Management of Hand & Wrist Nerve Injuries | Operative Guide
Key Takeaway
Peripheral nerve injuries in the hand and wrist demand meticulous clinical evaluation and precise microsurgical reconstruction. This guide details the preoperative assessment, Seddon and Sunderland classifications, and step-by-step operative techniques for primary repair and nerve grafting. Emphasizing evidence-based protocols, it provides orthopedic surgeons with the critical biomechanical and anatomical insights required to optimize sensory and motor recovery following complex neurovascular trauma.
Introduction to Peripheral Nerve Injuries of the Hand
The management of peripheral nerve injuries in the hand, digits, palm, and wrist represents one of the most technically demanding frontiers in operative orthopedics and hand surgery. Restoring functional sensibility and precise motor control requires a profound understanding of neuroanatomy, the pathophysiology of Wallerian degeneration, and advanced microsurgical techniques.
This comprehensive guide delineates the essential principles of evaluating and treating nerve injuries distal to the forearm. While the foundational tenets of microsurgery apply universally, the unique biomechanical demands and compact anatomical architecture of the hand necessitate highly specific surgical strategies.
Clinical Pearl: A high index of suspicion is paramount in hand trauma. If a flexor tendon function deficit is observed following a volar laceration, the surgeon must assume that at least one adjacent digital nerve has been compromised until proven otherwise via direct surgical exploration.
Preoperative Clinical Evaluation
Challenges in Acute Assessment
Accurate preoperative evaluation of nerve injuries in the hand is frequently confounded by the acute trauma setting. Factors that obscure a reliable clinical examination include life- or limb-threatening concomitant injuries, patient intoxication, extreme anxiety, altered mental status, and the inherent difficulties of examining pediatric patients. Furthermore, extensive crush or avulsion injuries may mask isolated nerve deficits.
If conditions preclude a definitive examination during the initial trauma bay assessment, the hand must be systematically re-examined within a reasonable timeframe once the patient is stabilized and cooperative. Documenting the precise neurological deficit prior to any surgical intervention is a medicolegal and clinical imperative, establishing a baseline to monitor spontaneous recovery or dictate the necessity of surgical exploration.
Mechanisms and Classification of Injury
Nerve injuries in civilian practice typically arise from direct trauma (blunt force, fractures, ballistic wounds), sharp lacerations, severe traction/stretch, or acute compression/entrapment. Determining the mechanism of injury is critical for prognosticating recovery and timing surgical intervention.
To standardize communication and guide treatment, injuries are classified according to the Seddon and Sunderland grading systems:
- Neurapraxia (Sunderland First-Degree): A focal conduction block without axonal disruption. Typically caused by mild compression or traction (e.g., "funny bone" contusion). Recovery is spontaneous and complete, usually within days to weeks.
- Axonotmesis (Sunderland Second to Fourth-Degree): Disruption of the axons with variable preservation of the connective tissue framework (endoneurium, perineurium, epineurium). Wallerian degeneration occurs distally. Recovery depends on the integrity of the supporting stroma.
- Neurotmesis (Sunderland Fifth-Degree): Complete transection of the nerve trunk. Spontaneous recovery is impossible; surgical coaptation is mandatory.
- Sunderland Sixth-Degree (Mackinnon Modification): A mixed injury pattern involving combinations of the above within different fascicles of the same nerve.
Sensibility Testing and Autonomous Zones
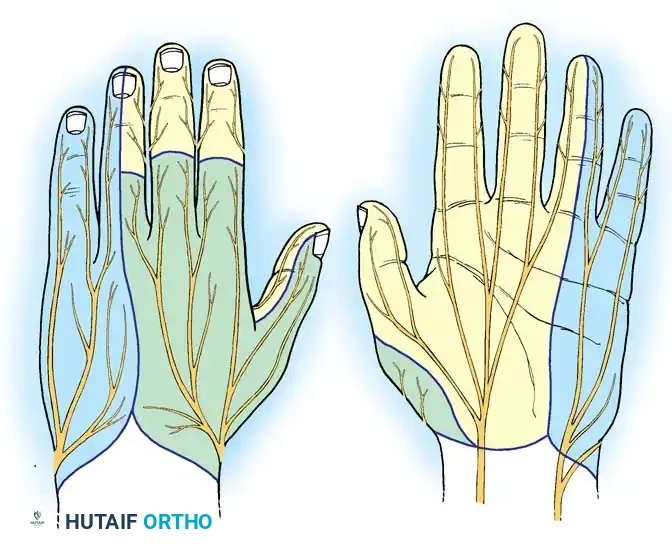
Evaluating sensibility requires a thorough mastery of the classic sensory distributions of the median, radial, and ulnar nerves.

Beyond the general distribution, the surgeon must test the autonomous sensory zones—areas exclusively innervated by a single nerve, minimizing the confounding effect of overlapping innervation:
* Median Nerve: Volar pulp of the index finger.
* Ulnar Nerve: Volar pulp of the little finger.
* Radial Nerve: Dorsal thumb-index web space.
In cases of sharp transection, the clinical deficit is usually absolute and easily defined. However, closed injuries or partial lacerations may present with spotty preservation of light touch and pain.
Standardized testing modalities include:
1. Pinprick Testing: Assesses the spinothalamic tracts (pain).
2. Cotton-Tipped Applicator/Semmes-Weinstein Monofilaments: Assesses light touch and pressure thresholds.
3. Two-Point Discrimination (2PD): The gold standard for assessing functional tactile gnosis. Normal static 2PD in the digital pulp is 6 mm or less.
Surgical Warning: Patients with closed traction injuries or partial lacerations may exhibit markedly widened two-point discrimination despite intact light touch. Advanced imaging, such as high-resolution MRI or ultrasound, may be indicated to diagnose closed partial ruptures requiring surgical exploration.
Motor Function Assessment
Motor evaluation must isolate specific muscle functions to differentiate between median, ulnar, and radial nerve lesions. While proximal forearm muscles (e.g., pronator teres, flexor carpi radialis) are innervated by the median nerve, hand-specific motor testing focuses on the intrinsic musculature:
- Median Nerve (Recurrent Motor Branch): Test the abductor pollicis brevis (APB) by having the patient abduct the thumb perpendicular to the palm against resistance. Palpate the APB muscle belly to ensure the abductor pollicis longus (radial nerve) is not substituting.
- Ulnar Nerve (Deep Motor Branch): Test the first dorsal interosseous (FDI) by resisting index finger abduction. Test the adductor pollicis via Froment's sign (hyperflexion of the thumb IP joint during key pinch indicates ulnar nerve weakness, as the median-innervated FPL compensates for the paralyzed adductor pollicis).
- Radial Nerve: Test wrist extension (ECRL, ECRB, ECU) and digital extension at the metacarpophalangeal (MCP) joints (EDC, EIP, EDM).
Sudomotor Activity (Sweating)
Sudomotor function is mediated by sympathetic postganglionic fibers that travel within peripheral nerves. Complete nerve transection results in localized anhidrosis (loss of sweating) in the autonomous zone of the injured nerve.
This is particularly useful in pediatric patients or uncooperative adults where subjective sensory testing is unreliable. Sudomotor function can be assessed visually, via the tactile friction of the skin (denervated skin feels smooth and dry), or objectively using the Ninhydrin test or the O'Riain wrinkle test (denervated skin fails to wrinkle after prolonged immersion in warm water).
Principles of Nerve Regeneration and Repair Timing
Following transection, the distal nerve segment undergoes Wallerian degeneration, clearing axonal debris to prepare for regenerating growth cones. Axons regenerate at an approximate rate of 1 mm per day (or 1 inch per month) following a latent period of 3 to 4 weeks.
Primary and Delayed Primary Nerve Repair
- Primary Repair (Within 1 week): Indicated for sharp, clean lacerations (e.g., glass or knife wounds). Immediate repair prevents retraction of the nerve ends, utilizes the optimal biological window before extensive scarring occurs, and allows for precise fascicular alignment before the distal stump undergoes significant shrinkage.
- Delayed Primary Repair (1 to 3 weeks): Indicated for contaminated wounds, severe crush injuries, or when the extent of nerve damage is initially unclear. The wound is debrided and left to demarcate. The nerve ends may be tacked to adjacent fascia to prevent retraction. Repair is performed once the wound bed is clean and the zone of injury is clearly defined.
Secondary Nerve Repair
- Secondary Repair (>3 weeks): Often required for missed injuries, extensive avulsions, or severe blast trauma. By this stage, the nerve ends have retracted, and neuromas/gliomas have formed. The scarred ends must be resected back to healthy fascicles, frequently resulting in a gap that precludes tension-free primary coaptation, thereby necessitating a nerve graft.
Operative Management and Microsurgical Techniques
Patient Positioning and Preparation
- Anesthesia: General anesthesia or regional block (axillary or supraclavicular) is preferred. Local anesthesia with epinephrine (WALANT) can be used for isolated digital nerve repairs but may obscure the assessment of nerve vascularity.
- Positioning: Supine with the arm extended on a radiolucent hand table.
- Tourniquet: A pneumatic tourniquet is essential for a bloodless field. Exsanguinate the limb with an Esmarch bandage and inflate the tourniquet to 250 mm Hg (or 100 mm Hg above systolic pressure). Note: Tourniquet time should be strictly monitored and limited to 2 hours to prevent ischemic neuropraxia.
- Magnification: Operating loupes (minimum 3.5x to 4.5x) are mandatory for initial dissection. An operating microscope is highly recommended for the actual epineurial or fascicular coaptation.
Surgical Approach and Nerve Preparation
- Incision: Extend traumatic lacerations using Bruner zigzag incisions or mid-lateral incisions to prevent flexion contractures. Never cross flexion creases perpendicularly.
- Dissection: Identify the nerve in healthy, uninjured tissue proximally and distally, then trace it toward the zone of injury. This "outside-in" approach minimizes the risk of iatrogenic damage within the scarred or hemorrhagic wound bed.
- Preparation of Nerve Ends: For sharp lacerations, minimal trimming is required. For crush or delayed injuries, resect the proximal neuroma and distal glioma using a fresh scalpel blade or specialized nerve scissors until healthy, pouting fascicles ("mushrooming") are visualized.
Surgical Warning: Inadequate resection of scarred nerve ends is a primary cause of repair failure. The fascicles must be completely free of fibrotic tissue to allow axonal crossing.
Suturing Techniques
The goal of neurorrhaphy is a tension-free, anatomically aligned coaptation. Tension across the repair site causes ischemia and stimulates excessive scarring, blocking axonal regeneration.
- Alignment: Utilize surface landmarks, such as longitudinal epineurial blood vessels, to ensure correct rotational alignment. Matching fascicular topography is critical in mixed motor-sensory nerves (e.g., median or ulnar nerves at the wrist).
- Epineurial Repair: The standard technique for most hand nerve injuries.
- Place a colored silicone background material behind the nerve to enhance contrast.
- Use 8-0 or 9-0 monofilament nylon on a micro-spatula needle.
- Place the first two sutures 180 degrees apart to act as stay sutures.
- Place intermediate sutures to close the epineurium circumferentially, ensuring fascicles are contained within the repair and not protruding.
- Group Fascicular Repair: Indicated for proximal injuries of mixed nerves where distinct motor and sensory fascicular groups can be identified and matched. Requires higher magnification and 10-0 nylon.
Nerve Grafting Strategies
Indications and Donor Site Selection
If resection of the damaged nerve ends results in a gap that cannot be closed without tension (typically >1 to 2 cm in the hand, or any gap requiring joint flexion >20 degrees to close), a nerve graft is mandatory.
Common Donor Nerves:
* Sural Nerve: The workhorse for large defects. Provides up to 30-40 cm of graft material. Harvested from the posterolateral calf. Leaves a sensory deficit on the lateral aspect of the foot.
* Medial Antebrachial Cutaneous (MABC) Nerve: Excellent for upper extremity defects. Harvested from the medial arm/forearm.
* Lateral Antebrachial Cutaneous (LABC) Nerve: Harvested from the volar forearm.
* Posterior Interosseous Nerve (PIN) Terminal Branch: Useful for small digital nerve defects; harvested from the dorsal wrist capsule.
Surgical Technique for Nerve Grafting
- Cable Grafting: Because donor nerves are usually smaller in diameter than the recipient nerve (e.g., sural nerve to median nerve), multiple strands of the donor nerve are cut to length and sutured together to form a "cable" that matches the cross-sectional area of the recipient stump.
- Reversal of Graft: The nerve graft should be reversed (distal end of graft sutured to proximal stump of recipient) to prevent regenerating axons from escaping through small branching points within the graft.
- Coaptation: Suture the graft using the standard epineurial technique. Fibrin glue may be used to augment the repair and reduce the number of sutures required, minimizing foreign body reaction.
Management of Specific Nerve Injuries
Digital Nerves
Digital nerve lacerations are the most common peripheral nerve injuries in the hand.
* Approach: Volar Bruner or mid-lateral incisions. The neurovascular bundle lies volar to the Cleland ligaments.
* Repair: Primary epineurial repair using 8-0 or 9-0 nylon.
* Distal Injuries: Injuries distal to the distal interphalangeal (DIP) joint are technically challenging due to extensive arborization. Repair is generally recommended for the radial aspect of the index finger and the ulnar aspect of the thumb to preserve critical pinch sensibility.
Median Nerve at the Wrist and Palm
Injuries at the wrist (e.g., "spaghetti wrist" from glass lacerations or suicide attempts) often involve multiple flexor tendons.
* Approach: Standard carpal tunnel incision, extending proximally across the wrist crease in a zigzag fashion.
* Motor Branch: The recurrent motor branch must be carefully identified. Its anatomy is highly variable (Lanz classification: extraligamentous, subligamentous, transligamentous). Failure to repair the motor branch results in severe thenar atrophy and loss of thumb opposition.
* Repair: Group fascicular repair is highly beneficial here to separate the anterior motor fascicles from the posterior sensory fascicles.
Ulnar Nerve at the Wrist
The ulnar nerve enters the hand through Guyon's canal, bifurcating into a superficial sensory branch and a deep motor branch.
* Approach: Incision along the radial border of the flexor carpi ulnaris (FCU), extending into the palm. The volar carpal ligament is released.
* Deep Motor Branch: This branch dives deeply around the hook of the hamate. It is purely motor. Lacerations here are devastating to hand intrinsic function (claw hand deformity). Meticulous exploration and repair are mandatory.
Superficial Radial Nerve
The superficial sensory branch of the radial nerve emerges from beneath the brachioradialis in the distal third of the forearm.
* Pathology: It is highly susceptible to laceration, compression (Wartenberg's syndrome), and iatrogenic injury during De Quervain's release or distal radius fracture fixation.
* Neuroma Risk: The superficial radial nerve is notorious for developing exquisitely painful traumatic neuromas. If the nerve cannot be repaired, the proximal stump should be transposed deep into the brachioradialis muscle belly to prevent neuroma formation in the subcutaneous tissues.
Postoperative Rehabilitation Protocols
The success of a meticulous microsurgical repair can be entirely undone by improper postoperative management.
- Immobilization: The hand and wrist are immobilized in a custom orthosis to remove tension from the repair site. For volar repairs (median/ulnar), the wrist is typically splinted in 20-30 degrees of flexion. Immobilization is maintained for 3 weeks.
- Early Protected Motion: After 3 weeks, the splint is gradually adjusted to allow progressive extension. Active range of motion (ROM) is initiated to prevent tendon adhesions and joint contractures, ensuring the nerve glides smoothly within its bed.
- Sensory Re-education: As axons reach the distal targets (evidenced by an advancing Tinel's sign), the brain receives altered sensory signals. Formal sensory re-education programs, guided by a specialized hand therapist, are critical to help the patient interpret these new signals, improving functional tactile gnosis and spatial recognition.
- Motor Re-education: Biofeedback and targeted strengthening are employed once motor reinnervation is clinically detectable.
Rigorous adherence to these operative and rehabilitative principles ensures the highest probability of restoring complex neurovascular function following devastating hand trauma.
You Might Also Like

