Treatment of Enchondroma, Bone Cyst, and Giant Cell Tumor of the Distal Upper Extremity
Introduction and Epidemiology
As academic orthopedic surgeons and musculoskeletal oncologists, a nuanced understanding of benign and locally aggressive bone lesions of the distal upper extremity is imperative for optimal patient management. This comprehensive review focuses on the pathophysiology, clinical presentation, diagnostic evaluation, and surgical management of enchondroma, unicameral bone cyst (UBC), and giant cell tumor (GCT) of bone. These lesions represent a diverse spectrum of osseous pathology affecting the hand and wrist, each with distinct biological behaviors, genomic profiles, and clinical implications. A precise diagnosis and tailored management strategy—ranging from active surveillance to extended intralesional curettage with structural reconstruction—are paramount to preventing catastrophic structural failure, minimizing local recurrence, and preserving complex hand and wrist kinematics.
Enchondroma Epidemiology and Pathogenesis
Enchondromas are the most prevalent benign cartilaginous neoplasms of bone and represent the most frequently encountered primary osseous neoplasm in the hand. While they predominantly affect the small tubular bones of the hand (phalanges and metacarpals), they are also observed in the distal radius and ulna.
Pathogenetically, enchondromas are thought to originate from remnants of the epiphyseal growth plate (hyaline cartilage rests) that fail to undergo normal endochondral ossification and instead proliferate within the medullary cavity. Recent molecular advances have identified somatic mutations in the isocitrate dehydrogenase 1 and 2 (IDH1 and IDH2) genes in a significant proportion of solitary and syndromic enchondromas. Most solitary enchondromas remain asymptomatic and are identified incidentally. However, cortical expansion and endosteal scalloping can lead to structural compromise, presenting clinically as a pathologic fracture. The risk of malignant transformation of a solitary enchondroma into a secondary chondrosarcoma is exceedingly low (<1%). Conversely, syndromic presentations such as Ollier disease (multiple enchondromatosis) and Maffucci syndrome (multiple enchondromatosis associated with soft tissue hemangiomas) carry a substantially higher risk of malignant transformation (up to 25-30% and 100% respectively over a lifetime), mandating rigorous longitudinal surveillance.
Unicameral Bone Cyst Epidemiology and Pathogenesis
Unicameral bone cysts (UBCs), or simple bone cysts, are benign, fluid-filled intraosseous cavities lined by a thin fibrovascular membrane. They are most frequently diagnosed in the first two decades of life, predominantly affecting the metaphysis of long bones. While the proximal humerus and femur are classic locations, UBCs are occasionally encountered in the distal radius and, more rarely, the short tubular bones of the hand.
The precise etiology remains debated, but the prevailing theory implicates a localized disturbance in venous drainage leading to increased intramedullary pressure, subsequent transudation of fluid, and osteoclastic bone resorption. Cytokine analysis of cyst fluid often reveals elevated levels of prostaglandins, interleukin-1 (IL-1), and other bone-resorbing factors. Clinically, UBCs in the distal upper extremity frequently present following a minor traumatic event resulting in a pathologic fracture. Active cysts are located adjacent to the physis and possess growth potential, whereas latent cysts migrate diaphyseally as normal bone forms between the cyst and the growth plate.
Giant Cell Tumor Epidemiology and Pathogenesis
Giant cell tumor (GCT) of bone is a locally aggressive primary bone neoplasm characterized by a highly vascularized network of proliferating mononuclear stromal cells interspersed with numerous multinucleated osteoclast-like giant cells. GCTs typically arise in the epiphyseal-metaphyseal regions of long bones following skeletal maturity. The distal radius is the third most common site overall (following the distal femur and proximal tibia), accounting for approximately 10-12% of all GCTs.
Pathogenetically, the neoplastic mononuclear stromal cells express high levels of Receptor Activator of Nuclear factor Kappa-B Ligand (RANKL). This drives the recruitment, differentiation, and activation of the non-neoplastic, multinucleated osteoclast-like giant cells via the RANK/RANKL signaling pathway, resulting in aggressive, localized osteolysis. Furthermore, somatic mutations in the H3F3A gene (specifically the G34W mutation) are found in over 90% of GCTs, serving as a highly specific diagnostic marker. While histologically benign, GCTs exhibit aggressive local destruction and possess a 2-5% rate of benign pulmonary metastasis. Clinically, patients present with insidious, progressive pain, localized swelling, and restricted joint range of motion.

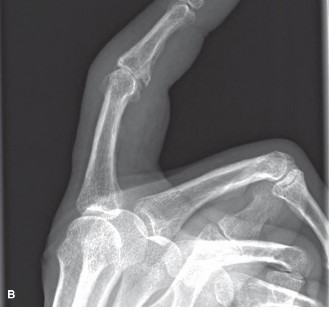
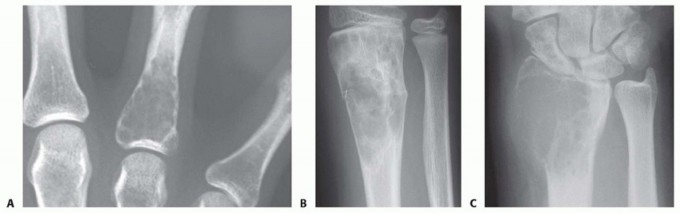
FIG 1 • A. Enchondroma of the proximal phalanx demonstrating characteristic central chondroid calcification. B. Unicameral bone cyst of the distal radius showing a well-defined, centrally located radiolucency. C. Giant cell tumor of the distal radius illustrating an eccentric, expansile, lytic lesion extending to the subchondral bone.
Surgical Anatomy and Biomechanics
A rigorous understanding of regional surgical anatomy and the biomechanical consequences of osteolytic lesions is fundamental for executing effective surgical interventions in the distal upper extremity. The intricate architecture of the hand and wrist demands meticulous technique to preserve critical neurovascular structures, tendon excursion, and articular congruity.
Anatomy of the Phalanges and Metacarpals
The phalanges and metacarpals are tubular bones subjected to complex loading patterns during prehension. Their diaphyseal cortices are relatively thin; thus, expansile intralesional masses rapidly compromise structural integrity. Surgical approaches must navigate the complex dorsal extensor apparatus (central slip, lateral bands) and the volar flexor tendon sheaths (A-pulleys). The neurovascular bundles (proper palmar digital arteries and nerves) run volarly and laterally, demanding precise dissection. Biomechanically, these bones endure significant bending and torsional moments during power grip and pinch. A lesion occupying more than 50% of the cortical diameter acts as a massive stress riser, exponentially increasing the risk of mechanical failure under physiologic loads.
Anatomy of the Distal Radius and Ulna
The distal radius forms the primary load-bearing articulation of the radiocarpal joint, transmitting approximately 80% of axial forces across the wrist. It features complex articular geometry, including the scaphoid and lunate fossae, and the sigmoid notch for the distal radioulnar joint (DRUJ). Surgical approaches must navigate the six dorsal extensor compartments, the superficial radial nerve, the median nerve, and the radial and ulnar arteries.
GCTs of the distal radius characteristically extend to the subchondral bone plate, posing a severe challenge for achieving adequate oncologic margins while simultaneously preserving joint viability. The subchondral collapse can lead to rapid radiocarpal arthrosis. UBCs in the distal radial metaphysis weaken the metaphyseal flare, predisposing the patient to insufficiency fractures that can alter radial inclination, volar tilt, and ulnar variance if not appropriately managed.
Biomechanical Considerations
Osteolytic lesions alter the moment of inertia and load-bearing capacity of the host bone.
* Enchondromas: In the small tubular bones, endosteal scalloping and cortical thinning create significant stress concentrators. Torsional forces are particularly poorly tolerated by tubular bones with structural defects, frequently resulting in spiral or oblique pathologic fractures.
* Unicameral Bone Cysts: The fluid-filled cavities of UBCs offer zero resistance to applied loads. The remaining thin cortical shell is highly susceptible to bending forces. The "fallen leaf" sign on radiographs represents a cortical fragment that has fractured and fallen into the fluid-filled cyst cavity.
* Giant Cell Tumors: The aggressive lytic nature of GCTs leads to extensive trabecular and cortical destruction. Biomechanically, the loss of subchondral support threatens the articular cartilage. Reconstruction must not only fill the void but also provide immediate structural support to withstand compressive joint reactive forces, often necessitating the use of polymethylmethacrylate (PMMA) or structural allografts combined with rigid internal fixation.
Indications and Contraindications
Surgical decision-making is predicated on a comprehensive assessment of symptomatology, biological aggressiveness, radiographic features, and the biomechanical stability of the affected segment.
Indications for Surgical Intervention
- Pathologic Fracture: The most frequent indication for operative management of enchondromas and UBCs. Surgical strategy must address both fracture stabilization and definitive tumor management (curettage and grafting) to prevent nonunion or recurrence.
- Impending Pathologic Fracture: Prophylactic intervention is indicated for lesions demonstrating high mechanical risk. Criteria include >50% cortical diameter involvement, cortical thickness <2mm, or progressive pain with weight-bearing or functional use.
- Progressive Pain: Mechanical pain suggests microfracture or impending structural failure. Biological pain (rest pain, night pain) raises suspicion for aggressive behavior or malignant transformation.
- Progressive Lesion Growth: Serial radiographic enlargement, particularly in skeletally mature patients, warrants intervention.
- Giant Cell Tumor: Due to their locally aggressive nature, high recurrence rate, and metastatic potential, virtually all GCTs require surgical intervention.
- Diagnostic Uncertainty: Any lesion exhibiting atypical radiographic features (e.g., cortical breakthrough, periosteal reaction, soft tissue extension, ill-defined zone of transition) requires biopsy and definitive surgical management.
Contraindications for Surgical Intervention
- Asymptomatic, Stable Lesions: Small, incidentally discovered enchondromas with classic radiographic features (stippled calcification, narrow zone of transition) in the absence of mechanical compromise are best managed with active surveillance.
- Resolved Pathologic Fracture: If a minor pathologic fracture through a stable enchondroma or UBC has healed spontaneously with acceptable alignment, continued observation may be appropriate.
- Severe Medical Comorbidities: In patients with prohibitive surgical risk, non-operative management with customized orthoses or systemic therapies (e.g., denosumab for GCT) may be employed.
Operative versus Non-Operative Indications
| Lesion Type | Operative Indications | Non-Operative Indications |
|---|---|---|
| Enchondroma | Pathologic fracture, impending fracture, progressive pain, diagnostic uncertainty (suspicion of secondary chondrosarcoma). | Asymptomatic incidental finding, classic radiographic features, stable size on serial imaging. |
| Unicameral Bone Cyst | Pathologic fracture, high risk of impending fracture (large metaphyseal cyst), failure of non-operative management. | Asymptomatic small cysts, latent cysts migrating diaphyseally, resolved fractures with progressive ossification. |
| Giant Cell Tumor | All confirmed GCTs require surgical management (extended curettage vs. resection). | None for definitive cure. Systemic RANKL inhibitors (denosumab) are reserved for unresectable disease, severe surgical morbidity, or as a neoadjuvant downstaging agent. |
Pre Operative Planning and Patient Positioning
Thorough preoperative planning, including advanced cross-sectional imaging and meticulous biopsy technique, is critical to achieving local control and optimizing reconstructive outcomes.
Diagnostic Workup and Staging
- Radiographs: Orthogonal plain radiographs remain the cornerstone of initial evaluation. They define the lesion's location (epiphyseal, metaphyseal, diaphyseal), margins (zone of transition), matrix production (chondroid calcifications), and effect on host bone (expansion, cortical destruction).
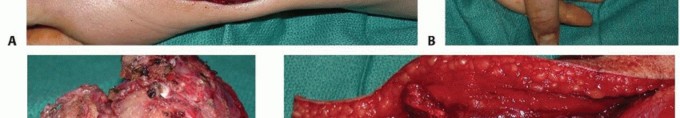
FIGURE 2: Plain radiographs demonstrating an enchondroma of the proximal phalanx, characterized by a well-circumscribed lytic lesion with internal chondroid calcifications (rings and arcs).
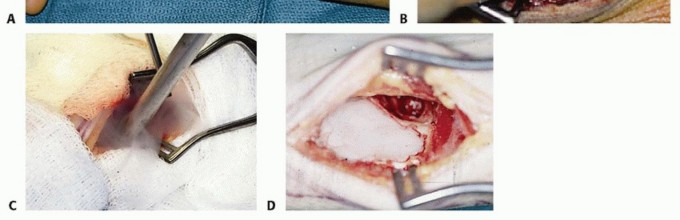
FIGURE 3: Anteroposterior and lateral radiographs of the distal radius revealing a unicameral bone cyst with evidence of cortical thinning, expansion, and a narrow zone of transition.
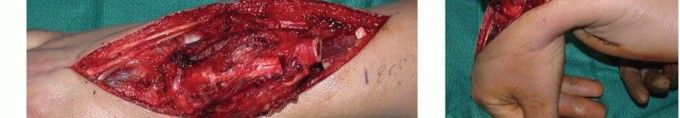
FIGURE 4: Radiographic appearance of a giant cell tumor of the distal radius, showing an eccentric, expansile, lytic lesion extending to the subchondral bone without a sclerotic rim (Campanacci Grade II). - Computed Tomography (CT): CT is unparalleled for assessing cortical integrity, subtle endosteal scalloping, subchondral bone stock, and the presence of occult microfractures. It is essential for preoperative templating of internal fixation and structural allografts.
- Magnetic Resonance Imaging (MRI): MRI defines the intraosseous and extraosseous extent of the tumor. It is critical for evaluating neurovascular bundle proximity, joint involvement, and "skip" metastases. Enchondromas typically show lobulated hyperintensity on T2-weighted images. GCTs exhibit heterogeneous intermediate signal on T1 and T2, often with secondary aneurysmal bone cyst (ABC) components (fluid-fluid levels).

FIGURE 5: MRI (T2-weighted sequence) demonstrating the extent of a distal radial unicameral bone cyst, highlighting its hyperintense fluid-filled nature and precise anatomical boundaries. - Biopsy: A definitive tissue diagnosis is mandatory for GCTs and atypical lesions prior to definitive resection. Core needle biopsy or open incisional biopsy must be meticulously planned. The biopsy tract must be placed in-line with the planned definitive surgical incision so the entire tract can be excised en bloc to prevent tumor seeding. Meticulous hemostasis is critical to prevent hematoma-associated tumor dissemination.
Anesthesia and Patient Positioning
- Anesthesia: A combination of regional anesthesia (e.g., supraclavicular or axillary brachial plexus block) and general anesthesia is optimal. Regional blocks provide profound intraoperative muscle relaxation, excellent postoperative analgesia, and vasodilation that can improve graft incorporation.
- Patient Positioning: The patient is positioned supine with the operative extremity on a radiolucent hand table. A pneumatic tourniquet is applied high on the brachium to ensure a bloodless field, which is an absolute requirement for adequate visualization during intralesional curettage and the application of chemical/thermal adjuvants.
- Fluoroscopy Setup: An intraoperative C-arm must be positioned to allow unhindered orthogonal views of the wrist and hand. Fluoroscopy confirms complete tumor evacuation, guides adjuvant application, and ensures precise hardware placement.
Detailed Surgical Approach and Technique
The surgical objective is to achieve complete oncologic clearance while maximizing functional preservation. For benign and locally aggressive lesions, extended intralesional curettage remains the gold standard.
General Principles of Extended Intralesional Curettage
- Adequate Cortical Window: The cortical window must be large enough to visualize the entire cavity. "Blind" curettage inevitably leaves residual tumor.
- Gross Curettage: Meticulous removal of all macroscopic tumor using sharp curettes of varying sizes and angles.
- High-Speed Burring: This is the most critical step in extended curettage. A high-speed burr (diamond or fluted) is used to systematically remove 1-2 mm of reactive bone from the entire cavity wall, breaking down bony ridges and exposing occult tumor lobules.
- Adjuvant Therapy: Chemical or physical adjuvants are utilized to achieve chemical or thermal necrosis of microscopic residual disease extending into the cancellous bone.
- Phenol (89%): A chemical cauterant that denatures proteins. It is applied carefully with cotton applicators, left for 1-2 minutes, and then thoroughly neutralized and irrigated with 70% isopropyl alcohol and saline.
- Cryotherapy (Liquid Nitrogen): Induces cell death via intracellular ice crystal formation and microvascular thrombosis. Requires careful protection of adjacent soft tissues to prevent iatrogenic necrosis.
- Argon Beam Coagulation: Delivers radiofrequency energy via ionized argon gas, producing a uniform depth of thermal necrosis (2-3 mm) without direct tissue contact.
- Reconstruction: The resultant cavitary defect must be reconstructed to restore biomechanical stability using autograft, allograft, bone graft substitutes, or PMMA.
Enchondroma of the Hand Phalanges and Metacarpals
Surgical Approach
- Phalanges: A dorsal longitudinal incision, splitting the extensor tendon longitudinally or elevating it via a chamfer incision, provides excellent exposure. Lateral approaches are utilized for lateral lesions, taking care to protect the neurovascular bundle.
- Metacarpals: Dorsal longitudinal incisions over the affected metacarpal. The internervous plane between the extensor digitorum communis tendons is utilized.
Technique
- Cortical Windowing: A small osteotome or high-speed burr is used to create an oval cortical window.

FIGURE 6: Intraoperative view showing an adequate cortical window created in the proximal phalanx for curettage of an enchondroma. - Curettage and Burring: The lobulated, avascular cartilaginous matrix is curetted. The cavity is then systematically burred until normal, bleeding cancellous bone is encountered.
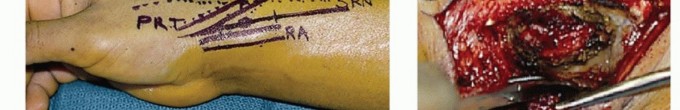
FIGURE 7: Post-curettage cavity after removal of an enchondroma, demonstrating clean bony margins ready for reconstruction. - Reconstruction: The defect is typically packed with cancellous autograft (harvested from the distal radius or olecranon) or demineralized bone matrix (DBM) allograft.
- Fracture Management: If a concomitant pathologic fracture exists, internal fixation with K-wires or low-profile plates is performed to restore length, alignment, and rotation.
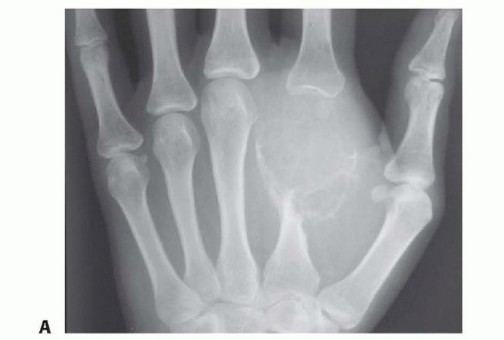
FIGURE 8: Radiograph demonstrating cancellous bone graft impaction within the defect after enchondroma excision, stabilized with K-wires for a concurrent pathologic fracture.
Unicameral Bone Cyst of the Distal Radius
Surgical Approach
- Volar Approach (Modified Henry): Provides excellent exposure of the metaphyseal flare. The interval between the flexor carpi radialis (FCR) and the radial artery is developed. The pronator quadratus is elevated to expose the volar cortex.
- Dorsal Approach: Utilized for predominantly dorsal cysts. The interval between the third and fourth extensor compartments is commonly used.
Technique
- Decompression: A cortical window is created. The characteristic serous or serosanguinous fluid is evacuated.
- Curettage: The thin, fibrous lining of the cyst must be meticulously scraped from the endosteal surface.
- Burring: High-speed burring is performed to disrupt the sclerotic cyst wall and open the medullary canal, promoting bleeding and ingress of osteoprogenitor cells.
- Reconstruction: The cavity is densely packed with bone graft. In large metaphyseal defects, structural support may be required to prevent collapse.
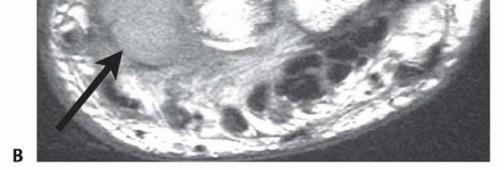
FIGURE 9: Intraoperative image showing the distal radial bone cyst cavity after thorough curettage and high-speed burring, prior to bone graft placement.
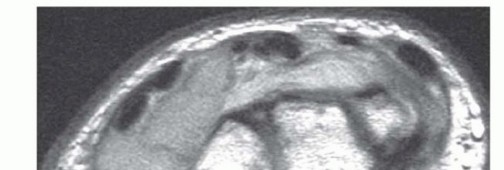
FIGURE 10: Postoperative radiograph demonstrating bone graft filling the UBC defect in the distal radius, augmented with supportive internal fixation (volar locking plate) to prevent insufficiency fracture.
Giant Cell Tumor of the Distal Radius
The surgical management of GCTs is highly demanding due to the requirement for aggressive oncologic clearance in close proximity to the radiocarpal joint.
Surgical Approach
An extensile volar or dorsal approach is dictated by the tumor's epicenter. A volar Henry approach is frequently utilized as it allows for the application of a volar locking plate, which is biomechanically advantageous for distal radius reconstruction.
Technique
- Extended Intralesional Curettage: A large cortical window is created. The highly vascular, friable tumor tissue is aggressively curetted.

FIGURE 11: Intraoperative image demonstrating extensive curettage of a giant cell tumor cavity in the distal radius. Note the size of the cortical window required for adequate visualization. - High-Speed Burring: Systematic, aggressive burring of the entire cavity is mandatory. Particular care is required at the subchondral bone plate to avoid intra-articular penetration while ensuring complete tumor removal.
- Adjuvant Therapy: Multiple adjuvants are often employed sequentially (e.g., phenol followed by argon beam coagulation) to maximize the depth of tumor necrosis.
- Reconstruction:
- PMMA Cementation: PMMA is frequently the reconstructive material of choice. The exothermic polymerization reaction provides a secondary thermal adjuvant effect. PMMA provides immediate structural stability to support the articular surface and facilitates early postoperative mobilization. Furthermore, the radiopaque nature of PMMA allows for easy radiographic detection of local recurrence (which appears as a new radiolucent halo at the cement-bone interface).
- Bone Grafting: If subchondral bone stock is severely depleted, a "sandwich" technique can be used: a layer of cancellous bone graft is placed against the subchondral plate to
Clinical & Radiographic Imaging
You Might Also Like