Operative Management of Ulnar Nerve Injuries at the Wrist: A Comprehensive Surgical Guide
Key Takeaway
Ulnar nerve transections at the wrist frequently accompany injuries to the ulnar artery and flexor carpi ulnaris. Because the nerve possesses both critical motor and sensory fascicles at this level, precise rotational alignment during microsurgical repair is paramount. This guide details the step-by-step surgical technique for primary neurorrhaphy, emphasizing fascicular matching, tension-free coaptation, and the specialized management of the deep motor branch to restore intrinsic hand function.
INTRODUCTION AND SURGICAL ANATOMY
Lacerations at the volar aspect of the wrist frequently result in devastating injuries to the neurovascular and tendinous structures, classically referred to as a "spaghetti wrist." When the ulnar artery and the tendon of the flexor carpi ulnaris (FCU) are severed at the wrist crease, the ulnar nerve is almost invariably transected due to its immediate anatomic proximity.
At the level of the distal forearm and wrist, the ulnar nerve is a mixed nerve, containing highly organized motor and sensory fascicles. Just proximal to or within Guyon’s canal, it bifurcates into its two terminal divisions:
* The Superficial Sensory Branch: Supplies sensation to the volar aspect of the hypothenar eminence, the small finger, and the ulnar half of the ring finger.
* The Deep Motor Branch: Dives deep between the abductor digiti minimi and the flexor digiti minimi brevis, coursing around the hook of the hamate. It provides critical motor innervation to the hypothenar muscles, all dorsal and volar interossei, the medial two lumbricals, the adductor pollicis, and the deep head of the flexor pollicis brevis.
These intrinsic muscles are fundamentally responsible for the sophisticated, quick, and skillful movements of the fingers, including powerful pinch and grip. While numerous tendon transfers have been devised to restore motor function lost by ulnar nerve interruption, direct, tension-free microsurgical repair of the nerve remains the gold standard and is highly desirable to restore native biomechanics.
Clinical Pearl: The ulnar nerve at the wrist is topographically highly organized. The motor fascicles are generally situated dorsally and ulnarly, while the sensory fascicles are located volarly. Recognizing this internal topography is the cornerstone of successful rotational alignment during neurorrhaphy.
PREOPERATIVE EVALUATION AND INDICATIONS
Clinical Assessment
A thorough preoperative neurological examination is mandatory. Complete transection of the ulnar nerve at the wrist presents with:
* Sensory Deficit: Loss of two-point discrimination over the volar small finger and ulnar half of the ring finger. (Note: Dorsal ulnar sensation may be preserved if the dorsal ulnar cutaneous nerve, which branches 5-8 cm proximal to the wrist, is spared).
* Motor Deficit: Inability to cross the fingers (loss of interossei), weak key pinch with compensatory interphalangeal joint flexion (Froment’s sign, due to adductor pollicis paralysis), and an abducted posture of the small finger (Wartenberg’s sign, due to unopposed extensor digiti minimi and paralyzed third palmar interosseous).
* Claw Deformity: Hyperextension of the metacarpophalangeal (MCP) joints and flexion of the interphalangeal (IP) joints of the ring and small fingers, resulting from the loss of intrinsic muscle balance.
Indications for Surgery
- Acute Lacerations: Immediate or early delayed (within 1-3 weeks) primary microsurgical repair is indicated for all sharp transections.
- Delayed Presentations: Secondary repair or nerve grafting is indicated if the injury is recognized late, provided the motor endplates have not undergone irreversible fibrosis (typically within 12-18 months).
PREOPERATIVE PREPARATION AND POSITIONING
- Anesthesia: General anesthesia or regional brachial plexus block.
- Positioning: The patient is positioned supine with the operative arm extended on a radiolucent hand table. The shoulder is abducted, and the forearm is supinated.
- Equipment: A well-padded pneumatic tourniquet is applied to the proximal arm. Loupe magnification (minimum 3.5x to 4.5x) is required for the initial dissection, and an operating microscope is mandatory for the intraneural dissection and fascicular repair. Microsurgical instruments, including diamond knives, microscissors, and jeweler's forceps, must be available.
SURGICAL TECHNIQUE: PRIMARY REPAIR OF THE ULNAR NERVE
1. Exposure and Incision
With the pneumatic tourniquet inflated to an appropriate pressure (typically 250 mmHg or 100 mmHg above systolic pressure), make proximal and distal extensile skin incisions.
* Avoid crossing the wrist flexion creases at a perpendicular angle. Utilize a Brunner-type zigzag incision or a lazy-S incision to prevent postoperative flexion contractures.
* Expose the proximal and distal segments of the ulnar nerve. At this stage, do not remove the nerve ends from their normal soft-tissue beds. Preserving the native bed maintains the longitudinal blood supply (vasa nervorum) for as long as possible.
2. Marking Rotational Alignment
Before any mobilization or resection of the nerve ends occurs, rotational alignment must be secured.
* Place a 6-0 or 7-0 marking suture through the epineurium at the exact anterior (volar) aspect of both the proximal and distal segments.
* Place these sutures some distance from the scarred or traumatized area so they are not excised during the preparation of the nerve stumps.
* This step is critical; a rotational mismatch of even a few degrees can direct regenerating motor axons into sensory end-organs, dooming functional recovery.
3. Preparation of the Nerve Stumps
Free each segment from the surrounding soft tissues, employing meticulous hemostasis. Transition from loupe magnification to the operating microscope.
* Using microscissors, a diamond knife, or a fresh scalpel blade on a sterile wooden tongue depressor, make clean, transverse cuts.
* Excise the traumatized tissue: remove the neuroma from the proximal segment and the glioma from the distal segment.
* Continue serial sectioning ("bread-loafing") by 1-2 mm increments until healthy, pouting fascicles are visualized. Healthy fascicles will exhibit a distinct "mushrooming" effect from the cut surface, and punctate bleeding from the vasa nervorum should be visible when the tourniquet is temporarily deflated.
Surgical Warning: Inadequate resection of the zone of injury is a primary cause of neurorrhaphy failure. You must resect back to normal, unscarred fascicular tissue, even if it creates a gap. A tension-free nerve graft is always superior to a primary repair performed under tension or through fibrotic tissue.
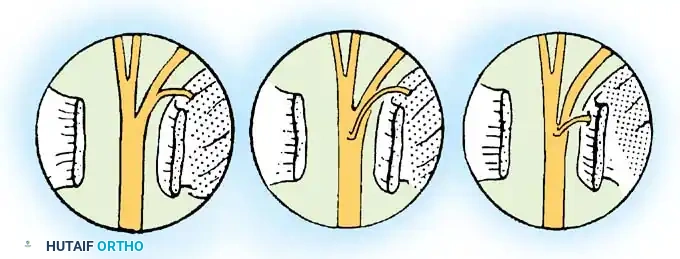
4. Fascicular Matching and Alignment
Inspect each cut end under high magnification (10x to 20x) for the pattern of large and small bundles.
* The ulnar nerve at the wrist typically presents with a distinct grouping of fascicles.
* By matching these fascicular patterns, mapping the vascular landmarks on the epineurium, and utilizing the two epineurial marking sutures placed earlier, perfect rotational alignment should be achieved.
5. Mobilization for Tension-Free Coaptation
A primary repair must be entirely tension-free. If a gap exists after debridement, length must be gained.
* Proximal Dissection: Dissect and mobilize the nerve more proximally into the forearm. Careful intraneural dissection of the muscular branches may allow significant distal advancement of the main trunk.
* Preservation of Motor Branches: When advancing the nerve distally, absolutely do not divide its branches to the flexor carpi ulnaris or the flexor digitorum profundus in the proximal forearm.
* Elbow Flexion: Flex the elbow as necessary to reduce tension. However, avoid excessive flexion (greater than 45-60 degrees), as extreme flexion can lead to postoperative joint contractures and increased tension upon mobilization.
* Anterior Transposition: If necessary, the ulnar nerve can be transplanted anteriorly from behind the medial epicondyle of the humerus. This maneuver can yield up to 2-3 cm of additional length at the wrist.
Pitfall: Extensive freeing of a nerve strips its segmental blood supply. While mobilization is necessary to overcome gaps, overzealous skeletonization will induce ischemia, leading to intraneural fibrosis and poor axonal regeneration. Balance mobilization with the preservation of the mesoneurium.
6. Microsurgical Coaptation
Use the operating microscope to align the major groups of fascicles.
* While four-quadrant epineurial traction sutures may be sufficient for a purely epineurial repair, a grouped fascicular (epiperineurial) repair is highly recommended at the wrist due to the distinct separation of motor and sensory components.
* Technique: It is often easier to start with the deep (dorsal) surface. Place a posterior epineurial suture and close the cut surface "like a book."
* Use a combination of 8-0 or 9-0 nylon for the epiperineurial alignment and 10-0 nylon for the perineurial (fascicular) coaptation.
* Ensure that the fascicles are gently approximated without buckling or overlapping. The sutures should merely hold the ends in apposition; the fibrin clot will provide the actual seal.
7. Managing the Bifurcation
When the ulnar nerve is severed near, but just distal to, its division into the volar superficial sensory branch and the deep motor branch, the complexity of the repair increases.
* Identify the two small proximal segments corresponding to these branches.
* Dissect them apart in a proximal direction (intraneural neurolysis) for ease of mobilization.
* Suture each branch separately to its corresponding distal stump using 10-0 nylon under the microscope. This guarantees that motor axons are directed exclusively into the deep branch.
SURGICAL TECHNIQUE: DEEP BRANCH OF THE ULNAR NERVE
The Boyes Technique and Intrinsic Restoration
Dr. Joseph H. Boyes extensively documented the feasibility and critical importance of repairing the deep branch of the ulnar nerve. Because this branch supplies the intrinsic muscles of the hand not innervated by the median nerve (the medial two lumbricals, all interossei, the hypothenar muscles, and the adductor pollicis), its restoration is paramount for grip strength and fine motor dexterity.
- Anatomic Challenges: The deep branch is small, deeply situated, and surrounded by dense fibrous tissue as it passes through the piso-hamate hiatus and around the hook of the hamate.
- Exposure: To adequately expose the distal stump of a severed deep branch, the origin of the hypothenar muscles must often be partially elevated or split. The pisohamate ligament may need to be divided to trace the nerve into the deep palmar space.
- Repair Strategy: Due to the deep location, placing circumferential sutures is exceptionally difficult. Often, two or three well-placed 10-0 or 11-0 perineurial sutures on the accessible anterior and lateral surfaces are sufficient, provided the nerve ends lie in a tension-free bed. Fibrin glue can be used as an adjunct to augment the repair in this difficult-to-reach area.
Clinical Pearl: If the deep motor branch is avulsed or destroyed such that primary repair or grafting is impossible, early consideration should be given to an end-to-side nerve transfer (e.g., anterior interosseous nerve to deep ulnar nerve transfer) to preserve the motor endplates of the intrinsic muscles while awaiting definitive tendon transfers.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a meticulous microsurgical repair can be entirely undone by improper postoperative care.
Immobilization Phase (Weeks 0-3)
- The wrist and forearm are immobilized in a well-padded dorsal blocking splint.
- The wrist is placed in 20 to 30 degrees of flexion to minimize tension on the repair site. If the elbow was flexed to gain length, a long-arm splint is utilized with the elbow at 45-60 degrees of flexion.
- Digital motion is encouraged immediately to prevent tendon adhesions, particularly if concomitant flexor tendon repairs (e.g., FCU) were performed.
Mobilization Phase (Weeks 3-6)
- At 3 weeks, the splint is gradually adjusted to bring the wrist into a neutral position.
- If a long-arm splint was used, the elbow is gradually extended by 15 degrees per week.
- Gentle, active range of motion of the wrist is initiated. Passive stretching is strictly prohibited to avoid traction injury to the regenerating axons.
Strengthening and Re-education (Weeks 6 and Beyond)
- By 6 weeks, the splint is discontinued during the day.
- Nerve gliding exercises are initiated to prevent tethering of the ulnar nerve in the scar bed.
- Sensory re-education begins once advancing Tinel's sign reaches the palm and protective sensation returns to the digits.
- Motor recovery is monitored clinically and via serial electromyography (EMG) starting at 3 to 4 months postoperatively.
COMPLICATIONS AND SALVAGE PROCEDURES
Despite optimal microsurgical technique, complications can arise:
* Neuroma-in-Continuity: Presents with severe pain and a halted Tinel's sign. May require re-exploration, neurolysis, or excision and grafting.
* Incomplete Motor Recovery: Intrinsic muscle atrophy may persist due to the distance regenerating axons must travel (approximately 1 mm per day) and the irreversible degradation of motor endplates if reinnervation takes longer than 12-18 months.
* Salvage Tendon Transfers: If intrinsic function does not return, secondary reconstructive procedures are indicated to correct the claw deformity and restore pinch strength. Common procedures include the Zancolli lasso procedure (FDS to A1 pulley), the Bouvier maneuver, or the Smith-Hastings transfer (ECRB to adductor pollicis) to restore powerful key pinch.
CONCLUSION
The operative management of ulnar nerve injuries at the wrist demands a profound understanding of fascicular anatomy, rigorous adherence to microsurgical principles, and meticulous handling of soft tissues. By prioritizing exact rotational alignment, ensuring tension-free coaptation, and specifically addressing the deep motor branch, the orthopaedic surgeon provides the patient with the highest possible probability of restoring the intricate biomechanics and sensory fidelity of the human hand.
You Might Also Like