Mastering Flexor Tendon Injuries: Pediatric Considerations and Traumatic Ruptures

Key Takeaway
Flexor tendon injuries in children and traumatic flexor tendon ruptures present unique surgical challenges. Successful management requires a deep understanding of tendon biomechanics, precise surgical timing, and meticulous repair techniques. This guide details the classification of avulsion injuries, step-by-step surgical approaches including tendon retrieval and reinsertion, and evidence-based postoperative rehabilitation protocols essential for restoring optimal hand function and preventing late complications.
INTRODUCTION TO FLEXOR TENDON TRAUMA
The management of flexor tendon injuries remains one of the most exacting disciplines within hand and orthopedic surgery. While sharp lacerations form the bulk of flexor tendon trauma, specialized scenarios—namely, injuries in the pediatric population and closed traumatic ruptures (avulsions) in adults—demand a highly nuanced approach. The margin for error is exceptionally narrow; surgical precision must be coupled with a profound understanding of tendon biomechanics, vascularity, and postoperative rehabilitation to prevent the dual nemeses of tendon rupture and restrictive adhesion formation.
This comprehensive guide delineates the evidence-based protocols for managing flexor tendon injuries in children and traumatic flexor tendon ruptures, providing practicing consultants, fellows, and residents with a masterclass in surgical decision-making, operative technique, and postoperative care.
FLEXOR TENDON INJURIES IN CHILDREN
Unique Pediatric Challenges
The management of injured flexor tendons in children younger than 10 years old is notoriously difficult and technically demanding. While the fundamental biomechanical and biological principles of tendon healing remain identical to those in adults, the pediatric patient presents a triad of unique challenges:
1. Miniature Anatomy: The diminutive caliber of pediatric tendons requires microsurgical precision, specialized instrumentation, and finer suture materials (e.g., 5-0 or 6-0 core sutures).
2. Hypertrophic Healing Response: Children possess a robust fibroblastic response, increasing the propensity for dense, restrictive peritendinous adhesions.
3. Absolute Lack of Compliance: Postoperative rehabilitation protocols, which rely heavily on controlled passive motion and patient cooperation, are nearly impossible to execute reliably in toddlers and young children.
Clinical Pearl: The success of flexor tendon repair is as dependent on postoperative rehabilitation as it is on surgical execution. In the pediatric population, the inability to comply with early active or controlled passive motion protocols significantly alters the risk-benefit ratio of immediate primary repair.
Timing of Surgery and Delayed Reconstruction
Due to the profound challenges associated with postoperative immobilization and rehabilitation in infants and toddlers, a subset of hand surgeons advocates for delaying flexor tendon surgery until the child reaches 3 to 4 years of age.
The rationale for this delayed approach includes:
* Improved Technical Repair: Larger anatomical structures allow for a more robust, multi-strand core suture technique.
* Enhanced Cooperation: A 4-year-old is significantly more capable of participating in modified controlled motion protocols than an infant.
* Predictable Outcomes: The results following flexor tenolysis tend to be markedly better in older children who can actively participate in their recovery.
However, delaying surgery is not without consequence. Retracted tendons undergo myostatic contracture, and the native flexor sheath may collapse or obliterate. Should tendon grafting be required during delayed reconstruction, the surgeon must be acutely aware that the sources of autologous tendon grafts (e.g., palmaris longus, plantaris) are limited and anatomically variable in the pediatric population.
BIOMECHANICS OF TENDON HEALING AND CONTROLLED MOTION
To mitigate adhesion formation while protecting the fragile anastomosis, controlled passive motion is paramount. The biomechanical goal is to achieve sufficient tendon excursion to prevent the tendon from adhering to the surrounding fibro-osseous sheath, without applying tension that exceeds the tensile strength of the repair.

Controlled Passive Motion Dynamics:
* Metacarpophalangeal (MCP) Joint Positioning: The MCP joint should remain in a normal, balanced, flexed position to remove tension from the flexor system.
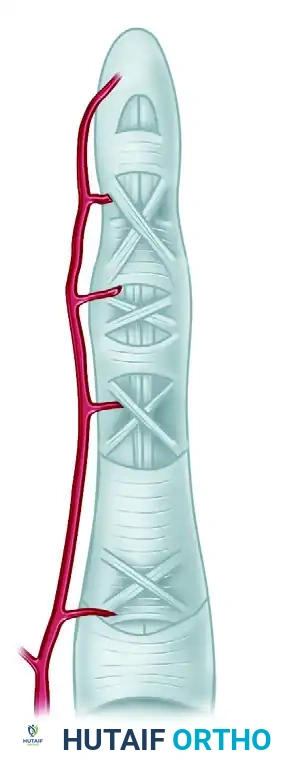
* Distal Interphalangeal (DIP) Joint Extension: Extension of the DIP joint alone is sufficient to move the flexor digitorum profundus (FDP) anastomosis 3 to 5 mm. This excursion is the critical threshold required to prevent dense adhesion formation.
* Differential Glide: During isolated DIP motion, note the distal migration of the FDP anastomosis away from the flexor digitorum sublimis (FDS) tendon.

- Proximal Interphalangeal (PIP) Joint Extension: When the middle phalanx is extended (with the MCP flexed), both the FDS and FDP anastomoses glide distally. This moves the repair sites away from fixed structures (such as the A2 pulley) that may have been injured during the initial trauma. Elastic traction is then utilized to safely return the finger to its original resting flexed position.
TRAUMATIC FLEXOR TENDON RUPTURES
Epidemiology and Pathoanatomy
Although closed ruptures of flexor tendons are less common than their extensor counterparts (e.g., mallet finger), they are a well-documented entity that is frequently misdiagnosed in the acute setting.
The most common presentation is the avulsion of the flexor digitorum profundus (FDP) at its insertion on the distal phalanx, colloquially known as "Jersey Finger." This injury predominantly affects athletes and occurs when a forcefully flexing digit is subjected to a sudden, violent extension force (eccentric load)—such as a player grabbing an opponent's jersey as they pull away.
Key Epidemiological Factors:
* Digit Involvement: The ring finger is involved in approximately 75% of cases. This is due to the anatomical tethering of the ring finger FDP by a common muscle belly and the lumbrical anatomy, which limits its independent extension, subjecting it to greater force during forced extension.
* Demographics: These ruptures occur most frequently in men in their 20s and 30s.
* Associated Conditions: Approximately 20% of non-traumatic or low-energy ruptures may be associated with underlying tenosynovitis or inflammatory arthropathies.
* Intratendinous Ruptures: While rare, mid-substance intratendinous ruptures of the FDP can occur in individuals performing activities that require forceful, sustained flexion against high resistance (e.g., rock climbing). Ruptures of the FDS or the flexor pollicis longus (FPL) are exceedingly rare but follow similar biomechanical principles.
Clinical Presentation and Diagnosis
A high index of suspicion is required, as the initial presentation can be deceptively benign.
* The "Missed" Diagnosis: Frequently, a patient’s primary complaint is a tender mass in the palm or volar finger, without an immediate awareness of the loss of isolated DIP joint flexion. The mass represents the retracted, coiled end of the avulsed tendon.
* Physical Examination: The hallmark sign is the inability to actively flex the DIP joint while the PIP joint is held in extension.
* Imaging:
* Radiographs: Standard AP and lateral views are mandatory. They may reveal a small bony avulsion or a larger intra-articular fracture fragment retracted proximally.
* MRI: Magnetic Resonance Imaging is highly sensitive and specific for defining the exact level of tendon retraction, the integrity of the vincula, and the presence of intratendinous ruptures.
Surgical Warning: Never assume an intact FDP based solely on a patient's ability to make a fist. The intact FDS will flex the PIP joint, and the intrinsic muscles will flex the MCP joint, masking the FDP avulsion if isolated DIP testing is not meticulously performed.
CLASSIFICATION OF FDP AVULSIONS (LEDDY AND PACKER)
The treatment strategy and prognosis are heavily dictated by four critical factors:
1. The length of time between injury and treatment.
2. The anatomical level to which the tendon has retracted.
3. The status of the blood supply to the avulsed tendon (vincula integrity).
4. The presence and size of bony fragments.
Based on these factors, FDP avulsions are classified into three primary types:
Type 1: Retraction into the Palm
- Pathoanatomy: The tendon avulses without a bone fragment and retracts completely into the palm, where it is held by the origin of the lumbrical muscle. Both the vinculum longum and vinculum breve are ruptured.
- Vascularity: The tendon end is entirely avascular.
- Prognosis: Poor if delayed. The tendon rapidly undergoes myostatic contracture, and the end becomes kinked, softened, and swollen.
Type 2: Retraction to the PIP Joint
- Pathoanatomy: The tendon retracts only to the level of the proximal interphalangeal (PIP) joint. It is prevented from further proximal migration by an intact vinculum longum or a small fragment of bone catching on the A3 or A2 pulley.
- Vascularity: Blood supply is preserved via the intact vinculum.
- Prognosis: Good. Because circulation is maintained and contracture is limited, these can often be repaired successfully even weeks or months after the initial injury.
Type 3: Bony Avulsion
- Pathoanatomy: A large bony fragment is avulsed with the tendon. The fragment is usually too large to pass through the A4 pulley, trapping the tendon at the level of the distal interphalangeal (DIP) joint or middle phalanx.
- Vascularity: Blood supply is highly preserved.
- Prognosis: Excellent. The fragment may be comminuted or noncomminuted, intra-articular or non-articular. Reattachment is highly successful even in delayed presentations.
SURGICAL MANAGEMENT AND TECHNIQUES
Direct repair, tendon grafting, or tendon transfer are the mainstays of treatment. The choice of procedure is dictated by the injury type and chronicity.
Management of Type 1 Injuries (Acute)
For Type 1 injuries, primary reinsertion into the distal phalanx is strongly recommended, provided the injury is detected within a strict window of 7 to 10 days. Beyond this period, the avascular distal end becomes structurally compromised (kinked and softened), prohibiting safe delivery through the fibro-osseous sheath and secure attachment to bone.
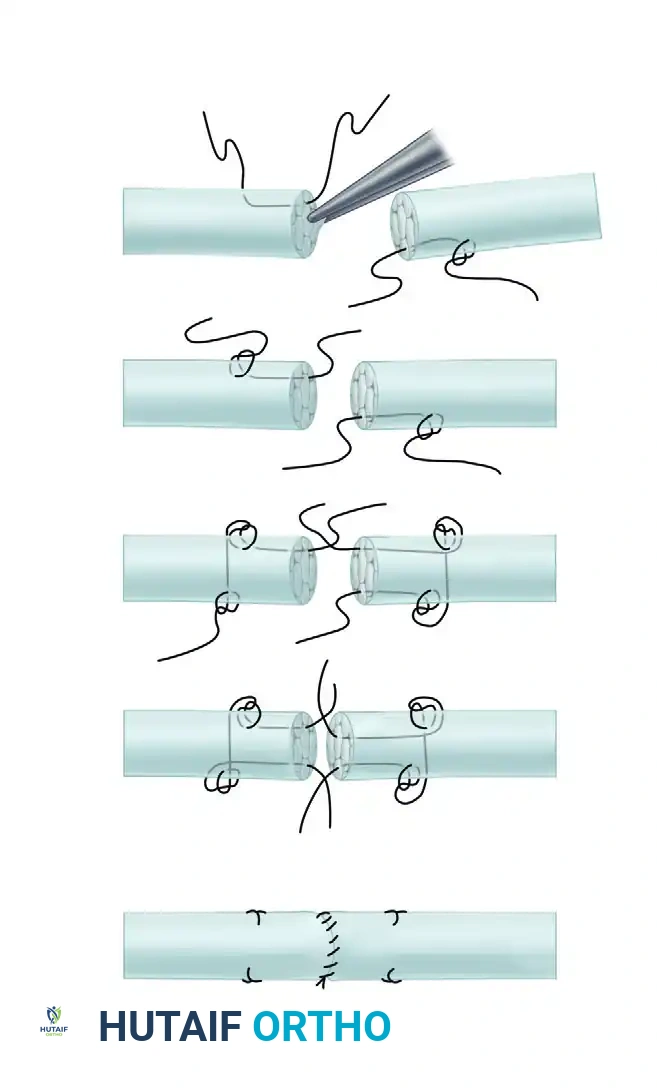
Step-by-Step Surgical Technique:
1. Incision: A midlateral or volar oblique (Bruner) incision is utilized over the affected digit.
2. Sheath Exposure: The flexor sheath is carefully opened through a transverse incision just distal to the A2 pulley. If the tendon end is absent in this window, retraction into the palm is confirmed.
3. Palmar Retrieval: A separate transverse incision is made near the distal palmar crease to expose the flexor sheath proximal to the A1 pulley. The retracted tendon end is located here.
4. Antegrade Passage: Passing the swollen tendon back through the intact pulley system (A1, A2, A3, A4) without causing iatrogenic trauma is critical.
* Technique: Use the retrograde passage of a pediatric feeding tube (or intravenous tubing) from the distal incision to the palmar incision. The tendon end is sutured to the tubing, allowing smooth, atraumatic antegrade passage of the tendon back into the finger. Alternatively, flexible wire loops can be used.
5. Distal Reattachment: The tendon is attached to its anatomical footprint on the distal phalanx using a pull-out wire technique. An antegrade pull-out wire is preferred over the traditional retrograde Bunnell technique to minimize trauma to the sterile matrix of the nail bed. The wire is secured over a button on the dorsal aspect of the fingertip.
Management of Type 2 and Type 3 Injuries
- Type 2 Injuries: Because the vinculum longum is intact and circulation is maintained, Type 2 injuries can be repaired at a significantly later time than Type 1 injuries. Literature demonstrates satisfactory primary repairs even several months post-injury, provided the tendon remains pliable.
- Type 3 Injuries: The presence of a bony fragment simplifies the repair. The fragment is reduced into its fracture bed on the distal phalanx and secured. Depending on fragment size, fixation can be achieved with a pull-out wire, mini-screws, or K-wires. Early passive motion is encouraged.
Salvage Procedures for Late Presentations
If a Type 1 injury presents late (>10-14 days), or if any delayed presentation reveals a tendon that cannot be safely advanced to the distal phalanx without excessive tension (resulting in a flexion contracture), primary repair must be abandoned.
Options include:
1. Flexor Tendon Grafting: In young, highly motivated, and cooperative patients (particularly in the 10- to 20-year age range), a free tendon graft (e.g., palmaris longus) can be routed through the intact FDS tendon in the index, long, or ring fingers.
2. DIP Joint Arthrodesis: For patients requiring absolute stability and grip strength (e.g., manual laborers), fusing the DIP joint in slight flexion (10-15 degrees) provides a highly functional, pain-free digit.
3. Tenodesis: Anchoring the distal stump of the FDP to the middle phalanx to provide a stable post for pinch.
POSTOPERATIVE REHABILITATION PROTOCOLS
The postoperative protocol is as critical as the surgical repair. For primary reinsertions utilizing a pull-out wire, the following regimen is standard:
- Immobilization Phase: The limb is placed in a dorsal blocking splint immediately postoperatively.
- Wrist: 20 to 30 degrees of flexion.
- MCP Joints: 70 to 80 degrees of flexion.
- IP Joints: Full extension (to prevent PIP flexion contractures).
- Duration: The splint and the pull-out wire are left in place for 3 to 4 weeks.
- Early Passive Motion: Duran and Houser popularized the concept of early passive motion to prevent adhesions.
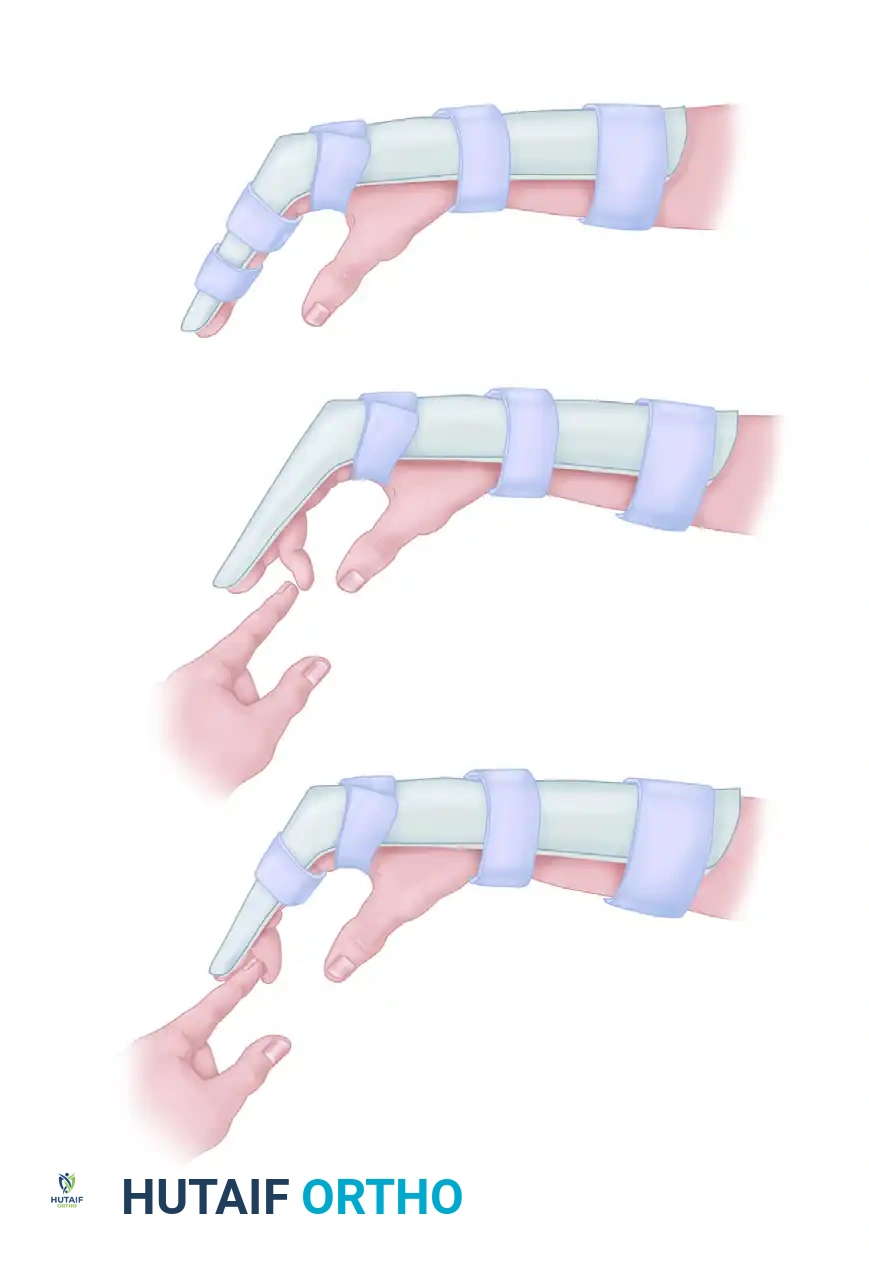
As illustrated above, passive flexion of the interphalangeal joints is performed several times each day for 4 to 5 weeks. The patient uses their uninjured hand to passively flex the PIP and DIP joints within the constraints of the dorsal blocking splint, ensuring tendon glide without active contractile tension.
COMPLICATIONS: POSTREPAIR RUPTURE AND TENOLYSIS
Postrepair Rupture
Tendon rupture following primary repair is a devastating complication, usually occurring between days 10 and 21 when the tendon is at its weakest (the fibroblastic phase of healing) and the patient begins to feel more confident in their motion.
- Acute Detection: If the rupture is detected promptly (within days), the finger should be re-explored. If the tendon ends are viable, a direct end-to-end re-repair can yield satisfactory results.
- Delayed Detection: If detection is delayed, the tendon ends rapidly degenerate and retract. End-to-end repair is rarely possible, and two-stage tendon graft reconstruction (utilizing a silicone Hunter rod) is usually required.
Rupture Following Tenolysis
Tenolysis is performed to release restrictive adhesions in a healed but stiff tendon. However, aggressive tenolysis can devascularize the tendon or mechanically weaken it, leading to spontaneous rupture during postoperative rehabilitation.
Surgical Pitfall: If a tendon ruptures in a densely scarred, previously operated bed (post-tenolysis), satisfactory function after re-exploration and direct repair is highly unlikely. The biological environment is hostile to primary healing. In these scenarios, the surgeon should immediately pivot to delayed tendon grafting or arthrodesis.
Flexor Pollicis Longus (FPL) Considerations
The FPL is unique due to its single tendon anatomy and powerful muscle belly. When an FPL rupture is seen early, it can be successfully reattached to the distal phalanx. However, when seen late, the FPL muscle belly undergoes rapid and severe myostatic shortening, accompanied by tendon degeneration. In late FPL ruptures, primary repair is impossible, and a tendon graft or FDS tendon transfer is necessary to restore thumb interphalangeal flexion.
📚 Medical References
- Flexor tendon ruptures in rheumatoid arthritis, Hand Clin 5:177, 1989.
- Ertel AN, Millender LH, Nalebuff E, et al: Flexor tendon ruptures in patients with rheumatoid arthritis, J Hand Surg 13A:860, 1988.
- Ferlic DC: Boutonniere deformities in rheumatoid arthritis, Hand Clin 5:215, 1989.
- Gibbons CER, Gosal HS, Choudri AH, et al: Trapeziectomy for basal thumb joint osteoarthritis: 3to 19-year follow-up, Int Orthop 23:216, 1999.
- Hartigan BJ, Stern PJ, Kiefhaber TR: Thumb carpometacarpal osteoarthritis: arthrodesis compared with ligament reconstruction and tendon interposition, J Bone Joint Surg 83A:1470, 2001.
- Kiefhaber TR, Strickland JW: Soft tissue reconstruction for rheumatoid swan-neck and boutonniere deformities: long-term results, J Hand Surg 18A:984, 1993.
- King JA, Tomaino MT: Surgical treatment of the rheumatoid thumb, Hand Clin 17:275, 2001.
- King PJ, Eglseder WA, House HO: Extensor carpi radialis longus tendinoplasty for thumb basal joint arthritis, Am J Orthop 30:213, 2001.
- Lane LB, Henley DH: Ligament reconstruction of the painful, unstable, nonarthritic thumb carpometacarpal joint, J Hand Surg 26A:686, 2001.
- Leslie BM: Rheumatoid extensor tendon ruptures, Hand Clin 5:191, 1989.
- Manueddu CA, Bogoch ER, Hastings DE: Restoration of metacarpophalangeal extension of the thumb in infl ammatory arthritis, J Hand Surg 21B:633, 1996.
- Melone CP Jr, Taras JS:
You Might Also Like