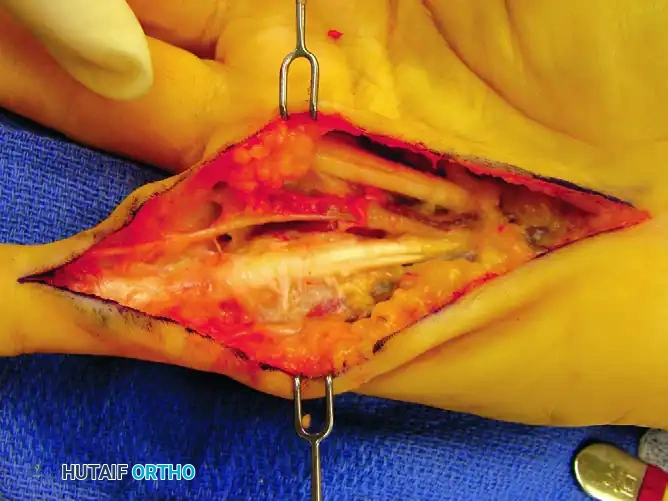
FIGURE 75-5 C: Following meticulous surgical resection of the cord, the ulnar digital nerve returns to its normal anatomical position.
Surgical Approaches and Incision Design
The choice of incision is critical. It must provide wide exposure of the diseased fascia, allow for the management of contracted skin, and prevent postoperative scar contracture.

FIGURE 75-6 A: Multiple Z-plasties can be used to provide exposure and convert longitudinal skin contractures into zigzag closures. Only one extension typically needs to be made into the palm.

FIGURE 75-6 B: When joint or skin contracture is not a major problem, a standard zigzag pattern (Bruner incision) can be used for exposure.

FIGURE 75-6 C: The shaded area demonstrates the extent of possible skin undermining required to separate the dermis from the underlying diseased fascia.

FIGURE 75-6 D: When only the palm is involved, transverse incisions placed within the natural palmar creases provide excellent cosmetic and functional results.

FIGURE 75-6 E: Initiation of the V-Y plasty method of Mukerjea for lengthening contracted palmar skin.

FIGURE 75-6 F: Completion of the V-Y plasty, demonstrating the advancement of the skin flap to accommodate the newly extended digit.
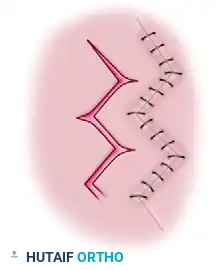


Various alternative incision patterns and skin flap designs utilized to optimize exposure while minimizing tension on the palmar skin during closure.
When tightness of the palmar skin limits extension of a finger, or when there is a significant PIP joint contracture, a midline longitudinal incision converted to appropriate Z-plasties is highly indicated. Dissection is made significantly easier by the use of loupe magnification (minimum 2.5x to 3.5x), and meticulous hemostasis is required to avoid neurovascular injury.
Definitive Surgical Procedures
Surgical procedures commonly used in treating Dupuytren contracture include:
1. Subcutaneous fasciotomy
2. Partial (selective) fasciectomy
3. Complete fasciectomy
4. Fasciectomy with skin grafting (Dermofasciectomy)
5. Amputation
6. Joint resection and arthrodesis
The appropriate procedure depends on the degree of contracture, the nutritional status of the palmar skin, the presence of bony deformities, and the patient’s age, occupation, and general health.
1. Subcutaneous Fasciotomy
The least extensive procedure, subcutaneous fasciotomy, is commonly used for elderly patients who are not concerned with the cosmetic appearance of the disease or in patients with poor general health who cannot tolerate prolonged anesthesia. The results are best in the residual phase when dense, mature cords are present. In subcutaneous tenotomy, the pretendinous cords are simply divided blindly or under minimal exposure to correct MCP joint contractures.
2. Partial (Selective) Fasciectomy
Partial fasciectomy is the most frequently performed operation for Dupuytren disease. It is usually indicated when only the ulnar one or two digits are involved. Postoperative morbidity is significantly less, and complications are fewer than after complete fasciectomy.
In this operation, only the mature deforming tissue is excised. It is important to note that biochemically or microscopically involved fascia may not be clinically apparent at the time of surgery and is left behind. Consequently, the rate of recurrence after partial fasciectomy is approximately 50%; however, the need for a secondary surgical procedure is only 15%.
Skoog described a specific variant of selective fasciectomy in which only the pretendinous fibers of the palmar fascia are excised. Skoog identified a definite surgical plane between the pretendinous longitudinal fibers and the transverse palmar ligament. By preserving the transverse palmar ligament, the normal architecture of the palm is maintained.
3. Complete Fasciectomy
Complete fasciectomy is rarely, if ever, indicated in modern orthopedic practice. It is frequently associated with severe complications, including massive hematoma, profound joint stiffness, and delayed wound healing. Furthermore, despite its radical nature, it does not completely prevent the recurrence of the disease.
4. Fasciectomy with Skin Grafting (Dermofasciectomy)
Dermofasciectomy may be indicated for young patients with a poor prognosis due to a strong Dupuytren diathesis (characterized by early onset, aggressive recurrence, epilepsy, alcoholism, or ectopic deposits like Ledderhose or Peyronie disease). The skin and underlying abnormal fascia are excised en bloc, and a full-thickness or split-thickness skin graft is applied. Recurrence is exceptionally rare beneath a full-thickness skin graft.
5. Salvage Procedures: Amputation and Arthrodesis
Amputation is rarely necessary but may be indicated if flexion contracture of the PIP joint—especially of the little finger—is severe, long-standing, and cannot be corrected enough to make the finger useful. A 40-degree flexion contracture is usually tolerated fairly well, but contractures exceeding 70-80 degrees often result in a functionally useless digit. The skin from the filleted finger can be folded into the palm as a pedicle flap to cover palmar skin defects.
Alternatively, joint resection and arthrodesis can be performed. This results in a shortened, fused finger but avoids the potential for recurrent PIP joint contracture and eliminates the risk of a painful amputation neuroma.
Management of the Proximal Interphalangeal (PIP) Joint
Incomplete extension of the PIP joint following fasciectomy is a common and frustrating complication. Projections of isolated cords passing volar to the rotation axis of the PIP joint are frequent causes of residual flexion deformity. These problematic cords often insert laterally onto the flexor tendon sheath or the middle phalanx.
Dissection of faintly detectable deforming cords intimately associated with the skin may be required. If significant residual PIP flexion contracture persists after complete fascial excision, a volar joint capsulotomy may be necessary.
Evidence-Based Outcome: According to Weinzweig, Culver, and Fleegler, PIP joint flexion contractures of more than 60 degrees are correctable to only about 50% of the existing contracture, regardless of whether a concomitant PIP joint capsulotomy is performed. Furthermore, capsulotomy patients experienced an average 16-degree loss of preoperative flexion compared with an 8-degree loss in the non-capsulotomy group. Therefore, capsulotomy should be approached with caution.
Step-by-Step Surgical Technique
🔪 Surgical Technique 75-1: Subcutaneous Fasciotomy (Luck)
1. Incision and Puncture:
Using a pointed scalpel (No. 11 blade), make precise skin puncture wounds on the ulnar side of the diseased palmar fascia at the following levels:
* Just distal to the apex of the palmar fascia between the thenar and hypothenar eminences.
* At or near the level of the proximal palmar crease.
* At the level of the distal palmar crease.
Warning: Digital nerves are highly susceptible to transection at the distal palm where they become superficial and intertwine with diseased tissue.2. Instrument Insertion:
Insert a small tenotomy knife or a Luck fasciotome (which resembles a myringotome) with its blade parallel to the palm through each puncture wound. Pass the cutting instrument across the palm beneath the skin but strictly superficial to the fascia.3. Cord Division:
Turn the edge of the blade dorsally toward the palmar fascia. Extend the patient's fingers to place the involved tissue under maximum tension. Carefully divide the fascial cords by pressing the blade onto the tense cords using gentle pressure or a slight rocking motion. Never use a sawing motion. As the cord is divided, the surgeon will feel the gritty, firm resistance suddenly yield, indicating complete passage through the diseased fascia.4. Skin Release:
Return the fasciotome blade to a plane parallel with the skin and sweep it to free the dermis from the underlying fascia. The corrugated skin, although very thin, can be safely undermined and released with minimal risk of skin necrosis.5. Digital Fasciotomy:
In the fingers, subcutaneous fasciotomy is safe only for a fascial cord located strictly in the midline. Insert the blade through a puncture wound adjacent to the cord and divide it obliquely. For laterally placed cords, a short longitudinal incision must be used to divide the diseased segment under direct vision to protect the neurovascular bundle. Enucleate larger nodules in both the fingers and palm under direct vision.
Postoperative Care and Rehabilitation
Meticulous postoperative care is as critical as the surgical execution. Immediately following surgery, a bulky, compressive pressure dressing is applied and maintained for 24 to 48 hours to prevent hematoma formation—the most common complication leading to delayed healing and stiffness.
Following the removal of the pressure dressing, a lighter dressing is applied, and active range-of-motion (ROM) therapy is initiated immediately. A custom-molded thermoplastic extension splint is fabricated by a certified hand therapist. The splint is typically worn continuously (except during exercise) for the first 3 weeks, and then transitioned to nighttime wear for an additional 3 to 6 months to prevent the recurrence of flexion contractures as the scar tissue matures. Aggressive passive stretching should be avoided early in the postoperative phase to prevent inflammatory flare reactions.
You Might Also Like
