Mastering Peripheral Nerve Repair: Primary, Delayed Primary, and Secondary Strategies
Key Takeaway
The timing of peripheral nerve repair is a critical determinant of functional recovery. Primary repair is indicated for clean, sharp transections, while secondary repair is reserved for crush, traction, or highly contaminated injuries requiring scar demarcation. This guide details the evidence-based indications, biomechanical principles of tension-free coaptation, step-by-step surgical techniques, and postoperative rehabilitation protocols essential for optimizing motor and sensory outcomes in peripheral nerve surgery.
INTRODUCTION TO PERIPHERAL NERVE REPAIR
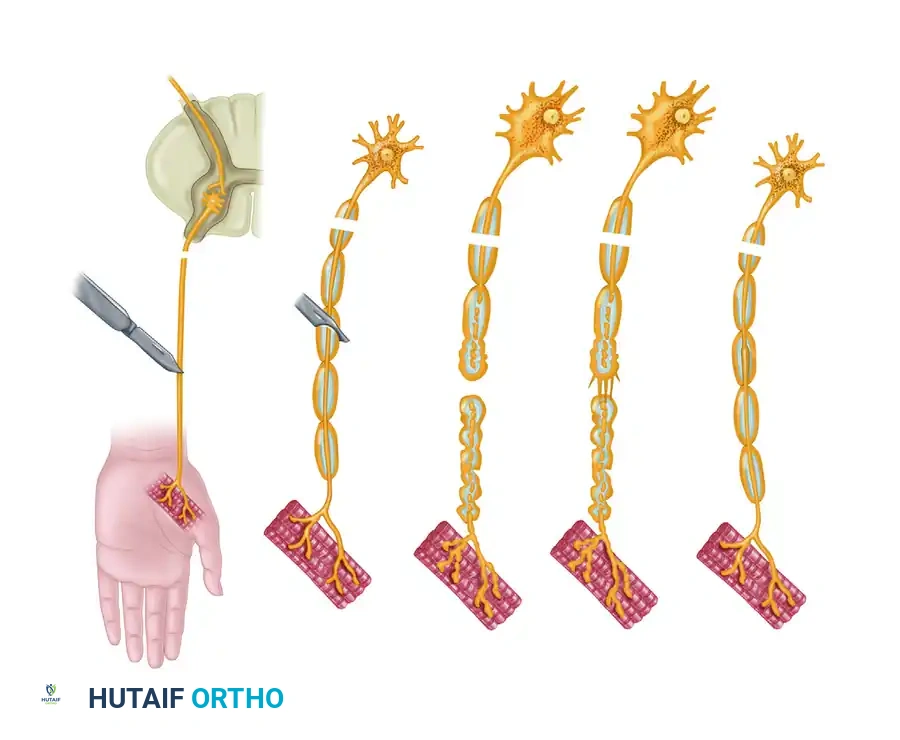
The management of peripheral nerve injuries remains one of the most technically demanding and biologically complex challenges in operative orthopaedics and microsurgery. The ultimate goal of nerve repair is to restore motor and sensory function by facilitating the precise regeneration of axons across the zone of injury into the distal endoneurial tubes.
The controversy regarding the optimal timing of nerve repair has historically divided surgical opinion. Experimental models, notably the foundational work of Grabb, Müller, and Grubel, heavily advocate for immediate primary repair to capitalize on the optimal biological milieu. Conversely, extensive clinical observations derived from wartime trauma have demonstrated the catastrophic failure rates of primary repair in the setting of high-energy, contaminated wounds, thereby establishing the role of delayed and secondary repairs.
Modern peripheral nerve surgery dictates that the timing of intervention must be meticulously tailored to the mechanism of injury, the condition of the soft-tissue envelope, and the physiological status of the patient.
CLASSIFICATION OF REPAIR TIMING
The nomenclature applied to the timing of nerve repair is strictly defined by the interval between the injury and the surgical intervention:
- Primary Repair: Performed immediately after the injury, typically within the first 6 to 12 hours.
- Delayed Primary Repair: Performed within the first 2 to 2.5 weeks post-injury.
- Secondary Repair: Performed after 2.5 to 3 weeks, often extending up to 3 to 6 months post-injury.
PRIMARY AND DELAYED PRIMARY NERVE REPAIR
Indications and Patient Selection
Primary or delayed primary repair is the gold standard for clean, sharp nerve transections (e.g., glass lacerations, scalpel injuries, or clean knife wounds). In these scenarios, the zone of trauma is highly localized, and the extent of intraneural damage is minimal and immediately apparent.
We advocate for repairing injured nerves on the day of injury or within the first 5 to 7 days, provided the wound is uncontaminated and the soft-tissue envelope is viable. A delay of up to 2.5 weeks (delayed primary repair) is acceptable and often necessitated by logistical factors, such as the stabilization of a polytraumatized patient, the availability of specialized microsurgical instruments, or the need for a fellowship-trained microsurgeon.
Clinical Pearl:
The decision to perform a primary repair hinges entirely on the mechanism of injury. A sharp laceration allows for immediate, tension-free coaptation. If there is any element of crush, avulsion, or blast injury, primary repair is strictly contraindicated due to the unpredictable extent of intraneural necrosis.
Neurobiological and Biomechanical Considerations
The fundamental biomechanical principle of peripheral nerve surgery is the absolute avoidance of tension at the coaptation site. Tension compromises the delicate intraneural microcirculation (vasa nervorum), leading to ischemia, fibroblast proliferation, and subsequent scarring that physically blocks axonal regeneration.
The seminal work of Millesi and Meissl revolutionized nerve surgery by demonstrating that nerve grafts performed without tension yield vastly superior functional outcomes compared to primary repairs performed under tension. This holds true despite the biological disadvantage of requiring regenerating axons to cross two separate suture lines in a grafted nerve.
The Biological Cost of Delay
While delayed primary repair is acceptable, extending the delay beyond 3 weeks introduces significant biological and anatomical hurdles. Authors advocating for early intervention highlight the following progressive pathological changes:
- Muscle Atrophy: Denervated muscle undergoes progressive atrophy. According to Sunderland, irreversible fibrotic changes occur in the muscle cells after 12 to 18 months, rendering motor recovery impossible even if axonal reinnervation eventually occurs.
- Endoneurial Contraction: The distal endoneurial tubules progressively shrink in diameter, creating a size mismatch that impedes the entry of regenerating proximal axons.
- Nerve Retraction: The elasticity of the nerve causes the proximal and distal stumps to retract over time, significantly increasing the gap length and necessitating nerve grafting.
- Joint Contractures: Prolonged denervation leads to muscular imbalance and rigid joint contractures, negating the functional benefits of a successful nerve repair.
- Fascicular Misalignment: Intraneural scarring and anatomical distortion make precise fascicular alignment exponentially more difficult in delayed settings.
Surgical Warning:
While sensory end-organs can survive and accept reinnervation up to 2 years post-injury, motor endplates undergo irreversible apoptosis by 12 to 18 months. Therefore, motor nerve repairs must be prioritized and expedited.
SECONDARY NERVE REPAIR
Indications for Secondary Repair
Secondary nerve repair is indicated when the initial injury characteristics preclude safe and effective primary coaptation. The surgeon must actively choose to delay the repair in the presence of the following conditions:
- Extensive Soft-Tissue Injury: Nerve repair is futile without a well-vascularized soft-tissue bed. Skin coverage (via local or free flaps) takes absolute precedence.
- Severe Wound Contamination: Infection at the repair site will destroy the coaptation. Serial debridements must be completed before nerve reconstruction.
- Polytrauma (Damage Control Orthopaedics): In patients with multiple life-threatening injuries, the priority is physiological stabilization, hemorrhage control, and skeletal fixation.
- Crush and Traction Injuries: These mechanisms cause longitudinal intraneural damage that extends far beyond the macroscopic zone of injury.
- Unknown Previous Surgical Intervention: If a patient is referred after an initial exploration by another surgeon, and the quality of the repair is unknown, secondary exploration is mandated.
The Principle of Scar Demarcation
The most critical rationale for delaying repair in crush, traction, or blast injuries is the concept of "scar demarcation." At the time of acute injury, it is visually impossible to differentiate between healthy, viable fascicles and those that have suffered irreversible ultrastructural damage.
By waiting 3 to 6 weeks, Wallerian degeneration occurs distally, and the zone of proximal trauma declares itself as a fibrotic neuroma. During a secondary repair, the surgeon can serially section the nerve stumps until healthy, pouting fascicles ("mushrooming") are visualized, ensuring that the coaptation is performed in healthy tissue.
Pitfall:
Attempting primary repair on a stretched or crushed nerve inevitably results in suturing necrotic tissue to necrotic tissue. This guarantees the formation of a neuroma-in-continuity and complete failure of axonal regeneration.
SURGICAL TECHNIQUE: STEP-BY-STEP APPROACH
1. Preoperative Preparation and Positioning
- Anesthesia: General anesthesia is preferred to prevent patient movement during microsurgical coaptation. Avoid long-acting paralytics if intraoperative nerve stimulation is planned.
- Tourniquet: A pneumatic tourniquet is essential for a bloodless field during the initial exposure. However, it should be deflated prior to final hemostasis and nerve coaptation to identify and control bleeding from the vasa nervorum.
- Magnification: Operating loupes (minimum 3.5x to 4.5x) are required for exposure. An operating microscope is mandatory for perineurial or grouped fascicular repairs.
2. Surgical Exposure
- Incision: Utilize extensile incisions. Never incise directly over the nerve injury.
- Proximal and Distal Control: Always identify the nerve in pristine, uninjured tissue proximally and distally. Once identified, trace the nerve carefully toward the zone of injury (the "outside-in" approach).
- Handling: Handle the nerve only by the external epineurium. Never grasp the fascicles with forceps.
3. Preparation of the Nerve Stumps
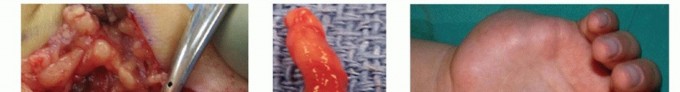
- Neuroma Excision: In secondary repairs, the proximal neuroma and distal glioma must be resected.
- Serial Sectioning: Using a fresh scalpel blade or specialized nerve scissors, perform serial 1-mm step-cuts until healthy tissue is reached.
- Visual Cues of Viability: Healthy nerve ends will exhibit "pouting" or "mushrooming" of the fascicles out of the epineurial sheath. The tissue should bleed briskly from the epineurial and interfascicular vessels.
4. Fascicular Alignment (Topography)
Accurate rotational alignment is the most critical technical step to prevent motor axons from regenerating into sensory end-tubes (and vice versa).
* Surface Landmarks: Match the longitudinal epineurial blood vessels.
* Fascicular Pattern: Match the size and spatial arrangement of the fascicular bundles.
* Intraoperative Stimulation: In awake patients or under specific anesthetic protocols, stimulation of the distal stump (up to 72 hours post-injury) can identify motor fascicles.
5. Coaptation and Suturing
- Epineurial Repair: The standard technique for most peripheral nerves. Place 8-0 or 9-0 non-absorbable monofilament sutures (e.g., Nylon) through the epineurium.
- Technique: Place the first two sutures 180 degrees apart to act as traction sutures. Place intermediate sutures to close the epineurial gap. Avoid placing too many sutures, which increases foreign body reaction and scarring.
- Grouped Fascicular Repair: Indicated for major mixed nerves (e.g., the ulnar nerve at the wrist) where distinct motor and sensory fascicular groups can be identified and repaired individually using 10-0 sutures through the interfascicular epineurium.
6. Managing the Gap: Nerve Grafting
If the nerve ends cannot be brought together without tension (defined as the ability to hold the nerve ends together with a single 8-0 suture), a nerve graft is absolutely indicated.
* Donor Sites: The reversed sural nerve is the workhorse autograft. Other options include the medial antebrachial cutaneous nerve or the lateral antebrachial cutaneous nerve.
* Cable Grafting: Because the donor nerve is usually smaller in diameter than the recipient nerve, multiple strands (cables) are sutured together to match the cross-sectional area of the injured nerve.
* Reversal: Grafts must be reversed to prevent regenerating axons from escaping through the transected branches of the donor nerve.
POSTOPERATIVE PROTOCOL AND REHABILITATION
The success of a meticulously performed nerve repair relies heavily on the postoperative rehabilitation protocol.
Phase 1: Immobilization (Weeks 0-3)
- The limb is immobilized in a well-padded orthosis to remove all tension from the repair site.
- For flexor surface repairs (e.g., median nerve at the wrist), the joint is immobilized in slight flexion.
- Strict elevation is maintained to minimize edema, which can compress the microcirculation of the repair.
Phase 2: Early Mobilization (Weeks 3-6)
- The orthosis is gradually adjusted to bring the joint to a neutral position.
- Active and passive range of motion exercises are initiated for adjacent, uninvolved joints to prevent contractures.
- Nerve gliding exercises are introduced cautiously to prevent the repair site from adhering to the surrounding soft-tissue bed.
Phase 3: Re-education and Strengthening (Weeks 6+)
- Sensory Re-education: As regenerating axons reach the distal targets, patients often experience hypersensitivity or altered sensation. Desensitization protocols (using various textures and vibration) are critical.
- Motor Biofeedback: Electromyographic (EMG) biofeedback can assist patients in recruiting newly reinnervated motor units.
- Monitoring: Tinel's sign should be monitored monthly. The advancing front of a Tinel's sign indicates the progression of regenerating axons (typically advancing at a rate of 1 mm per day, or 1 inch per month).
PROGNOSIS AND OUTCOMES
The prognosis following peripheral nerve repair is multifactorial. Key determinants of success include:
1. Age of the Patient: Pediatric patients possess a robust neuroplasticity and consistently demonstrate superior functional recovery compared to adults.
2. Level of Injury: Distal injuries (e.g., wrist level) have a shorter distance for axonal regeneration, resulting in better outcomes than proximal injuries (e.g., brachial plexus).
3. Timing of Repair: Repairs performed within the first 3 months yield the highest percentage of functional motor recovery.
4. Mechanism of Injury: Sharp transections fare significantly better than crush or avulsion injuries.
While sensory recovery can be observed even when repairs are performed up to 2 years post-injury, the window for motor recovery is strictly limited by the biological lifespan of the denervated motor endplate. Therefore, the orthopaedic surgeon must approach peripheral nerve injuries with a sense of urgency, a deep understanding of neurobiology, and an uncompromising commitment to microsurgical principles.
You Might Also Like