Dupuytren Contracture: Prognosis, Indications, and Surgical Techniques
Key Takeaway
Dupuytren contracture management requires a nuanced understanding of disease histopathology, prognostic factors, and precise surgical execution. Operative intervention is typically indicated for metacarpophalangeal joint contractures exceeding 30 degrees or any proximal interphalangeal joint contracture causing functional impairment. Surgical options range from percutaneous needle aponeurotomy to partial fasciectomy and dermatofasciectomy. Meticulous dissection, particularly regarding neurovascular bundle displacement by spiral cords, is paramount to minimizing complications and optimizing postoperative functional recovery.
INTRODUCTION TO DUPUYTREN DISEASE
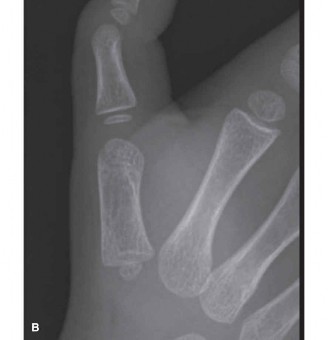
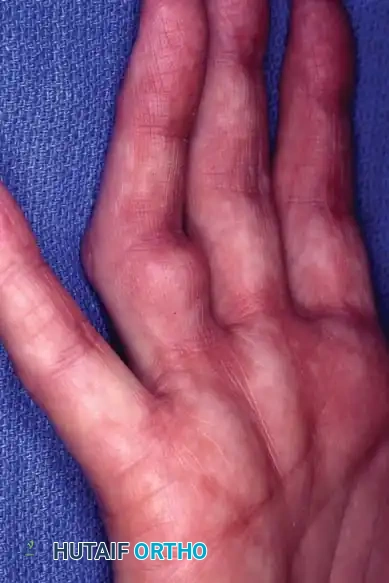
Dupuytren disease is a benign, fibroproliferative disorder of the palmar fascia characterized by the progressive formation of nodules and cords, ultimately leading to debilitating flexion contractures of the digits. The fundamental histopathology involves the transformation of normal fascial fibroblasts into myofibroblasts, which exhibit aggressive contractile properties.
Successful management of Dupuytren contracture requires the orthopedic surgeon to possess a profound understanding of palmar fascial anatomy, the biomechanical behavior of pathological cords, and the patient-specific prognostic factors that dictate the timing and extent of surgical intervention.
PROGNOSIS AND RISK FACTORS (DUPUYTREN'S DIATHESIS)
The prognosis in Dupuytren contracture is highly variable. The concept of "Dupuytren's diathesis" refers to a constellation of constitutional risk factors that predispose a patient to aggressive disease, early onset, and a high rate of postoperative recurrence. The extent of any surgical operation must be tailored to these prognostic indicators.
- Heredity: A strong family history, particularly with an autosomal dominant inheritance pattern and variable penetrance, indicates that the lesion is likely to progress rapidly. Early age of onset in these patients is a hallmark of aggressive disease.
- Sex: The lesion typically begins later in life and progresses more slowly in women, who often accommodate better to the resulting deformity. However, Zemel et al. demonstrated that long-term surgical outcomes are paradoxically worse in women than in men, with postoperative flare reactions (including complex regional pain syndrome) being twice as likely.
- Epilepsy: Historically, earlier literature positively associated Dupuytren contracture with epilepsy and the use of barbiturates. However, contemporary epidemiological studies by Geoghegan et al. have conclusively debunked this, showing that neither epilepsy nor antiepileptic medications are independently associated with the disease.
- Diabetes Mellitus: Diabetes is a well-established risk factor. The severity correlates with the medical management required; patients requiring insulin therapy are significantly more likely to develop Dupuytren disease compared to those managed with diet, metformin, or sulfonylureas.
- Alcoholism and Smoking: Microvascular ischemia and free radical damage play a role in pathogenesis. Godtfredsen et al. found a strict dose-dependent relationship between alcohol intake, smoking, and disease severity. The combination of these two factors conveys a remarkably high risk for rapid progression and postoperative recurrence.
- Location and Extent of Disease: Bilateral hand involvement, especially when associated with ectopic fibroproliferative deposits such as knuckle pads (Garrod's nodes) or plantar fibromatosis (Ledderhose disease), signifies a severe diathesis. Progression is characteristically more rapid on the ulnar side of the hand (ring and small fingers).
- Behavior of Disease: The historical biological behavior of the disease in a given patient—whether previously treated or observed—is the most reliable predictor of its future clinical course.

INDICATIONS FOR TREATMENT
Nonoperative Management
Nonoperative treatment regimens focus on altering the fundamental histopathology of the myofibroblast. Bisson et al. demonstrated that nodule-derived fibroblasts possess significantly greater contractile properties and force generation than cord-derived or normal carpal tunnel ligament fibroblasts.
- Corticosteroid Injections: Ketchum and Donahue studied the modification of Dupuytren nodules via intralesional injection of triamcinolone acetonide. After an average of 3.2 injections per nodule, 97% of hands exhibited softening or flattening. While complete resolution is rare, nodule reactivation was delayed by up to 3 years in 50% of patients.
- Clostridial Collagenase Injections: Enzymatic fasciotomy using Clostridium histolyticum collagenase has revolutionized nonoperative care. Badalamente et al. established the safety and efficacy of this enzymatic degradation, which provides prompt metacarpophalangeal (MCP) and proximal interphalangeal (PIP) joint contracture release.
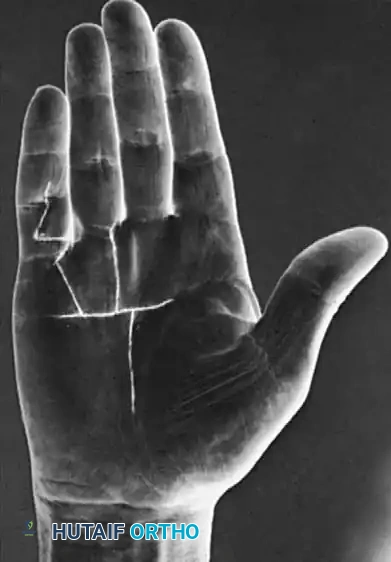
Operative Indications and Timing
In the absence of contractures, surgical treatment is not indicated. Painless palmar nodules alone do not warrant surgery unless they cause severe discomfort, skin pitting, or maceration.
Clinical Pearl: Timing is critical. Operating during the active, highly cellular "proliferative stage" often results in ill-defined surgical planes, massive postoperative flare reactions, and paradoxical stiffening. Ideally, surgery is delayed until the disease matures into the "residual phase" (dense, avascular cords).
Standard Surgical Indications:
* MCP Joint Contracture: ≥ 30 degrees.
* PIP Joint Contracture: ≥ 15 to 20 degrees (PIP contractures are notoriously difficult to correct and rapidly become disabling).
SURGICAL OPTIONS AND PREOPERATIVE PLANNING
The appropriate procedure depends on the degree of contracture, the nutritional status of the palmar skin, the presence of joint subluxation, and the patient’s diathesis.
The five primary surgical procedures are:
1. Subcutaneous Fasciotomy
2. Partial (Selective) Fasciectomy
3. Complete Fasciectomy (Largely abandoned due to high complication rates)
4. Dermatofasciectomy with Skin Grafting
5. Amputation or Arthrodesis
Incision Design and Exposure
Exposure is paramount. The incision must allow full visualization of the neurovascular bundles while preventing postoperative scar contracture.
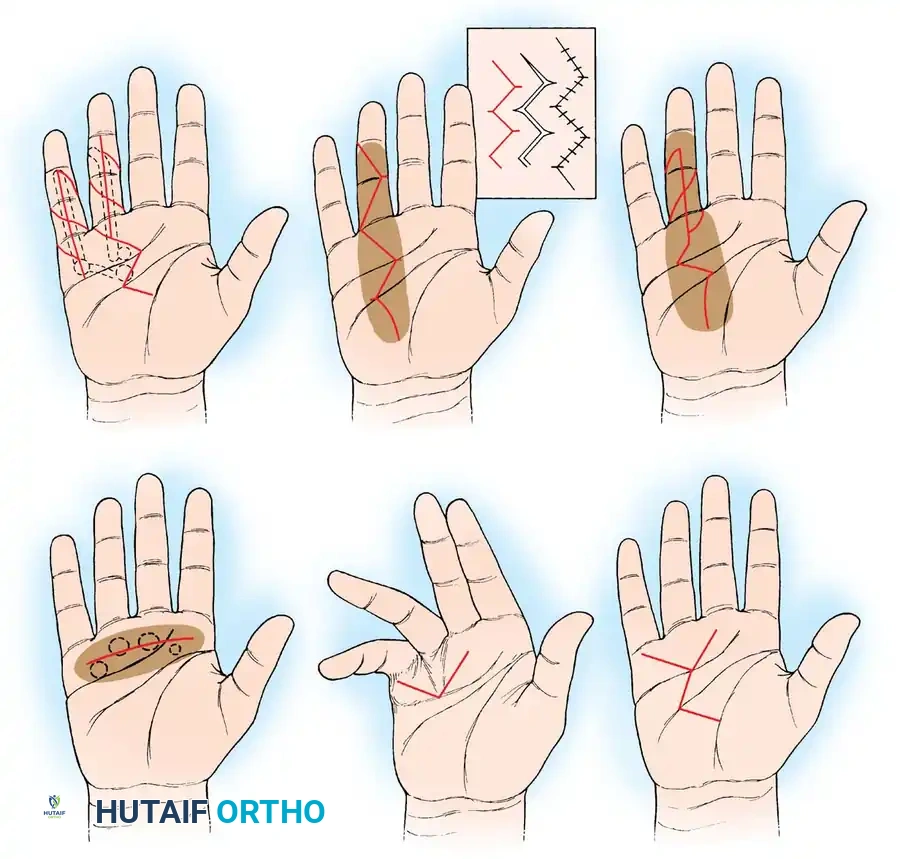
- Zigzag (Bruner) Incisions: Preferred for digital exposure. They prevent linear scar contracture but must be designed so the apices do not cross flexion creases at right angles.
- Z-Plasties: Used to lengthen contracted palmar skin. A midline longitudinal incision can be converted into multiple Z-plasties during closure.
- Transverse Incisions: Useful in the palm when the disease is well-defined and can be safely dissected from the neurovascular bundles.
SURGICAL TECHNIQUES
1. Subcutaneous Fasciotomy (Luck Technique)
This minimally invasive procedure is primarily indicated for elderly patients, those with severe medical comorbidities, or as a preliminary staging procedure for severe contractures. It is most effective for isolated, mature pretendinous cords causing MCP joint flexion.
Surgical Warning: Subcutaneous fasciotomy is strictly contraindicated for laterally placed digital cords due to the high risk of digital nerve transection. It should only be performed for centrally located palmar cords.
Technique:
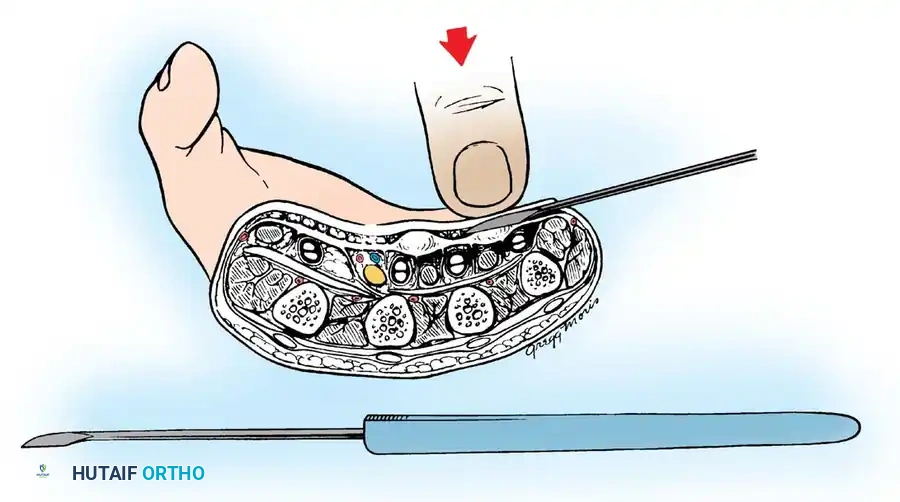
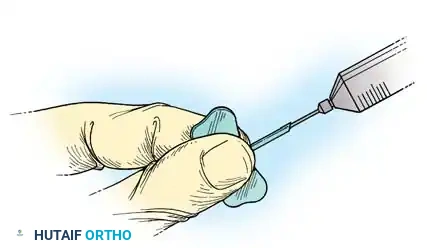
1. Using a pointed scalpel (e.g., #11 blade), make small puncture wounds on the ulnar side of the diseased palmar fascia at three levels: distal to the palmar fascia apex, near the proximal palmar crease, and at the distal palmar crease.
2. Insert a small tenotomy knife or a Luck fasciotome (resembling a myringotome) through the puncture wounds. The blade must remain parallel to the palm, passing beneath the skin but superficial to the fascia.
3. Extend the fingers to place the fascial cord under maximal tension.
4. Turn the blade edge toward the palmar fascia and divide the cord using gentle pressure or a rocking motion. Never use a sawing motion.
5. A sudden loss of gritty resistance indicates complete division of the cord.
6. Undermine the corrugated skin using the blade parallel to the dermis to release skin tethering.
2. Partial (Selective) Fasciectomy
This is the gold standard and most frequently performed operation. It excises only the macroscopically diseased fascia, balancing adequate release with minimized surgical morbidity.
Technique:
1. Preparation: Exsanguinate the limb and inflate the pneumatic tourniquet. Loupe magnification (minimum 2.5x to 3.5x) is mandatory.
2. Incision: Outline a Bruner zigzag incision over the digit, extending into the palm. Incorporate skin pits into the flap design so they can be excised or released.
3. Flap Elevation: Elevate full-thickness skin flaps. The dissection plane is immediately deep to the dermis. Great care must be taken to preserve the subdermal vascular plexus to prevent flap necrosis.
4. Neurovascular Identification: This is the most critical step. The normal anatomy is distorted by the pathological cords.
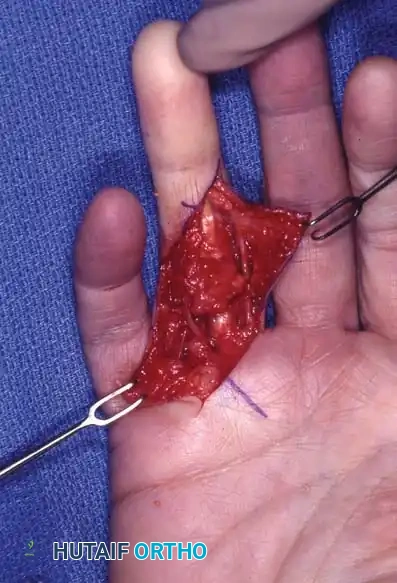
The Spiral Cord Biomechanics: The spiral cord (formed by the pretendinous band, spiral band, lateral digital sheet, and Grayson's ligament) wraps around the neurovascular bundle. As it contracts, it displaces the digital nerve centrally, superficially, and proximally. The nerve is at extreme risk of transection at the level of the natatory ligament.
- Dissection: Identify the neurovascular bundle in the proximal palm where the anatomy remains normal. Trace it distally into the diseased tissue.
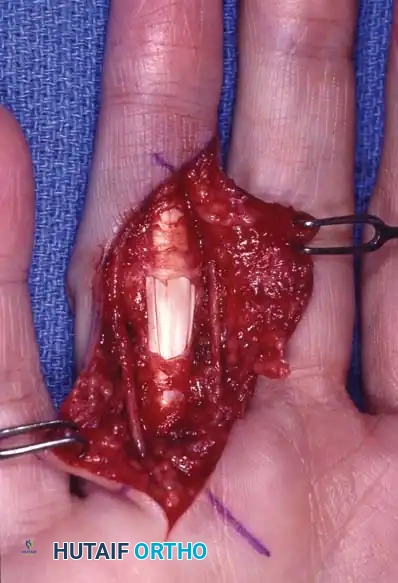
- Excision: Carefully dissect the diseased cord away from the nerve and artery. Retract the neurovascular bundle safely and excise the cord in its entirety.
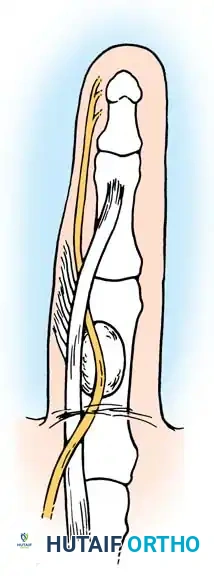
- Skoog's Modification: Skoog emphasized that a definitive cleavage plane exists between the longitudinal pretendinous fibers and the transverse palmar ligament. The transverse palmar ligament is rarely involved in Dupuytren's and should be preserved to protect the underlying neurovascular structures.
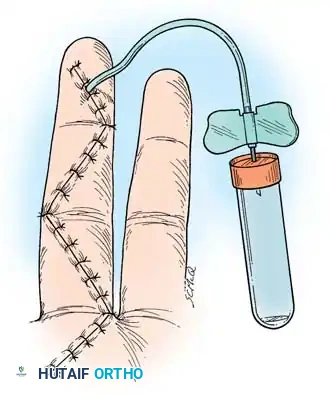
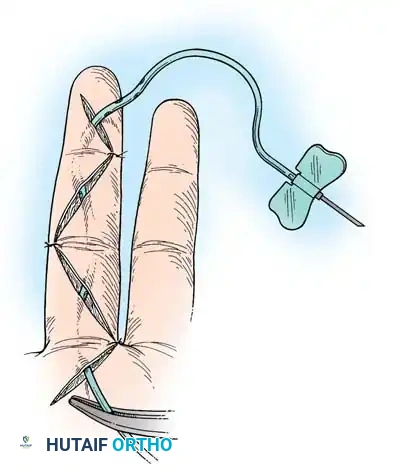
Management of the Stiff PIP Joint
Following fasciectomy, residual PIP joint flexion deformity is common. This is often due to isolated cords passing volar to the rotation axis of the PIP joint, inserting onto the flexor tendon sheath or middle phalanx.
If excision of all fascial cords (including the retrovascular cords of Thomine) fails to correct the contracture, a volar joint capsulotomy may be required. However, Weinzweig et al. demonstrated that PIP contractures >60 degrees are typically only correctable to about 50% of the existing deformity, regardless of capsulotomy. Furthermore, capsulotomy patients lose an average of 16 degrees of terminal flexion compared to 8 degrees in non-capsulotomy patients.
3. Dermatofasciectomy with Skin Grafting (Hueston's Procedure)
Advocated by Hueston, this procedure is indicated for young patients with a severe Dupuytren's diathesis, recurrent disease, or when the palmar skin is so intimately involved with the fascia that it cannot be salvaged.
Technique:
The diseased fascia and the overlying involved skin are excised en bloc. The defect is covered with a full-thickness skin graft (FTSG) or a thick split-thickness skin graft (STSG). Recurrence beneath a skin graft is exceptionally rare, as the graft appears to act as a biological "firebreak" against myofibroblast proliferation.
4. Salvage Procedures: Amputation and Arthrodesis
In cases of severe, neglected PIP joint contractures (e.g., >90 degrees in the small finger) where the digit is non-functional and interferes with daily activities, salvage procedures are indicated.
- Amputation: A ray amputation or amputation through the PIP joint may be necessary. The skin from the filleted finger can be preserved as a vascularized pedicle flap to cover large palmar skin defects.
- Arthrodesis: Resection of the contracted PIP joint and arthrodesis in a functional position (typically 30-40 degrees of flexion) shortens the finger, relieving tension on the neurovascular bundles, and permanently eliminates the risk of recurrent contracture.
CLOSURE AND POSTOPERATIVE PROTOCOL
Meticulous hemostasis is mandatory before closure. Hematoma formation is the most common complication and serves as a potent stimulus for excessive scarring, flap necrosis, and severe flare reactions.
- Deflate the tourniquet and achieve pinpoint hemostasis using bipolar electrocautery.
- If a transverse palmar incision was used, it may be left open to heal by secondary intention (McCash open palm technique) to allow hematoma drainage and prevent skin tension.
- Close zigzag incisions with interrupted non-absorbable sutures.
- Insert a small closed-suction drain or a passive capillary drain if dead space is significant.
Rehabilitation:
* Apply a bulky, non-compressive soft dressing with a volar plaster splint holding the MCP joints in extension and the PIP joints in extension (or slight flexion if neurovascular tension is a concern).
* Elevate the hand strictly for 48-72 hours.
* Initiate active and passive range-of-motion exercises within 3 to 5 days postoperatively under the guidance of a certified hand therapist.
* Night splinting in extension is maintained for 3 to 6 months to counteract the physiological forces of scar contracture.
You Might Also Like